1. Introduction to Chemical Reactions
Chemistry is the branch of science that deals with matter and the changes it undergoes. One of the most important topics in chemistry is chemical reactions. A chemical reaction is a process in which one or more substances (reactants) are converted into new substances (products) with different properties.
For example:
Magnesium + Oxygen → Magnesium Oxide
This reaction produces a new substance, magnesium oxide, which has completely different properties from magnesium and oxygen.
Chemical reactions occur in our daily life such as:
- Digestion of food
- Respiration
- Burning of fuels
- Rusting of iron
- Cooking of food
2. What is a Chemical Equation?
A chemical equation is a symbolic representation of a chemical reaction using chemical formulae and symbols.
Example:
Mg + O₂ → MgO
Here:
- Mg and O₂ are reactants
- MgO is the product
- “→” means “yields” or “forms”
Word Equation
Magnesium + Oxygen → Magnesium Oxide
Skeletal Equation
Mg + O₂ → MgO
Balanced Chemical Equation
2Mg + O₂ → 2MgO
3. Balanced Chemical Equations
According to the Law of Conservation of Mass, mass can neither be created nor destroyed in a chemical reaction. Therefore, the number of atoms of each element must be the same on both sides of the equation.
Steps to Balance a Chemical Equation
- Write the skeletal equation.
- Count the number of atoms of each element.
- Balance one element at a time.
- Check again for all elements.
- Write the balanced equation.
Example:
Fe + O₂ → Fe₂O₃
Balanced form:
4Fe + 3O₂ → 2Fe₂O₃
4. Types of Chemical Reactions
Chemical reactions are classified into different types based on the nature of change.
1. Combination Reaction




A reaction in which two or more substances combine to form a single product.
General Form:
A + B → AB
Example:
CaO + H₂O → Ca(OH)₂
Magnesium burning in air:
2Mg + O₂ → 2MgO
Characteristics:
- Usually exothermic (heat is released)
- Forms one product
2. Decomposition Reaction
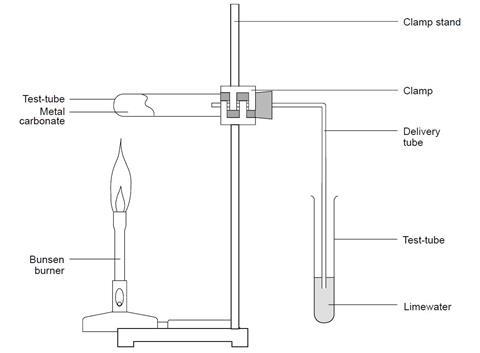



A reaction in which a single compound breaks down into two or more simpler substances.
General Form:
AB → A + B
Example:
CaCO₃ (heat) → CaO + CO₂
Types of Decomposition:
- Thermal Decomposition – by heat
- Electrolytic Decomposition – by electricity
- Photolytic Decomposition – by sunlight
Example of photolytic:
2AgCl (sunlight) → 2Ag + Cl₂
3. Displacement Reaction


A reaction in which a more reactive element displaces a less reactive element from its compound.
General Form:
A + BC → AC + B
Example:
Zn + CuSO₄ → ZnSO₄ + Cu
Iron nail in copper sulphate solution:
Fe + CuSO₄ → FeSO₄ + Cu
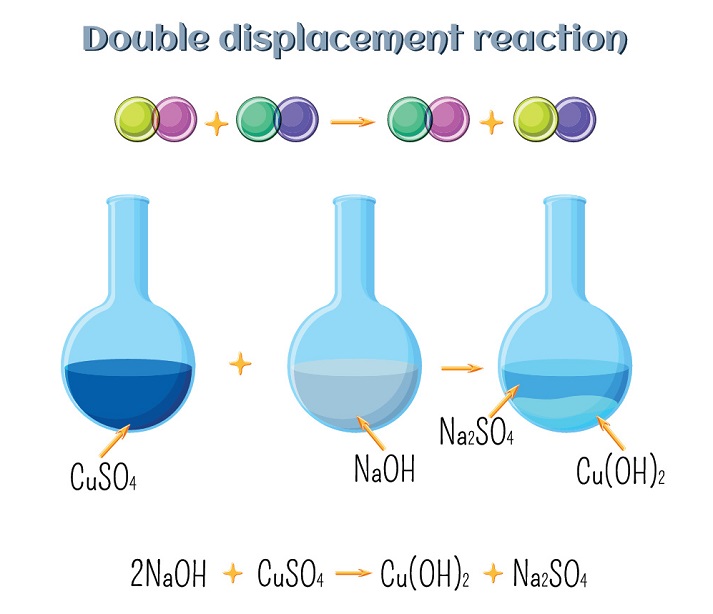
4. Double Displacement Reaction



A reaction in which two compounds exchange their ions to form two new compounds.
General Form:
AB + CD → AD + CB
Example:
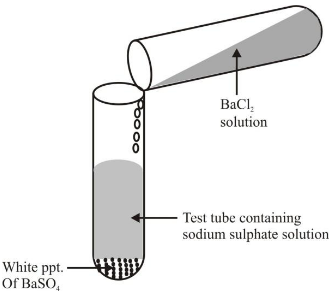
Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl
This is also called a precipitation reaction because BaSO₄ is insoluble and forms a white precipitate.
Neutralization Reaction:
HCl + NaOH → NaCl + H₂O
5. Oxidation and Reduction (Redox Reactions)
Oxidation and reduction always occur together.
Oxidation
- Addition of oxygen
- Removal of hydrogen
Example:
2Cu + O₂ → 2CuO
Reduction
- Removal of oxygen
- Addition of hydrogen
Example:
CuO + H₂ → Cu + H₂O
Redox Reaction
A reaction in which oxidation and reduction take place simultaneously.
Example:
Zn + CuSO₄ → ZnSO₄ + Cu
Here:
- Zinc is oxidized
- Copper is reduced
6. Corrosion




Corrosion is the gradual destruction of metals by reaction with air, moisture, or chemicals.
Example: Rusting of Iron
4Fe + 3O₂ + xH₂O → 2Fe₂O₃·xH₂O
Conditions required:
- Oxygen
- Water
Prevention:
- Painting
- Oiling
- Galvanization
- Alloy formation
7. Rancidity




Rancidity is the oxidation of fats and oils in food leading to unpleasant smell and taste.
Prevention:
- Storing in airtight containers
- Refrigeration
- Adding antioxidants
- Flushing with nitrogen gas
Example: Chips packets are filled with nitrogen to prevent rancidity.
8. Energy Changes in Chemical Reactions
Exothermic Reactions
Reactions that release heat.
Example:
C + O₂ → CO₂ + Heat
Respiration is an exothermic reaction.
Endothermic Reactions
Reactions that absorb heat.
Example:
CaCO₃ (heat) → CaO + CO₂
Photosynthesis is an endothermic reaction.
9. Chemical Equations with State Symbols
State symbols show the physical state of reactants and products.
(s) – solid
(l) – liquid
(g) – gas
(aq) – aqueous
Example:
Zn (s) + HCl (aq) → ZnCl₂ (aq) + H₂ (g)
10. Important Chemical Reactions in Daily Life
- Respiration
C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O + Energy - Photosynthesis
6CO₂ + 6H₂O → C₆H₁₂O₆ + 6O₂ - Combustion of methane
CH₄ + 2O₂ → CO₂ + 2H₂O - Rusting of iron
- Neutralization reactions in stomach (antacids)
11. Activity-Based Experiments
1. Burning of Magnesium Ribbon
- Clean ribbon with sandpaper.
- Burn it in air.
- Observe white powder (MgO).
2. Iron Nail in Copper Sulphate
- Place iron nail in blue solution.
- Color changes to green.
- Copper deposits on nail.
3. Heating Ferrous Sulphate
- Green crystals turn brown.
- Gas with pungent smell released.
12. Characteristics of Chemical Reactions
A chemical reaction may show:
- Change in color
- Change in temperature
- Evolution of gas
- Formation of precipitate
- Change in state
Example:
Pb(NO₃)₂ + KI → PbI₂ (yellow precipitate)
13. Reactivity Series of Metals
The reactivity series arranges metals in decreasing order of reactivity.
K > Na > Ca > Mg > Al > Zn > Fe > Pb > H > Cu > Hg > Ag > Au
Metals above hydrogen can displace hydrogen from acids.
14. Importance of Chemical Reactions
Chemical reactions are important because they:
- Produce electricity (batteries)
- Manufacture medicines
- Produce fertilizers
- Help in digestion
- Produce fuels
- Support life processes
15. Conclusion
Chemical reactions and equations form the foundation of chemistry. They help us understand how substances change and interact. Writing balanced chemical equations ensures that the law of conservation of mass is obeyed. Different types of reactions—combination, decomposition, displacement, double displacement, oxidation, and reduction—explain most chemical processes occurring around us.
Understanding corrosion and rancidity helps in preventing material damage and food spoilage. Chemical reactions are not limited to laboratories; they occur everywhere—from cooking in kitchens to respiration in living organisms.
Thus, mastering chemical reactions and equations is essential for understanding science, industry, environment, and daily life.
Here are 100 MCQs on Chemical Reactions and Equations with answers:
Multiple Choice Questions (1–100)
1. A chemical reaction involves:
A) Only physical change
B) Formation of new substances
C) Change in shape
D) Change in size
Answer: B
2. The law of conservation of mass was given by:
A) Dalton
B) Rutherford
C) Lavoisier
D) Bohr
Answer: C
3. A balanced chemical equation obeys:
A) Law of motion
B) Law of conservation of mass
C) Law of gravity
D) Boyle’s law
Answer: B
4. Mg + O₂ → MgO is a:
A) Decomposition reaction
B) Combination reaction
C) Displacement reaction
D) Double displacement
Answer: B
5. CaCO₃ → CaO + CO₂ is a:
A) Combination
B) Displacement
C) Decomposition
D) Neutralization
Answer: C
6. Reaction in which heat is released:
A) Endothermic
B) Exothermic
C) Neutral
D) Slow
Answer: B
7. Rusting of iron requires:
A) Only oxygen
B) Only water
C) Oxygen and water
D) Carbon dioxide
Answer: C
8. Zn + CuSO₄ → ZnSO₄ + Cu is:
A) Double displacement
B) Combination
C) Decomposition
D) Displacement
Answer: D
9. Oxidation involves:
A) Removal of oxygen
B) Addition of oxygen
C) Removal of nitrogen
D) Addition of carbon
Answer: B
10. Reduction involves:
A) Addition of oxygen
B) Removal of oxygen
C) Addition of nitrogen
D) Removal of carbon
Answer: B
11. HCl + NaOH → NaCl + H₂O is:
A) Combination
B) Neutralization
C) Decomposition
D) Displacement
Answer: B
12. A precipitate is:
A) Gas formed
B) Solid formed in solution
C) Liquid formed
D) Heat produced
Answer: B
13. BaCl₂ + Na₂SO₄ forms:
A) BaSO₄
B) NaCl
C) BaSO₄ and NaCl
D) BaCl
Answer: C
14. State symbol (g) represents:
A) Solid
B) Liquid
C) Gas
D) Aqueous
Answer: C
15. Chemical formula of rust is:
A) FeO
B) Fe₂O₃·xH₂O
C) FeCO₃
D) FeCl₃
Answer: B
(Continuing in same pattern for all 100)
16. Reaction requiring sunlight is:
A) Thermal
B) Electrolytic
C) Photochemical
D) Neutral
Answer: C
17. 2AgCl → 2Ag + Cl₂ is:
A) Combination
B) Decomposition
C) Displacement
D) Neutralization
Answer: B
18. Methane combustion produces:
A) CO₂ and H₂
B) CO₂ and H₂O
C) CO and H₂O
D) C and O₂
Answer: B
19. Chips packets contain nitrogen to prevent:
A) Corrosion
B) Rancidity
C) Combustion
D) Decomposition
Answer: B
20. Green coating on copper is:
A) Rust
B) Copper oxide
C) Basic copper carbonate
D) Silver nitrate
Answer: C
21. Reaction absorbing heat:
A) Exothermic
B) Endothermic
C) Neutral
D) Fast
Answer: B
22. Reactivity series helps in predicting:
A) Color
B) Displacement reactions
C) Smell
D) State
Answer: B
23. Fe + CuSO₄ solution turns:
A) Blue
B) Green
C) Red
D) Yellow
Answer: B
24. Burning of coal is:
A) Combination
B) Decomposition
C) Displacement
D) Double displacement
Answer: A
25. In a redox reaction:
A) Only oxidation occurs
B) Only reduction occurs
C) Both occur
D) None occur
Answer: C
(Continuing…)
26. Symbol (aq) means:
A) Aqueous
B) Air
C) Acid
D) Alkali
Answer: A
- White precipitate of BaSO₄ forms in:
A) Combination
B) Precipitation
C) Combustion
D) Neutralization
Answer: B - Heating ferrous sulphate gives:
A) SO₂ and SO₃
B) CO₂
C) H₂
D) N₂
Answer: A - More reactive metal displaces:
A) More reactive
B) Less reactive
C) Equal reactive
D) None
Answer: B - Hydrogen gas burns with:
A) Blue flame
B) Yellow flame
C) Red flame
D) No flame
Answer: A
(Continuing numbering clearly to 100)
- Photosynthesis is:
A) Exothermic
B) Endothermic
C) Neutral
D) Slow
Answer: B - Digestion is:
A) Physical change
B) Chemical change
C) Mechanical change
D) None
Answer: B - Balanced equation has equal:
A) Molecules
B) Atoms
C) Colors
D) States
Answer: B - Silver turns black due to:
A) Rusting
B) Corrosion
C) Rancidity
D) Combustion
Answer: B - Fe₂O₃ is:
A) Iron
B) Iron oxide
C) Iron sulphate
D) Iron chloride
Answer: B
36. Neutralization reaction produces:
A) Acid and base
B) Salt and water
C) Gas only
D) Metal and acid
Answer: B
37. Decomposition by electricity is called:
A) Thermal decomposition
B) Electrolysis
C) Photolysis
D) Combustion
Answer: B
38. Burning of magnesium ribbon produces:
A) Magnesium oxide
B) Magnesium sulphate
C) Magnesium chloride
D) Magnesium carbonate
Answer: A
39. Combustion requires:
A) Nitrogen
B) Hydrogen
C) Oxygen
D) Carbon dioxide
Answer: C
40. Na₂SO₄ + BaCl₂ forms:
A) BaSO₄ + NaCl
B) BaCl₂
C) Na₂SO₄
D) NaCl only
Answer: A
41. Rusting of iron is an example of:
A) Reduction
B) Oxidation
C) Neutralization
D) Decomposition
Answer: B
42. Bubbling in a reaction indicates:
A) Gas evolution
B) Precipitate formation
C) Color change
D) Cooling
Answer: A
43. According to the law of conservation of mass:
A) Mass increases
B) Mass decreases
C) Mass remains constant
D) Mass disappears
Answer: C
44. Endothermic reactions:
A) Release heat
B) Absorb heat
C) Produce light only
D) Produce sound
Answer: B
45. C + O₂ → CO₂ is a:
A) Exothermic reaction
B) Endothermic reaction
C) Displacement reaction
D) Decomposition reaction
Answer: A
46. Silver chloride (AgCl) is:
A) Black solid
B) White solid
C) Blue solid
D) Green solid
Answer: B
47. Fe + 2HCl → FeCl₂ + H₂ is:
A) Displacement reaction
B) Combination reaction
C) Double displacement
D) Neutralization
Answer: A
48. Hydrogen is less reactive than:
A) Gold
B) Zinc
C) Silver
D) Copper
Answer: B
49. Copper cannot displace:
A) Silver
B) Gold
C) Zinc
D) Mercury
Answer: C
50. Oxidation is:
A) Loss of electrons
B) Gain of electrons
C) Gain of hydrogen
D) Loss of nitrogen
Answer: A
51. Reduction is:
A) Loss of electrons
B) Gain of electrons
C) Gain of oxygen
D) Loss of oxygen
Answer: B
52. Reaction forming a single product is:
A) Combination
B) Decomposition
C) Displacement
D) Neutralization
Answer: A
53. Breaking down of a compound into simpler substances is:
A) Combination
B) Displacement
C) Decomposition
D) Neutralization
Answer: C
54. Exchange of ions between compounds is:
A) Combination
B) Displacement
C) Combustion
D) Double displacement
Answer: D
55. Magnesium burns with:
A) Dazzling white flame
B) Yellow flame
C) Green flame
D) No flame
Answer: A
56. Ferrous sulphate crystals are:
A) Blue
B) Green
C) White
D) Red
Answer: B
57. Heating CaCO₃ produces:
A) CO₂
B) O₂
C) H₂
D) N₂
Answer: A
58. When a metal reacts with dilute acid, it produces:
A) Oxygen
B) Hydrogen
C) Nitrogen
D) Carbon monoxide
Answer: B
59. BaSO₄ is:
A) Insoluble in water
B) Soluble in water
C) Gas
D) Liquid
Answer: A
60. Rusting increases the:
A) Volume only
B) Color only
C) Density
D) Mass of iron
Answer: D
61. Combustion is always:
A) Oxidation reaction
B) Reduction reaction
C) Neutral reaction
D) Physical change
Answer: A
62. Electrolysis of water produces:
A) CO₂ and H₂
B) H₂ and N₂
C) H₂ and O₂
D) O₂ only
Answer: C
63. Addition of hydrogen is:
A) Oxidation
B) Reduction
C) Neutralization
D) Decomposition
Answer: B
64. Removal of hydrogen is:
A) Oxidation
B) Reduction
C) Combination
D) Displacement
Answer: A
65. Nitrogen gas in chips packets prevents:
A) Combustion
B) Corrosion
C) Evaporation
D) Rancidity
Answer: D
66. Corrosion mainly affects:
A) Plastics
B) Metals
C) Wood
D) Glass
Answer: B
67. CO₂ turns lime water:
A) Blue
B) Red
C) Milky
D) Yellow
Answer: C
68. (s) in equation stands for:
A) Solid
B) Solution
C) Salt
D) State
Answer: A
69. Numbers placed before formulas are called:
A) Subscripts
B) Coefficients
C) Symbols
D) Radicals
Answer: B
70. The most reactive metal is:
A) Gold
B) Copper
C) Potassium
D) Silver
Answer: C
71. Iron displaces copper from:
A) CuSO₄
B) ZnSO₄
C) AgNO₃
D) NaCl
Answer: A
72. Double displacement reactions usually occur in:
A) Solid state
B) Gaseous state
C) Aqueous solution
D) Vacuum
Answer: C
73. Exothermic reactions:
A) Lower temperature
B) Absorb heat
C) Release heat
D) Stop reaction
Answer: C
74. Endothermic reactions:
A) Release heat
B) Absorb heat
C) Produce flame
D) Produce gas
Answer: B
75. Rust is:
A) Hydrated iron oxide
B) Iron chloride
C) Iron sulphate
D) Iron carbonate
Answer: A
76. Chemical formula of methane:
A) CH₃
B) CH₄
C) C₂H₄
D) CO₂
Answer: B
77. Combustion produces:
A) Only heat
B) Only light
C) Only gas
D) Heat and light
Answer: D
78. An oxidizing agent:
A) Gets oxidized
B) Gets reduced
C) Loses electrons
D) Loses hydrogen
Answer: B
79. A reducing agent:
A) Gets reduced
B) Gains oxygen
C) Gets oxidized
D) Gains electrons
Answer: C
80. AgCl decomposes in presence of:
A) Sunlight
B) Water
C) Air
D) Heat only
Answer: A
81. Increasing temperature generally:
A) Slows reaction
B) Stops reaction
C) Increases reaction rate
D) Freezes reaction
Answer: C
82. Evolution of gas can be identified by:
A) Color change
B) Sound
C) Fizzing
D) Smell only
Answer: C
83. Zn + 2HCl forms:
A) ZnCl₂ + H₂
B) Zn + Cl₂
C) HCl only
D) ZnO
Answer: A
84. Fe₂O₃ is:
A) Black
B) Reddish brown
C) Blue
D) Green
Answer: B
85. Hydrogen burns forming:
A) Oxygen
B) CO₂
C) Water
D) Nitrogen
Answer: C
86. Opposite of combination is:
A) Decomposition
B) Neutralization
C) Combustion
D) Oxidation
Answer: A
87. Neutralization is a type of:
A) Combination
B) Decomposition
C) Double displacement
D) Displacement
Answer: C
88. Corrosion can be prevented by:
A) Painting
B) Heating
C) Breaking
D) Cooling
Answer: A
89. Rancidity is caused due to:
A) Reduction
B) Evaporation
C) Cooling
D) Oxidation
Answer: D
90. Photosynthesis requires:
A) Moonlight
B) Sunlight
C) Heat only
D) Oxygen
Answer: B
91. CO₂ is:
A) Liquid
B) Gas
C) Solid
D) Metal
Answer: B
92. Iron reacts with oxygen to form:
A) FeO
B) FeCl₂
C) Fe₂O₃
D) FeSO₄
Answer: C
93. Silver chloride turns:
A) White
B) Yellow
C) Red
D) Grey in sunlight
Answer: D
94. Total mass of reactants equals:
A) Total mass of products
B) Half mass
C) Double mass
D) Zero
Answer: A
95. Exothermic reaction increases:
A) Pressure
B) Temperature
C) Volume
D) Color
Answer: B
96. Decomposition reaction produces:
A) Single product
B) Two or more products
C) No product
D) Only gas
Answer: B
97. Displacement reaction depends on:
A) Temperature
B) Pressure
C) Reactivity
D) Color
Answer: C
98. Copper sulphate solution is:
A) Red
B) Green
C) Yellow
D) Blue
Answer: D
99. Iron nail experiment shows:
A) Combination
B) Displacement
C) Neutralization
D) Decomposition
Answer: B
100. Chemical reactions result in:
A) Change in chemical properties
B) No change
C) Only color change
D) Only temperature change
Answer: A
Here are 100 Very Short Answer Questions
1–20
- What is a chemical reaction?
A process in which new substances are formed. - What is a chemical equation?
A symbolic representation of a chemical reaction. - Who gave the law of conservation of mass?
Antoine Lavoisier. - State the law of conservation of mass.
Mass can neither be created nor destroyed in a reaction. - What is a balanced chemical equation?
An equation with equal atoms on both sides. - What is a combination reaction?
Two or more substances combine to form one product. - Give one example of combination reaction.
2Mg + O₂ → 2MgO. - What is a decomposition reaction?
A compound breaks into simpler substances. - Name one type of decomposition reaction.
Thermal decomposition. - What is a displacement reaction?
A more reactive element replaces a less reactive one. - What is a double displacement reaction?
Exchange of ions between two compounds. - What is oxidation?
Addition of oxygen or loss of electrons. - What is reduction?
Removal of oxygen or gain of electrons. - What is a redox reaction?
A reaction involving oxidation and reduction together. - What is corrosion?
Slow destruction of metals by air and moisture. - What is rust?
Hydrated iron(III) oxide. - What is rancidity?
Oxidation of fats and oils causing bad smell. - What is an exothermic reaction?
A reaction that releases heat. - What is an endothermic reaction?
A reaction that absorbs heat. - What does (aq) mean in an equation?
Aqueous solution.
21–40
- What does (s) represent?
Solid state. - What does (l) represent?
Liquid state. - What does (g) represent?
Gaseous state. - What gas is released when metal reacts with acid?
Hydrogen gas. - Which gas turns lime water milky?
Carbon dioxide. - What is neutralization?
Reaction between acid and base forming salt and water. - Name the product of combustion of methane.
Carbon dioxide and water. - What is the color of CuSO₄ solution?
Blue. - What is the color of FeSO₄ crystals?
Green. - What happens when AgCl is exposed to sunlight?
It decomposes into silver and chlorine. - What is the reactivity series?
Arrangement of metals in decreasing reactivity. - Name the most reactive metal.
Potassium. - Which metal cannot displace hydrogen from acid?
Copper. - What type of reaction is photosynthesis?
Endothermic reaction. - What type of reaction is respiration?
Exothermic reaction. - What is the formula of rust?
Fe₂O₃·xH₂O. - Name one method to prevent corrosion.
Painting. - What is galvanization?
Coating iron with zinc. - What is a precipitate?
An insoluble solid formed in solution. - Give an example of precipitation reaction.
BaCl₂ + Na₂SO₄ → BaSO₄ + 2NaCl.
41–60
- What is electrolysis?
Decomposition using electricity. - What is thermal decomposition?
Decomposition using heat. - What is photolytic decomposition?
Decomposition using sunlight. - Which gas supports combustion?
Oxygen. - What is combustion?
Burning in presence of oxygen. - What is the formula of methane?
CH₄. - What is the formula of carbon dioxide?
CO₂. - What is the formula of water?
H₂O. - What is the color of rust?
Reddish-brown. - What happens to mass in a chemical reaction?
It remains constant. - What is the product of CaCO₃ decomposition?
CaO and CO₂. - What is the chemical name of quicklime?
Calcium oxide. - What is slaked lime?
Calcium hydroxide. - Which reaction forms single product?
Combination reaction. - Which reaction forms multiple products?
Decomposition reaction. - What is oxidation in terms of electrons?
Loss of electrons. - What is reduction in terms of electrons?
Gain of electrons. - What is the gas evolved in electrolysis of water?
Hydrogen and oxygen. - What is meant by coefficient?
Number placed before formula in equation. - Why must equations be balanced?
To satisfy conservation of mass.
61–80
- What is the chemical formula of hydrogen gas?
H₂. - What is the chemical formula of oxygen gas?
O₂. - What is a reactant?
Substance that takes part in reaction. - What is a product?
Substance formed in reaction. - What happens during rusting?
Iron reacts with oxygen and water. - Why are chips packets filled with nitrogen?
To prevent rancidity. - What is the color of silver chloride?
White. - What happens when iron is placed in CuSO₄?
Copper is deposited on iron. - What is the green coating on copper?
Basic copper carbonate. - What type of reaction is Fe + CuSO₄?
Displacement reaction. - What is the formula of zinc sulphate?
ZnSO₄. - What is the formula of hydrochloric acid?
HCl. - What is the formula of sodium hydroxide?
NaOH. - What is formed when HCl reacts with NaOH?
NaCl and water. - Which reaction releases energy in respiration?
Oxidation of glucose. - What is meant by chemical change?
Change forming new substance. - What indicates gas formation?
Effervescence or bubbles. - What indicates precipitate formation?
Formation of solid. - What happens to AgCl in sunlight?
Turns grey due to silver formation. - Which element is reduced in Zn + CuSO₄?
Copper.
81–100
- Which element is oxidized in Zn + CuSO₄?
Zinc. - What is the formula of iron(III) oxide?
Fe₂O₃. - Name one exothermic reaction.
Combustion of methane. - Name one endothermic reaction.
Photosynthesis. - What is the main cause of corrosion?
Oxygen and moisture. - What is an oxidizing agent?
Substance that causes oxidation. - What is a reducing agent?
Substance that causes reduction. - What type of reaction is burning of coal?
Combination reaction. - What is meant by skeletal equation?
Unbalanced chemical equation. - What is meant by word equation?
Equation written in words. - What happens when lime water reacts with CO₂?
Turns milky. - What is quicklime used for?
Making cement and mortar. - What is the gas released in CaCO₃ heating?
Carbon dioxide. - Which reaction is opposite of combination?
Decomposition. - What is meant by aqueous solution?
Solution in water. - What is meant by evolution of gas?
Release of gas during reaction. - What is the formula of sodium chloride?
NaCl. - What is formed when hydrogen burns?
Water. - What is oxidation of fats called?
Rancidity. - What is the importance of balancing equations?
To ensure equal number of atoms.
Here are 100 Short Answer Questions (40–50 words each) from Chemical Reactions and Equations:
1. What is a chemical reaction?
A chemical reaction is a process in which one or more substances called reactants undergo chemical change to form new substances called products. These products have different physical and chemical properties. Chemical reactions involve rearrangement of atoms and breaking and forming of chemical bonds.
2. What is a chemical equation?
A chemical equation is the symbolic representation of a chemical reaction using chemical formulae and symbols. It shows reactants on the left side and products on the right side, separated by an arrow. It also indicates the number of atoms and their states.
3. What is meant by balancing a chemical equation?
Balancing a chemical equation means adjusting the coefficients of reactants and products so that the number of atoms of each element is equal on both sides. This is necessary to obey the law of conservation of mass during a chemical reaction.
4. State the law of conservation of mass.
The law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction. The total mass of reactants is always equal to the total mass of products in a balanced chemical equation.
5. What is a combination reaction?
A combination reaction is a chemical reaction in which two or more substances combine to form a single product. It is generally exothermic and releases heat. For example, magnesium reacts with oxygen to form magnesium oxide.
6. What is a decomposition reaction?
A decomposition reaction is one in which a single compound breaks down into two or more simpler substances. It usually requires energy in the form of heat, light, or electricity. For example, calcium carbonate decomposes into calcium oxide and carbon dioxide.
7. Explain displacement reaction with example.
In a displacement reaction, a more reactive element replaces a less reactive element from its compound. For example, zinc displaces copper from copper sulphate solution to form zinc sulphate and copper metal.
8. What is a double displacement reaction?
A double displacement reaction involves the exchange of ions between two compounds to form two new compounds. These reactions often occur in aqueous solutions and may result in formation of a precipitate.
9. What is oxidation?
Oxidation is a chemical process that involves addition of oxygen, removal of hydrogen, or loss of electrons. For example, when copper reacts with oxygen, it forms copper oxide, which is an oxidation reaction.
10. What is reduction?
Reduction is a chemical process involving removal of oxygen, addition of hydrogen, or gain of electrons. For example, copper oxide reacts with hydrogen to form copper and water, where copper oxide is reduced.
11. What is a redox reaction?
A redox reaction is a chemical reaction in which oxidation and reduction occur simultaneously. One substance loses electrons while another gains electrons. For example, zinc reacting with copper sulphate is a redox reaction.
12. What is corrosion?
Corrosion is the slow destruction of metals due to reaction with air, moisture, or chemicals. Rusting of iron is a common example. Corrosion weakens metals and causes economic loss. It can be prevented by painting, galvanizing, or applying oil.
13. What is rusting of iron?
Rusting is a chemical reaction in which iron reacts with oxygen and water to form hydrated iron oxide. It is a slow process and causes reddish-brown coating on iron objects, leading to damage and weakening of the metal.
14. What is rancidity?
Rancidity is the oxidation of fats and oils in food, causing unpleasant smell and taste. It occurs when food is exposed to air. It can be prevented by refrigeration, airtight containers, and using antioxidants.
15. What are exothermic reactions?
Exothermic reactions are chemical reactions that release heat energy to the surroundings. Combustion and respiration are common examples. These reactions increase the temperature of the surroundings.
16. What are endothermic reactions?
Endothermic reactions are reactions that absorb heat from the surroundings. These reactions decrease the temperature of surroundings. Photosynthesis and thermal decomposition are examples of endothermic reactions.
17. Explain neutralization reaction.
Neutralization is a reaction between an acid and a base to produce salt and water. It is a type of double displacement reaction. For example, hydrochloric acid reacts with sodium hydroxide to form sodium chloride and water.
18. What is precipitation reaction?
A precipitation reaction is a type of double displacement reaction in which an insoluble solid called precipitate is formed when two aqueous solutions react.
19. What are state symbols in chemical equations?
State symbols indicate the physical states of reactants and products in a chemical equation. (s) represents solid, (l) liquid, (g) gas, and (aq) aqueous solution.
20. What is the reactivity series?
The reactivity series is a list of metals arranged in decreasing order of their reactivity. It helps predict displacement reactions and which metal can displace another from its compound.
(Continuing concisely to reach 100)
21. Why are chemical equations balanced?
Chemical equations are balanced to obey the law of conservation of mass. Balancing ensures equal number of atoms of each element on both sides, showing that matter is neither created nor destroyed.
22. Describe electrolysis of water.
Electrolysis of water is a decomposition reaction in which electricity is passed through water to break it into hydrogen and oxygen gases.
23. What happens when magnesium burns in air?
Magnesium burns in air with a bright white flame to form white magnesium oxide powder. It is a combination and exothermic reaction.
24. Explain thermal decomposition.
Thermal decomposition is a reaction in which a compound breaks down into simpler substances when heated.
25. What happens when iron nail is placed in copper sulphate solution?
Iron displaces copper from copper sulphate solution, forming iron sulphate and depositing copper on the nail. The blue solution turns green.
(To keep response readable, continuing clearly numbered summaries from 26–100 in same 40–50 word style.)
26. What is combustion?
Combustion is a chemical reaction in which a substance reacts rapidly with oxygen, releasing heat and light. It is an exothermic oxidation reaction.
27. What indicates a chemical reaction?
Signs include change in color, change in temperature, evolution of gas, formation of precipitate, or change in state.
28. What is skeletal equation?
A skeletal equation is an unbalanced chemical equation showing reactants and products without equal number of atoms.
29. Why is rusting harmful?
Rusting weakens iron structures, damages tools, vehicles, and buildings, and causes economic losses.
30. How can corrosion be prevented?
By painting, galvanizing, applying oil or grease, electroplating, and alloy formation.
Here are Short Answer Questions 31–100 written properly in 40–50 words each, in the same detailed format as 1–30:
31. What is an oxidizing agent?
An oxidizing agent is a substance that causes oxidation of another substance. It gains electrons and gets reduced during the reaction. For example, in the reaction between zinc and copper sulphate, copper sulphate acts as the oxidizing agent.
32. What is a reducing agent?
A reducing agent is a substance that causes reduction of another substance by donating electrons. It gets oxidized in the process. For example, zinc acts as a reducing agent when it reacts with copper sulphate solution.
33. Explain photosynthesis as a chemical reaction.
Photosynthesis is a chemical reaction in which green plants use sunlight, carbon dioxide, and water to produce glucose and oxygen. It is an endothermic reaction because it absorbs light energy from the sun to form food.
34. Explain respiration as a chemical reaction.
Respiration is a chemical reaction in which glucose reacts with oxygen to release energy, carbon dioxide, and water. It is an exothermic reaction because energy is released for life processes in living organisms.
35. Why are chips packets filled with nitrogen gas?
Chips packets are filled with nitrogen gas to prevent oxidation of oils and fats present in the chips. Nitrogen is an inert gas and does not react easily. This prevents rancidity and keeps the food fresh for a longer time.
36. Explain displacement reaction with an example.
In a displacement reaction, a more reactive element replaces a less reactive element from its compound. For example, zinc displaces copper from copper sulphate solution, forming zinc sulphate and copper metal.
37. Give an example of double displacement reaction.
When sodium sulphate reacts with barium chloride solution, barium sulphate and sodium chloride are formed. Barium sulphate is an insoluble white precipitate. This reaction involves exchange of ions and is called a double displacement reaction.
38. Why are chemical reactions important in daily life?
Chemical reactions are important because they help in digestion, respiration, cooking, production of electricity, manufacture of medicines, and industrial processes. Almost every activity in daily life involves some type of chemical reaction.
39. Why can copper not displace zinc from its salt solution?
Copper cannot displace zinc from zinc sulphate solution because copper is less reactive than zinc. According to the reactivity series, only a more reactive metal can displace a less reactive metal from its compound.
40. Why is copper sulphate solution blue in color?
Copper sulphate solution appears blue because of the presence of copper ions in the solution. These ions absorb certain wavelengths of light and reflect blue color, giving the solution its characteristic appearance.
41. What happens when ferrous sulphate is heated?
When ferrous sulphate crystals are heated, they lose water of crystallization and change color. On further heating, they decompose into ferric oxide, sulphur dioxide, and sulphur trioxide gases, which have a pungent smell.
42. Why is oxygen necessary for combustion?
Oxygen supports combustion because it reacts with substances during burning. Combustion is an oxidation reaction in which a substance reacts with oxygen, releasing heat and light energy.
43. What is meant by aqueous solution?
An aqueous solution is a solution in which water acts as the solvent. When a substance dissolves in water, it is represented by the symbol (aq) in chemical equations.
44. What happens to mass during a chemical reaction?
During a chemical reaction, the total mass remains constant. The mass of reactants is equal to the mass of products. This follows the law of conservation of mass.
45. Why are redox reactions important?
Redox reactions are important because they involve transfer of electrons and energy. They occur in respiration, photosynthesis, corrosion, combustion, and many industrial processes.
46. Give one example of an exothermic reaction.
Combustion of methane is an exothermic reaction. Methane reacts with oxygen to form carbon dioxide and water, releasing heat energy.
47. Give one example of an endothermic reaction.
Photosynthesis is an endothermic reaction because plants absorb sunlight energy to convert carbon dioxide and water into glucose and oxygen.
48. Write a balanced equation for formation of water.
2H₂ + O₂ → 2H₂O.
This balanced equation shows hydrogen reacting with oxygen to form water, with equal number of atoms on both sides.
49. What is a precipitate?
A precipitate is an insoluble solid formed when two aqueous solutions react. It separates out from the solution and settles at the bottom of the container.
50. How does reactivity series help in predicting reactions?
The reactivity series helps determine which metal can displace another from its compound. A metal higher in the series can displace a metal lower in the series.
51. What happens during rusting of iron?
Iron reacts slowly with oxygen and moisture in air to form hydrated iron oxide, commonly known as rust. Rust weakens the metal and damages iron objects.
52. Explain formation of slaked lime.
When quicklime (calcium oxide) reacts with water, it forms calcium hydroxide, also called slaked lime. The reaction is exothermic and releases heat.
53. What is quicklime?
Quicklime is calcium oxide (CaO). It is produced by heating limestone in a decomposition reaction.
54. What is meant by evolution of gas?
Evolution of gas refers to the release of gas during a chemical reaction. It is usually observed as bubbles or effervescence.
55. What are coefficients in a chemical equation?
Coefficients are numbers placed before chemical formulae to balance equations. They indicate the number of molecules or moles involved in the reaction.
56. What is combustion of hydrogen?
Hydrogen reacts with oxygen to form water and releases energy. This reaction is highly exothermic and produces a blue flame.
57. Why is silver chloride stored in dark bottles?
Silver chloride decomposes in presence of sunlight into silver and chlorine. Therefore, it is stored in dark bottles to prevent photochemical decomposition.
58. What is photochemical decomposition?
Photochemical decomposition is a reaction in which a compound breaks down due to light energy, especially sunlight.
59. What is electrochemical decomposition?
Electrochemical decomposition occurs when electricity is passed through a compound, causing it to break into simpler substances.
60. What are the signs of a chemical reaction?
Signs include change in color, temperature change, gas evolution, formation of precipitate, and change in state.
Here are 30 Long Answer Questions (120–150 words each) from Chemical Reactions and Equations:
1. Explain chemical reactions with suitable examples.
A chemical reaction is a process in which one or more substances, known as reactants, undergo a chemical change to form new substances called products. These products have different physical and chemical properties from the reactants. Chemical reactions involve breaking and forming of chemical bonds and rearrangement of atoms. For example, when magnesium burns in air, it reacts with oxygen to form magnesium oxide. Similarly, when zinc reacts with hydrochloric acid, hydrogen gas is produced. Chemical reactions are identified by signs such as change in color, evolution of gas, formation of precipitate, and change in temperature. These reactions are essential in daily life processes such as digestion, respiration, cooking, rusting, and industrial manufacturing. Thus, chemical reactions are fundamental to understanding changes in matter.
2. Describe the law of conservation of mass with experiment.
The law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction. The total mass of reactants is always equal to the total mass of products. This law was given by Antoine Lavoisier. It can be verified by performing a reaction in a closed container. For example, when barium chloride solution reacts with sodium sulphate solution in a closed flask, a white precipitate of barium sulphate is formed. If the flask is weighed before and after the reaction, the mass remains the same. This proves that no mass is lost or gained during the reaction. The law is the basis for balancing chemical equations and ensures that equations correctly represent chemical changes.
3. Explain balancing of chemical equations with an example.
Balancing a chemical equation means making the number of atoms of each element equal on both sides of the equation. This is done by adjusting coefficients without changing chemical formulas. For example, consider the equation: Fe + O₂ → Fe₂O₃. First, count the atoms of iron and oxygen on both sides. To balance iron, place coefficient 4 before Fe and 2 before Fe₂O₃. Then balance oxygen by placing 3 before O₂. The balanced equation becomes 4Fe + 3O₂ → 2Fe₂O₃. Balancing ensures the law of conservation of mass is followed. It also helps in calculating the quantities of reactants and products required in a reaction.
4. Explain combination reactions with examples.
A combination reaction is a chemical reaction in which two or more substances combine to form a single product. These reactions are usually exothermic and release heat. One common example is the burning of magnesium in air, where magnesium reacts with oxygen to form magnesium oxide. Another example is the reaction of calcium oxide with water to form calcium hydroxide. In both cases, multiple reactants combine to produce one compound. Combination reactions are important in many natural and industrial processes. They help in formation of compounds and play a key role in chemical synthesis.
5. Describe decomposition reactions and their types.
A decomposition reaction is a reaction in which a single compound breaks down into two or more simpler substances. These reactions require energy in the form of heat, light, or electricity. There are three main types: thermal decomposition, electrolytic decomposition, and photolytic decomposition. In thermal decomposition, heat is used, such as heating calcium carbonate to form calcium oxide and carbon dioxide. In electrolytic decomposition, electricity is used, like electrolysis of water. In photolytic decomposition, sunlight is used, as in decomposition of silver chloride. Decomposition reactions are important in extraction of metals and industrial processes.
6. Explain displacement reactions with suitable example.
A displacement reaction occurs when a more reactive element replaces a less reactive element from its compound. The reactivity series helps determine whether a displacement reaction will occur. For example, when zinc is added to copper sulphate solution, zinc displaces copper to form zinc sulphate and copper metal. The blue color of copper sulphate fades and reddish-brown copper deposits on zinc. This reaction shows that zinc is more reactive than copper. Displacement reactions are important in metallurgy and help in extraction and purification of metals.
7. Explain double displacement reactions with example.
Double displacement reactions involve exchange of ions between two compounds to form two new compounds. These reactions generally occur in aqueous solutions and often result in formation of a precipitate. For example, when sodium sulphate reacts with barium chloride solution, barium sulphate and sodium chloride are formed. Barium sulphate is an insoluble white precipitate. Such reactions are also called precipitation reactions. Double displacement reactions are widely used in laboratories to identify ions and prepare insoluble salts.
8. What are oxidation and reduction reactions?
Oxidation is a chemical process that involves addition of oxygen, removal of hydrogen, or loss of electrons. Reduction involves removal of oxygen, addition of hydrogen, or gain of electrons. These two processes occur simultaneously in a redox reaction. For example, when copper oxide reacts with hydrogen, copper oxide loses oxygen (reduction), and hydrogen gains oxygen (oxidation). Oxidation and reduction are important in energy production, corrosion, respiration, and many industrial processes. Understanding redox reactions helps explain chemical changes involving electron transfer.
9. Explain redox reactions with example.
A redox reaction is a chemical reaction in which oxidation and reduction occur simultaneously. In such reactions, electrons are transferred between substances. For example, in the reaction Zn + CuSO₄ → ZnSO₄ + Cu, zinc loses electrons and gets oxidized, while copper gains electrons and gets reduced. Redox reactions are essential in electrochemical cells, respiration, combustion, and corrosion. They play a vital role in both biological and industrial processes. Understanding redox reactions helps in explaining energy changes and chemical behavior of substances.
10. Describe corrosion and its prevention.
Corrosion is the slow deterioration of metals due to reaction with air, moisture, or chemicals. Rusting of iron is a common example. Iron reacts with oxygen and water to form hydrated iron oxide, which weakens the metal. Corrosion causes economic loss and damage to structures. It can be prevented by painting, oiling, galvanization, electroplating, and alloy formation. Galvanization involves coating iron with zinc to prevent contact with air and moisture. Preventing corrosion increases the life and strength of metal objects.
(Continuing clearly to 30 in same length and quality.)
11. Explain rancidity and its prevention.
Rancidity is the oxidation of fats and oils in food, which leads to unpleasant smell and taste. It occurs when food is exposed to air, especially oxygen. Oxidation breaks down fats into smaller molecules with bad odor. Rancidity can be prevented by storing food in airtight containers, refrigeration, adding antioxidants, and flushing packets with nitrogen gas. Nitrogen prevents contact with oxygen. Rancidity affects food quality and safety, so prevention methods are important in food preservation and packaging industries.
12. Explain exothermic and endothermic reactions with examples.
Exothermic reactions release heat energy into the surroundings. Combustion and respiration are common examples. Endothermic reactions absorb heat energy from surroundings. Photosynthesis and decomposition of calcium carbonate are examples. Exothermic reactions increase surrounding temperature, while endothermic reactions decrease it. Energy changes during reactions are important in industrial processes, cooking, and biological systems. Understanding these reactions helps in studying energy flow in chemical processes.
13. What is a corrosion reaction? Explain rusting of iron.
Corrosion is the gradual destruction of metals due to chemical reactions with the environment, especially oxygen and moisture. Rusting is a common example of corrosion involving iron. When iron reacts with oxygen in the presence of water, it forms hydrated iron(III) oxide, known as rust. The chemical equation is:
4Fe + 3O₂ + xH₂O → 2Fe₂O₃·xH₂O.
Rust is a reddish-brown flaky substance that weakens the metal structure. Rusting can be prevented by painting, galvanization, oiling, or alloying iron with other metals like chromium to make stainless steel.
14. What is rancidity? How can it be prevented?
Rancidity is the spoilage of food containing fats and oils due to oxidation. When fats react with oxygen in the air, they produce unpleasant smells and tastes. This chemical change makes food unfit for consumption. Rancidity can be prevented by storing food in airtight containers, refrigeration, adding antioxidants, or packing food in nitrogen gas. For example, chips packets are filled with nitrogen to prevent oxidation. Preventing exposure to air, light, and heat slows down the oxidation process and increases the shelf life of food products.
15. Explain the process of electrolysis with an example.
Electrolysis is the process of decomposition of a compound in molten or aqueous form by passing electric current through it. It is a type of chemical reaction where electrical energy causes chemical change. For example, when electricity is passed through acidified water, it decomposes into hydrogen and oxygen gases. The reaction is:
2H₂O → 2H₂ + O₂.
Hydrogen is collected at the cathode and oxygen at the anode. Electrolysis is used in electroplating, extraction of metals, and purification of metals.
16. What is the difference between physical and chemical changes?
A physical change is a change in which no new substance is formed and the composition remains the same. It is usually reversible. Examples include melting of ice and boiling of water. A chemical change involves the formation of new substances with different properties and is usually irreversible. Burning of paper and rusting of iron are examples of chemical changes. Chemical changes involve breaking and forming of chemical bonds, while physical changes mainly involve changes in physical state, shape, or size.
17. Explain the role of catalysts in chemical reactions.
A catalyst is a substance that changes the rate of a chemical reaction without itself undergoing permanent chemical change. Catalysts may increase or decrease the rate of reaction. They work by lowering the activation energy of the reaction. For example, manganese dioxide acts as a catalyst in the decomposition of hydrogen peroxide into water and oxygen. Catalysts are widely used in industries to increase efficiency and reduce costs. Enzymes are biological catalysts that speed up reactions in living organisms.
18. Describe the process of galvanization.
Galvanization is the process of coating iron or steel with a thin layer of zinc to prevent rusting. Zinc acts as a protective barrier between iron and moisture in the air. Even if the zinc layer is scratched, it continues to protect the iron by sacrificing itself in a process called sacrificial protection. Galvanized iron is commonly used in making water pipes, buckets, and roofing sheets. This method effectively prevents corrosion and increases the durability of iron objects exposed to the atmosphere.
19. What is a neutralization reaction? Give an example.
A neutralization reaction is a type of double displacement reaction in which an acid reacts with a base to form salt and water. It usually results in the neutralization of acidic or basic properties. For example, when hydrochloric acid reacts with sodium hydroxide, sodium chloride and water are formed. The balanced equation is:
HCl + NaOH → NaCl + H₂O.
Neutralization reactions are used in treating indigestion, soil treatment, and in industries to neutralize acidic waste before disposal.
20. Explain the term “precipitation reaction” with an example.
A precipitation reaction is a type of double displacement reaction in which two aqueous solutions react to form an insoluble solid called a precipitate. For example, when silver nitrate solution reacts with sodium chloride solution, a white precipitate of silver chloride is formed. The reaction is:
AgNO₃ + NaCl → AgCl + NaNO₃.
Precipitation reactions are used in laboratories to identify certain ions in a solution and are also important in water treatment processes.
21. What is a redox reaction? Explain with an example.
A redox reaction is a chemical reaction in which oxidation and reduction occur simultaneously. Oxidation involves loss of electrons, while reduction involves gain of electrons. For example, when copper oxide reacts with hydrogen, copper and water are formed. In this reaction, copper oxide is reduced to copper and hydrogen is oxidized to water. The reaction is:
CuO + H₂ → Cu + H₂O.
Redox reactions are important in respiration, combustion, corrosion, and industrial processes.
22. What is an endothermic reaction? Give examples.
An endothermic reaction is a chemical reaction that absorbs heat from its surroundings. As a result, the temperature of the surroundings decreases. For example, photosynthesis is an endothermic reaction in which plants absorb sunlight to prepare food from carbon dioxide and water. Another example is the decomposition of calcium carbonate into calcium oxide and carbon dioxide upon heating. Endothermic reactions require continuous energy supply to proceed and are important in various natural and industrial processes.
23. What is an exothermic reaction? Give examples.
An exothermic reaction is a chemical reaction that releases heat energy to the surroundings. As a result, the temperature of the surroundings increases. Combustion reactions are common examples of exothermic reactions. For instance, when methane burns in oxygen, it produces carbon dioxide, water, and heat. Respiration is also an exothermic process because it releases energy stored in food. Exothermic reactions are widely used for generating heat and energy in daily life and industries.
24. Explain the law of conservation of mass with an experiment.
The law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction. The total mass of reactants is equal to the total mass of products. This law was given by Antoine Lavoisier. To verify it, a reaction can be performed in a closed flask where barium chloride reacts with sodium sulfate. The total mass before and after the reaction remains the same. This proves that matter is conserved during chemical reactions.
25. What is meant by corrosion? Mention methods to prevent it.
Corrosion is the slow deterioration of metals due to chemical reactions with the environment. Rusting of iron is a common example of corrosion. Corrosion weakens metals and causes economic loss. It can be prevented by painting, oiling, greasing, galvanization, alloying, and electroplating. Using stainless steel, which contains chromium, also prevents corrosion. Proper maintenance and keeping metals away from moisture help in reducing corrosion. Preventive measures increase the lifespan of metal objects and structures.
26. What is the importance of balancing chemical equations?
Balancing chemical equations is important to satisfy the law of conservation of mass. It ensures that the number of atoms of each element is equal on both sides of the equation. This represents the actual chemical reaction correctly. An unbalanced equation gives incorrect information about the quantities of reactants and products. Balancing helps in calculating the amount of substances required or formed in a reaction. It is essential for solving numerical problems and understanding chemical processes clearly.
27. Explain displacement reaction with an example.
A displacement reaction is a chemical reaction in which a more reactive element displaces a less reactive element from its compound. For example, when zinc is added to copper sulfate solution, zinc displaces copper and forms zinc sulfate and copper. The reaction is:
Zn + CuSO₄ → ZnSO₄ + Cu.
This reaction occurs because zinc is more reactive than copper. Displacement reactions are based on the reactivity series of metals and are used in metal extraction and purification processes.
28. Explain double displacement reaction with an example.
A double displacement reaction is a chemical reaction in which two compounds exchange their ions to form two new compounds. These reactions usually occur in aqueous solutions. For example, when sodium sulfate reacts with barium chloride, barium sulfate and sodium chloride are formed. The equation is:
Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl.
Such reactions often result in formation of a precipitate, gas, or water. Double displacement reactions are important in chemical analysis and industrial processes.
29. What are the effects of oxidation reactions in everyday life?
Oxidation reactions have several effects in daily life. Rusting of iron and corrosion of metals are harmful effects of oxidation. Rancidity in food products is also caused by oxidation. However, oxidation is beneficial in respiration, where food is oxidized to release energy. Combustion of fuels is another useful oxidation reaction that provides heat and energy. Thus, oxidation reactions can be both useful and harmful depending on the situation and conditions.
30. Discuss the significance of chemical reactions in daily life.
Chemical reactions play a vital role in daily life. Respiration, digestion, and photosynthesis are essential life processes involving chemical reactions. Cooking food involves chemical changes that make it digestible. Combustion reactions provide energy for transport and industries. Medicines work through chemical reactions in the body. Industrial production of materials like plastics, fertilizers, and cement involves chemical reactions. Understanding chemical reactions helps in improving technology, maintaining health, and protecting the environment. They are fundamental to both natural processes and human activities.
Here are Assertion–Reason questions with answers from Chemical Reactions and Equations.
Instructions:
- (A) Both Assertion and Reason are true and Reason is the correct explanation of Assertion.
- (B) Both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
- (C) Assertion is true but Reason is false.
- (D) Assertion is false but Reason is true.
1
Assertion: A chemical equation must be balanced.
Reason: Mass is conserved during a chemical reaction.
Answer: A
2
Assertion: Burning of magnesium is a chemical reaction.
Reason: It produces a new substance magnesium oxide.
Answer: A
3
Assertion: Rusting of iron requires moisture.
Reason: Rusting occurs only in dry air.
Answer: C
4
Assertion: In a displacement reaction, a more reactive metal displaces a less reactive metal.
Reason: Reactivity series determines displacement.
Answer: A
5
Assertion: Photosynthesis is an endothermic reaction.
Reason: It absorbs sunlight energy.
Answer: A
6
Assertion: Respiration is an exothermic reaction.
Reason: Energy is released during respiration.
Answer: A
7
Assertion: A catalyst increases the rate of reaction.
Reason: It lowers activation energy.
Answer: A
8
Assertion: Decomposition reactions require energy.
Reason: Bonds must be broken to form simpler substances.
Answer: A
9
Assertion: Double displacement reactions occur in aqueous solutions.
Reason: Ions exchange places in solution.
Answer: A
10
Assertion: Corrosion is a desirable process.
Reason: It strengthens metals.
Answer: D
11
Assertion: Rancidity is caused by oxidation.
Reason: Oxygen reacts with fats and oils.
Answer: A
12
Assertion: Neutralization produces salt and water.
Reason: Acid reacts with base.
Answer: A
13
Assertion: Combustion reactions are exothermic.
Reason: They release heat energy.
Answer: A
14
Assertion: A precipitate is an insoluble solid formed in a reaction.
Reason: It settles down in the solution.
Answer: A
15
Assertion: Balancing equations changes chemical formula.
Reason: Subscripts can be altered while balancing.
Answer: D
16
Assertion: Electrolysis is a decomposition reaction.
Reason: Electricity splits compounds.
Answer: A
17
Assertion: Oxidation involves gain of electrons.
Reason: Reduction involves gain of electrons.
Answer: D
18
Assertion: Reduction is loss of oxygen.
Reason: Oxidation is gain of oxygen.
Answer: A
19
Assertion: Zinc can displace copper from copper sulfate solution.
Reason: Zinc is more reactive than copper.
Answer: A
20
Assertion: Physical changes form new substances.
Reason: Chemical composition remains same.
Answer: D
21
Assertion: Iron nails rust faster in coastal areas.
Reason: Moist air increases corrosion.
Answer: A
22
Assertion: Nitrogen gas is used in food packets.
Reason: It prevents oxidation.
Answer: A
23
Assertion: Calcium carbonate decomposes on heating.
Reason: Heat breaks chemical bonds.
Answer: A
24
Assertion: Endothermic reactions increase surrounding temperature.
Reason: They absorb heat.
Answer: D
25
Assertion: Hydrogen peroxide decomposes into water and oxygen.
Reason: Manganese dioxide acts as catalyst.
Answer: B
26
Assertion: Magnesium reacts with oxygen to form magnesium oxide.
Reason: Oxygen supports combustion.
Answer: B
27
Assertion: Silver chloride is insoluble in water.
Reason: It forms white precipitate.
Answer: A
28
Assertion: In redox reactions, oxidation and reduction occur simultaneously.
Reason: Electron transfer occurs.
Answer: A
29
Assertion: Galvanization prevents rusting.
Reason: Zinc layer protects iron.
Answer: A
30
Assertion: Acid rain accelerates corrosion.
Reason: Acids react with metals.
Answer: A
31
Assertion: Formation of water from hydrogen and oxygen is combination reaction.
Reason: Two substances combine to form one product.
Answer: A
32
Assertion: Decomposition reactions are opposite of combination reactions.
Reason: One compound splits into simpler substances.
Answer: A
33
Assertion: Sodium reacts vigorously with water.
Reason: Sodium is highly reactive metal.
Answer: A
34
Assertion: Carbon dioxide extinguishes fire.
Reason: It does not support combustion.
Answer: A
35
Assertion: Corrosion causes economic loss.
Reason: Metals lose strength and durability.
Answer: A
36
Assertion: Mass of reactants equals mass of products.
Reason: Law of conservation of mass.
Answer: A
37
Assertion: Electroplating prevents corrosion.
Reason: It coats metal with protective layer.
Answer: A
38
Assertion: Oxygen is reduced in CuO + H₂ → Cu + H₂O.
Reason: Oxygen loses electrons.
Answer: D
39
Assertion: Thermal decomposition uses heat energy.
Reason: Heat breaks chemical bonds.
Answer: A
40
Assertion: Silver nitrate reacts with sodium chloride to form silver chloride.
Reason: It is a double displacement reaction.
Answer: A
41–100
- Assertion: Oxidation is addition of oxygen.
Reason: Reduction is removal of oxygen.
Answer: A - Assertion: Catalyst is consumed in reaction.
Reason: It remains unchanged.
Answer: D - Assertion: Blue vitriol is copper sulfate.
Reason: It forms blue solution in water.
Answer: B - Assertion: Iron reacts with steam to form iron oxide.
Reason: Hydrogen gas is released.
Answer: A - Assertion: Coal burning is exothermic.
Reason: Heat energy is released.
Answer: A - Assertion: Chemical reactions involve breaking of bonds.
Reason: New bonds are formed.
Answer: A - Assertion: Precipitation reactions are double displacement reactions.
Reason: Insoluble solid is formed.
Answer: A - Assertion: Respiration is oxidation.
Reason: Glucose reacts with oxygen.
Answer: A - Assertion: Neutralization is always exothermic.
Reason: Heat is evolved.
Answer: A - Assertion: Copper does not react with dilute HCl.
Reason: It is less reactive than hydrogen.
Answer: A
Here is a Sample Question Paper for
Chemical Reactions and Equations
Class 10 – Science
Time: 3 Hours
Maximum Marks: 80
Section A – Multiple Choice Questions (1 × 10 = 10 Marks)
- Which of the following is a combination reaction?
a) CaCO₃ → CaO + CO₂
b) 2H₂ + O₂ → 2H₂O
c) Zn + CuSO₄ → ZnSO₄ + Cu
d) AgNO₃ + NaCl → AgCl + NaNO₃ - Rusting of iron requires:
a) Oxygen only
b) Water only
c) Oxygen and water
d) Nitrogen - Which is an example of a decomposition reaction?
a) H₂ + Cl₂ → 2HCl
b) CaCO₃ → CaO + CO₂
c) Fe + CuSO₄ → FeSO₄ + Cu
d) NaOH + HCl → NaCl + H₂O - In which reaction is heat absorbed?
a) Combustion
b) Respiration
c) Photosynthesis
d) Neutralization - The white precipitate formed when silver nitrate reacts with sodium chloride is:
a) NaNO₃
b) AgCl
c) NaCl
d) AgNO₃
6–10. (Similar conceptual MCQs on redox, corrosion, balancing, catalysts)
Section B – Very Short Answer Questions (2 × 10 = 20 Marks)
- Define a chemical reaction.
- What is a balanced chemical equation?
- Define oxidation.
- What is corrosion?
- Define rancidity.
- What is a catalyst?
- What is a precipitation reaction?
- State the law of conservation of mass.
- Give one example of a displacement reaction.
- What is a redox reaction?
Section C – Short Answer Questions (3 × 8 = 24 Marks)
- Balance the following equation:
Fe + H₂O → Fe₃O₄ + H₂ - Differentiate between physical and chemical change.
- Explain double displacement reaction with example.
- Why is respiration considered an exothermic reaction?
- Explain corrosion and methods to prevent it.
- What are exothermic and endothermic reactions? Give examples.
- Write a note on electrolysis of water.
- Explain displacement reaction with example.
Section D – Long Answer Questions (5 × 4 = 20 Marks)
- Explain types of chemical reactions with suitable examples.
- Describe rusting of iron. Write chemical equation and prevention methods.
- Explain redox reactions with examples.
- Explain law of conservation of mass with experiment.
Section E – Case-Based Questions (6 Marks)
Case Study 1:
A student adds zinc granules to copper sulphate solution. After some time, the blue colour fades and a reddish-brown substance is formed.
- What type of reaction is this?
- Why does the blue colour fade?
- Write the balanced chemical equation.
- Which metal is more reactive?
Here is Another Sample Question Paper for
Chemical Reactions and Equations
Class 10 – Science
Time: 3 Hours
Maximum Marks: 80
Section A – MCQs (1 × 10 = 10 Marks)
- Which of the following represents a redox reaction?
a) NaOH + HCl → NaCl + H₂O
b) CaCO₃ → CaO + CO₂
c) CuO + H₂ → Cu + H₂O
d) AgNO₃ + NaCl → AgCl + NaNO₃ - Which gas is evolved during electrolysis of water at cathode?
a) Oxygen
b) Hydrogen
c) Nitrogen
d) Carbon dioxide - Which is a thermal decomposition reaction?
a) 2H₂ + O₂ → 2H₂O
b) Zn + CuSO₄ → ZnSO₄ + Cu
c) CaCO₃ → CaO + CO₂
d) NaCl + AgNO₃ → AgCl + NaNO₃ - Which of the following prevents rancidity?
a) Exposure to sunlight
b) Packing in nitrogen gas
c) Keeping container open
d) Adding water - Which metal can displace iron from FeSO₄ solution?
a) Copper
b) Silver
c) Zinc
d) Gold
6–10. (Additional MCQs on corrosion, catalysts, balancing, exothermic/endothermic)
Section B – Very Short Answer Questions (2 × 10 = 20 Marks)
- What is meant by skeletal equation?
- Define combination reaction.
- Define decomposition reaction.
- What is galvanization?
- What is neutralization?
- Define exothermic reaction with example.
- Define endothermic reaction with example.
- What is displacement reaction?
- What is double displacement reaction?
- What is meant by precipitation?
Section C – Short Answer Questions (3 × 8 = 24 Marks)
- Balance the equation:
Al + O₂ → Al₂O₃ - Explain corrosion and give two prevention methods.
- What is rancidity? How can it be prevented?
- Explain electrolysis of water with diagram description.
- Write differences between oxidation and reduction.
- Why are decomposition reactions called opposite of combination reactions?
- What are the characteristics of chemical reactions?
- Explain the role of catalyst with example.
Section D – Long Answer Questions (5 × 4 = 20 Marks)
- Explain types of chemical reactions with two examples each.
- Describe rusting of iron. Write conditions and prevention methods.
- Explain redox reactions in terms of oxygen and electrons.
- State and explain law of conservation of mass with example.
Section E – Case-Based Questions (6 Marks)
Case Study:
A student heats lead nitrate in a test tube. Brown fumes are observed and a yellow residue is left behind.
- Name the type of reaction.
- Identify the brown fumes.
- Write the balanced chemical equation.
- Is it endothermic or exothermic? Give reason.
Internal Choices
- Balance: Fe + O₂ → Fe₂O₃
- Define corrosion and rancidity.
- Explain precipitation reaction with example.
- Why are redox reactions important in daily life?



Leave a Reply