1. Introduction
Elements are broadly classified into metals, non-metals, and metalloids based on their physical and chemical properties. Most elements in the periodic table are metals. Metals are generally electropositive and tend to lose electrons, while non-metals are electronegative and tend to gain electrons. These differences explain their contrasting behavior in chemical reactions, bonding, and applications in daily life.
Understanding metals and non-metals is essential because they form the basis of materials used in construction, electronics, transportation, medicine, agriculture, and industry.
2. Physical Properties of Metals




Metals show the following physical properties:
(i) Lustre
Metals have a shiny surface called metallic lustre. For example, gold, silver, and copper are shiny when freshly cut.
(ii) Hardness
Most metals are hard (iron, copper), but some like sodium and potassium are soft and can be cut with a knife.
(iii) Malleability
Metals can be beaten into thin sheets. Gold and silver are highly malleable.
(iv) Ductility
Metals can be drawn into thin wires. Copper and aluminum are widely used in electrical wiring.
(v) Conductivity
Metals are good conductors of heat and electricity due to free electrons. Silver is the best conductor of electricity.
(vi) Sonority
Metals produce a ringing sound when struck. This property is called sonority.
(vii) High Melting and Boiling Points
Most metals have high melting and boiling points, except metals like mercury which is liquid at room temperature.
3. Physical Properties of Non-Metals




Non-metals generally show opposite properties:
(i) Non-lustrous
They are usually dull in appearance. Example: sulfur.
(ii) Brittle
They break easily when hammered.
(iii) Poor Conductors
Non-metals are poor conductors of heat and electricity, except graphite (an allotrope of carbon).
(iv) Non-sonorous
They do not produce sound when struck.
(v) Low Melting and Boiling Points
Most non-metals have low melting points. For example, bromine is liquid at room temperature.
4. Chemical Properties of Metals
(i) Reaction with Oxygen
Metals react with oxygen to form metal oxides.
Example:
4Na + O₂ → 2Na₂O
Metal oxides are generally basic in nature. Some like aluminum oxide are amphoteric.
(ii) Reaction with Water
Different metals react differently with water.
| Metal | Reaction |
|---|---|
| Sodium, Potassium | React violently with cold water |
| Calcium | Reacts less violently |
| Magnesium | Reacts with hot water |
| Iron | Reacts with steam |
| Copper | Does not react |
Example:
2Na + 2H₂O → 2NaOH + H₂
(iii) Reaction with Acids
Metals react with dilute acids to produce salt and hydrogen gas.
Zn + 2HCl → ZnCl₂ + H₂
Copper does not react with dilute HCl.
(iv) Reaction with Chlorine
Metals combine with chlorine to form metal chlorides.
2Na + Cl₂ → 2NaCl
(v) Reactivity Series



The Reactivity Series arranges metals in decreasing order of reactivity:
K > Na > Ca > Mg > Al > Zn > Fe > Pb > H > Cu > Hg > Ag > Au
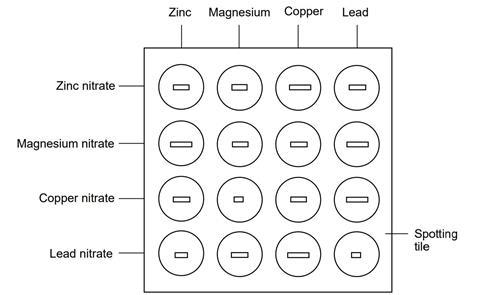
A more reactive metal displaces a less reactive metal from its salt solution.
Example:
Zn + CuSO₄ → ZnSO₄ + Cu
5. Chemical Properties of Non-Metals
(i) Reaction with Oxygen
Non-metals form non-metal oxides.
C + O₂ → CO₂
Non-metal oxides are generally acidic.
(ii) Reaction with Hydrogen
Non-metals react with hydrogen to form covalent hydrides.
H₂ + Cl₂ → 2HCl
(iii) Reaction with Metals
Non-metals react with metals to form ionic compounds.
2Na + Cl₂ → 2NaCl
6. Ionic Compounds


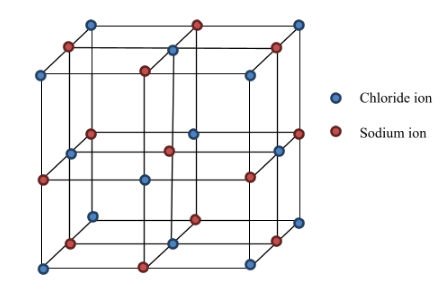
Ionic compounds are formed by transfer of electrons from metals to non-metals.
Properties:
- High melting and boiling points
- Soluble in water
- Conduct electricity in molten or aqueous state
- Hard and brittle
Example: Sodium chloride (NaCl)
7. Occurrence of Metals
Metals occur in nature in free or combined state.
- Native State: Gold, silver (less reactive metals)
- Combined State: Most metals occur as ores.
Ores and Minerals
An ore is a mineral from which metal can be extracted economically.
8. Extraction of Metals
(i) Concentration of Ore
Removal of impurities (gangue).
(ii) Roasting and Calcination
Roasting: Heating sulfide ores in air.
Calcination: Heating carbonate ores in limited air.
(iii) Reduction
Metal oxides are reduced to metals using carbon or by electrolysis.
Example:
Fe₂O₃ + 3C → 2Fe + 3CO
Highly reactive metals like sodium are extracted by electrolysis.
9. Corrosion




Corrosion is gradual destruction of metals due to reaction with air and moisture.
Example: Rusting of iron
4Fe + 3O₂ + xH₂O → 2Fe₂O₃·xH₂O
Prevention:
- Painting
- Oiling
- Galvanization
- Alloying
10. Alloys
An alloy is a homogeneous mixture of two or more metals or a metal and a non-metal.
Examples:
- Brass (Copper + Zinc)
- Bronze (Copper + Tin)
- Steel (Iron + Carbon)
Alloys improve strength, hardness, and corrosion resistance.
11. Properties and Uses of Important Metals
Iron
Used in construction, machinery, vehicles.
Copper
Used in electrical wiring.
Aluminum
Used in utensils, aircraft, packaging.
Gold and Silver
Used in jewelry and electronics.
12. Properties and Uses of Important Non-Metals
Oxygen
Essential for respiration.
Nitrogen
Used in fertilizers.
Carbon
Used as fuel and in making steel.
Chlorine
Used in water purification.
13. Differences Between Metals and Non-Metals
| Metals | Non-Metals |
|---|---|
| Lustrous | Dull |
| Malleable | Brittle |
| Good conductors | Poor conductors |
| Form cations | Form anions |
| Basic oxides | Acidic oxides |
14. Environmental Aspects
Mining causes deforestation and pollution. Recycling metals reduces environmental damage. Sustainable use of metals is important for conservation.
15. Importance in Daily Life
Metals are used in transportation, construction, electronics, and medical instruments. Non-metals are essential in respiration, fertilizers, fuels, and disinfectants.
Conclusion
Metals and non-metals differ significantly in their physical and chemical properties. Metals tend to lose electrons and form ionic compounds, while non-metals gain electrons and form covalent compounds. Their reactivity determines their extraction methods and applications. From construction materials to life-supporting gases, these elements are fundamental to human life and industrial development. Understanding their properties helps us use them efficiently and sustainably.
Here are 100 Multiple Choice Questions (MCQs) from the chapter Metals and Non-Metals (with answers):
🧪 METALS AND NON-METALS – 100 MCQs
🔹 Physical Properties of Metals
- Which of the following is a metal?
A) Oxygen
B) Nitrogen
C) Iron
D) Sulphur
Answer: C - Metals are generally:
A) Dull
B) Brittle
C) Lustrous
D) Soft
Answer: C - The property of being beaten into thin sheets is called:
A) Ductility
B) Malleability
C) Sonority
D) Hardness
Answer: B - The ability to draw metals into wires is called:
A) Malleability
B) Conductivity
C) Ductility
D) Sonority
Answer: C - Which metal is liquid at room temperature?
A) Mercury
B) Iron
C) Sodium
D) Zinc
Answer: A - Metals are good conductors of:
A) Heat only
B) Electricity only
C) Both heat and electricity
D) None
Answer: C - Which metal is soft and can be cut with a knife?
A) Iron
B) Copper
C) Sodium
D) Aluminium
Answer: C - Metals are generally:
A) Poor conductors
B) Non-sonorous
C) High melting point
D) Brittle
Answer: C - Which is NOT a metal?
A) Gold
B) Silver
C) Carbon
D) Copper
Answer: C - The shining property of metals is called:
A) Ductility
B) Malleability
C) Lustre
D) Hardness
Answer: C
🔹 Chemical Properties of Metals
- Metals react with oxygen to form:
A) Acids
B) Bases
C) Metal oxides
D) Salts
Answer: C - Metal oxides are generally:
A) Acidic
B) Basic
C) Neutral
D) Amphoteric only
Answer: B - When zinc reacts with dilute HCl, it produces:
A) Oxygen
B) Nitrogen
C) Hydrogen
D) Chlorine
Answer: C - Reaction of metals with water produces:
A) Salt and oxygen
B) Metal hydroxide and hydrogen
C) Salt only
D) Water only
Answer: B - Which metal reacts most vigorously with water?
A) Iron
B) Copper
C) Sodium
D) Gold
Answer: C - The gas evolved when metal reacts with acid is:
A) Oxygen
B) Hydrogen
C) Carbon dioxide
D) Nitrogen
Answer: B - Aluminium reacts with:
A) Acid only
B) Base only
C) Both acid and base
D) None
Answer: C - Rusting is an example of:
A) Combustion
B) Corrosion
C) Reduction
D) Neutralization
Answer: B - Rust is:
A) Iron sulphide
B) Iron oxide
C) Hydrated iron oxide
D) Iron carbonate
Answer: C - Galvanization prevents:
A) Melting
B) Corrosion
C) Reaction
D) Ductility
Answer: B
🔹 Reactivity Series
- The most reactive metal is:
A) Gold
B) Copper
C) Potassium
D) Silver
Answer: C - The least reactive metal is:
A) Gold
B) Zinc
C) Magnesium
D) Sodium
Answer: A - A more reactive metal can displace:
A) Less reactive metal
B) More reactive metal
C) Hydrogen only
D) Oxygen
Answer: A - Which metal is above hydrogen in reactivity series?
A) Copper
B) Silver
C) Zinc
D) Gold
Answer: C - Copper cannot displace:
A) Silver
B) Gold
C) Iron
D) Mercury
Answer: C
🔹 Extraction of Metals
- Ores are:
A) Pure metals
B) Impure metals
C) Minerals from which metals are extracted
D) Compounds of oxygen
Answer: C - Removal of gangue is called:
A) Roasting
B) Concentration
C) Reduction
D) Refining
Answer: B - Heating ore in absence of air is:
A) Roasting
B) Calcination
C) Reduction
D) Smelting
Answer: B - Highly reactive metals are extracted by:
A) Heating
B) Electrolysis
C) Roasting
D) Refining
Answer: B - Iron is extracted in:
A) Kiln
B) Blast furnace
C) Boiler
D) Oven
Answer: B
🔹 Alloys
- Brass is an alloy of:
A) Copper + Tin
B) Copper + Zinc
C) Iron + Carbon
D) Aluminium + Copper
Answer: B - Bronze contains:
A) Copper + Tin
B) Iron + Zinc
C) Aluminium + Iron
D) Copper + Silver
Answer: A - Steel is alloy of:
A) Copper
B) Aluminium
C) Iron + Carbon
D) Gold
Answer: C - Alloys are generally:
A) Weaker
B) More reactive
C) Harder
D) Softer
Answer: C - Stainless steel contains:
A) Copper
B) Zinc
C) Chromium
D) Gold
Answer: C
🔹 Non-Metals
- Which is a non-metal?
A) Sodium
B) Nitrogen
C) Calcium
D) Iron
Answer: B - Non-metals are generally:
A) Lustrous
B) Malleable
C) Brittle
D) Ductile
Answer: C - Graphite is a form of:
A) Sulphur
B) Carbon
C) Oxygen
D) Nitrogen
Answer: B - Graphite conducts:
A) Heat
B) Electricity
C) Both
D) None
Answer: B - Non-metal oxides are generally:
A) Basic
B) Acidic
C) Neutral
D) Metallic
Answer: B
🔹 Ionic Compounds
- Ionic bond is formed by:
A) Sharing of electrons
B) Transfer of electrons
C) Losing neutrons
D) Gaining protons
Answer: B - NaCl is formed by:
A) Covalent bond
B) Ionic bond
C) Metallic bond
D) Hydrogen bond
Answer: B - Ionic compounds have:
A) Low melting point
B) High melting point
C) No melting point
D) Variable
Answer: B - Ionic compounds conduct electricity in:
A) Solid state
B) Molten state
C) Gas state
D) None
Answer: B - Sodium loses:
A) One electron
B) Two electrons
C) Three electrons
D) Four electrons
Answer: A
- Chlorine gains:
A) 1 electron
B) 2 electrons
C) 3 electrons
D) 4 electrons
Answer: A - Diamond is an allotrope of:
A) Sulphur
B) Carbon
C) Oxygen
D) Nitrogen
Answer: B - Coal is mainly composed of:
A) Oxygen
B) Nitrogen
C) Carbon
D) Sulphur
Answer: C - Sulphur is used in the manufacture of:
A) Nitric acid
B) Hydrochloric acid
C) Sulphuric acid
D) Acetic acid
Answer: C - Nitrogen is mainly used in making:
A) Plastics
B) Fertilizers
C) Paints
D) Soaps
Answer: B - Oxygen supports:
A) Photosynthesis
B) Respiration only
C) Combustion
D) Corrosion
Answer: C - Most metals form:
A) Anions
B) Cations
C) Neutral atoms
D) Molecules
Answer: B - Most non-metals form:
A) Cations
B) Anions
C) Protons
D) Neutrons
Answer: B - Aluminium ore is:
A) Haematite
B) Bauxite
C) Galena
D) Zinc blende
Answer: B - Iron ore is:
A) Bauxite
B) Haematite
C) Limestone
D) Graphite
Answer: B - Zinc ore is:
A) Zinc blende
B) Galena
C) Magnetite
D) Calcite
Answer: A - Corrosion of silver forms:
A) Silver oxide
B) Silver nitrate
C) Silver sulphide
D) Silver chloride
Answer: C - Green coating on copper is:
A) Copper oxide
B) Copper sulphate
C) Copper carbonate
D) Basic copper carbonate
Answer: D - A metal that produces sound when struck is called:
A) Ductile
B) Malleable
C) Sonorous
D) Hard
Answer: C - Potassium is stored in:
A) Water
B) Air
C) Kerosene
D) Alcohol
Answer: C - Sodium reacts with water:
A) Slowly
B) Moderately
C) Violently
D) Not at all
Answer: C - Metals are electropositive because they:
A) Gain electrons
B) Lose electrons
C) Share electrons
D) Accept protons
Answer: B - Non-metals are electronegative because they:
A) Gain electrons
B) Lose electrons
C) Share neutrons
D) Lose protons
Answer: A - The hardest natural substance is:
A) Iron
B) Graphite
C) Diamond
D) Gold
Answer: C - The most malleable metal is:
A) Iron
B) Copper
C) Silver
D) Gold
Answer: D - The best conductor of electricity is:
A) Copper
B) Gold
C) Silver
D) Aluminium
Answer: C - The lightest metal is:
A) Sodium
B) Lithium
C) Magnesium
D) Potassium
Answer: B - An example of amphoteric oxide is:
A) Na₂O
B) MgO
C) Al₂O₃
D) CO₂
Answer: C - Oxides of metals are generally:
A) Acidic
B) Basic
C) Neutral
D) Amphoteric only
Answer: B - Oxides of non-metals are generally:
A) Basic
B) Acidic
C) Neutral
D) Metallic
Answer: B - Alloying improves:
A) Color only
B) Strength
C) Weight
D) Density only
Answer: B - Rusting requires:
A) Water only
B) Air only
C) Air and moisture
D) Sunlight
Answer: C - Extraction of aluminium requires:
A) Heating
B) Roasting
C) Electrolysis
D) Filtration
Answer: C - Metals are mostly solids except:
A) Bromine
B) Mercury
C) Iodine
D) Sodium
Answer: B - Bromine is:
A) Solid metal
B) Liquid non-metal
C) Gas metal
D) Solid non-metal
Answer: B - Covalent bond involves:
A) Transfer of electrons
B) Sharing of electrons
C) Loss of electrons
D) Gain of electrons
Answer: B - Metal carbonates on heating give:
A) Oxygen
B) Hydrogen
C) Carbon dioxide
D) Nitrogen
Answer: C - Displacement reaction occurs when:
A) Less reactive replaces more reactive
B) More reactive replaces less reactive
C) Equal metals react
D) No reaction occurs
Answer: B - Silver nitrate reacts with copper forming:
A) Copper nitrate only
B) Silver metal
C) Copper oxide
D) No reaction
Answer: B - Sodium chloride is soluble in:
A) Oil
B) Alcohol
C) Water
D) Petrol
Answer: C - Ionic compounds are generally:
A) Soft
B) Liquid
C) Hard and brittle
D) Gaseous
Answer: C - Most reactive among K, Na, Mg is:
A) K
B) Na
C) Mg
D) Equal
Answer: A - Least reactive among Cu, Ag, Au is:
A) Cu
B) Ag
C) Au
D) Equal
Answer: C - Rust is:
A) Black
B) White
C) Green
D) Reddish brown
Answer: D - Carbon forms maximum compounds due to:
A) Valency
B) Catenation
C) Size
D) Density
Answer: B - Stainless steel resists:
A) Heat
B) Pressure
C) Corrosion
D) Electricity
Answer: C - Metals expand on heating:
A) True
B) False
C) Sometimes
D) Never
Answer: A - Non-metals are poor conductors except:
A) Diamond
B) Sulphur
C) Graphite
D) Phosphorus
Answer: C - Gold is used in jewellery because it is:
A) Reactive
B) Cheap
C) Non-reactive
D) Brittle
Answer: C - Bronze is mainly used for making:
A) Wires
B) Statues
C) Foil
D) Batteries
Answer: B - Iron reacts with steam to give:
A) Oxygen
B) Hydrogen
C) Nitrogen
D) Chlorine
Answer: B - NaCl is an example of:
A) Covalent compound
B) Ionic compound
C) Alloy
D) Metal
Answer: B - Diamond is used for cutting because it is:
A) Soft
B) Reactive
C) Hard
D) Brittle
Answer: C - Graphite is used in pencils because it is:
A) Hard
B) Slippery
C) Shiny
D) Strong
Answer: B - Chlorine is used for:
A) Cooking
B) Water purification
C) Painting
D) Welding
Answer: B - Most reactive metals react with water forming:
A) Oxide
B) Hydroxide and hydrogen
C) Salt only
D) Oxygen
Answer: B - Reduction means:
A) Loss of electrons
B) Gain of electrons
C) Loss of oxygen
D) Gain of hydrogen
Answer: B - Oxidation means:
A) Gain of electrons
B) Loss of electrons
C) Gain of neutrons
D) Loss of protons
Answer: B - Metals generally have:
A) 1–3 valence electrons
B) 5–7 valence electrons
C) 8 valence electrons
D) No electrons
Answer: A - Metals are generally:
A) Electronegative
B) Electropositive
C) Neutral
D) Inert
Answer: B
✅ 100 Very Short Answer Questions
🔹 Basic Concepts
- What are metals?
Answer: Elements that are generally hard, shiny, and good conductors of heat and electricity. - What are non-metals?
Answer: Elements that are generally dull, brittle, and poor conductors of heat and electricity. - What is lustre?
Answer: The shiny appearance of a metal. - What is malleability?
Answer: Ability of a metal to be beaten into thin sheets. - What is ductility?
Answer: Ability of a metal to be drawn into wires. - What is sonority?
Answer: Property of producing sound when struck. - Name one liquid metal.
Answer: Mercury. - Name one liquid non-metal.
Answer: Bromine. - Name the hardest natural substance.
Answer: Diamond. - Which non-metal conducts electricity?
Answer: Graphite.
🔹 Chemical Properties
- What is formed when metals react with oxygen?
Answer: Metal oxides. - What is the nature of metal oxides?
Answer: Basic. - What is formed when non-metals react with oxygen?
Answer: Non-metal oxides. - What is the nature of non-metal oxides?
Answer: Acidic. - What gas is evolved when metals react with dilute acids?
Answer: Hydrogen gas. - Write one example of a displacement reaction.
Answer: Zn + CuSO₄ → ZnSO₄ + Cu. - What is corrosion?
Answer: Slow destruction of metals due to air and moisture. - What is rust?
Answer: Hydrated iron(III) oxide. - What is galvanization?
Answer: Coating iron with zinc to prevent rusting. - What is oxidation?
Answer: Loss of electrons.
🔹 Reactivity Series
- Which is the most reactive metal?
Answer: Potassium. - Which is the least reactive metal?
Answer: Gold. - What is reactivity series?
Answer: Arrangement of metals in decreasing order of reactivity. - Can copper displace iron?
Answer: No. - Can zinc displace copper?
Answer: Yes.
🔹 Extraction of Metals
- What is an ore?
Answer: A mineral from which metal can be extracted. - What is gangue?
Answer: Unwanted impurities in an ore. - What is concentration of ore?
Answer: Removal of gangue from ore. - What is roasting?
Answer: Heating ore in presence of air. - What is calcination?
Answer: Heating ore in absence of air. - How are highly reactive metals extracted?
Answer: By electrolysis. - Name the furnace used for extraction of iron.
Answer: Blast furnace. - What is metallurgy?
Answer: Process of extracting metals from ores. - What is refining?
Answer: Purification of metal. - Which metal is extracted from bauxite?
Answer: Aluminium.
🔹 Alloys
- What is an alloy?
Answer: Mixture of two or more metals or metal and non-metal. - What is brass made of?
Answer: Copper and zinc. - What is bronze made of?
Answer: Copper and tin. - What is steel made of?
Answer: Iron and carbon. - Why are alloys used?
Answer: To improve strength and prevent corrosion.
🔹 Ionic Compounds
- What is an ionic bond?
Answer: Bond formed by transfer of electrons. - What type of bond is present in NaCl?
Answer: Ionic bond. - Do ionic compounds have high melting point?
Answer: Yes. - When do ionic compounds conduct electricity?
Answer: In molten or aqueous state. - What ion is formed by sodium?
Answer: Na⁺. - What ion is formed by chlorine?
Answer: Cl⁻. - What is valency of sodium?
Answer: 1. - What is valency of oxygen?
Answer: 2. - What type of bond is formed between two non-metals?
Answer: Covalent bond. - What is covalent bond?
Answer: Bond formed by sharing of electrons.
🔹 Important Metals
- What is the chemical symbol of iron?
Answer: Fe. - What is the symbol of copper?
Answer: Cu. - What is the symbol of aluminium?
Answer: Al. - Which metal is used in electrical wiring?
Answer: Copper. - Which metal is used in aircraft bodies?
Answer: Aluminium. - Which metal is most malleable?
Answer: Gold. - Which metal is best conductor of electricity?
Answer: Silver. - Which metal reacts violently with water?
Answer: Sodium. - Which metal is stored in kerosene?
Answer: Potassium. - Which metal forms rust?
Answer: Iron.
🔹 Important Non-Metals
- What is the chemical symbol of carbon?
Answer: C. - What is the symbol of nitrogen?
Answer: N. - What is the symbol of oxygen?
Answer: O. - Which non-metal is essential for respiration?
Answer: Oxygen. - Which non-metal is used in fertilizers?
Answer: Nitrogen. - Which non-metal is used in water purification?
Answer: Chlorine. - Name two allotropes of carbon.
Answer: Diamond and graphite. - Which allotrope of carbon is soft?
Answer: Graphite. - Which allotrope of carbon is hardest?
Answer: Diamond. - Which non-metal is yellow in color?
Answer: Sulphur.
🔹 Applications & Miscellaneous
- Why are metals good conductors?
Answer: Due to presence of free electrons. - Why are non-metals poor conductors?
Answer: They lack free electrons. - What is amphoteric oxide?
Answer: Oxide that reacts with both acids and bases. - Give one example of amphoteric oxide.
Answer: Aluminium oxide. - What is reduction?
Answer: Gain of electrons. - What happens when iron reacts with steam?
Answer: Forms iron oxide and hydrogen gas. - What is the colour of rust?
Answer: Reddish brown. - Why is gold used in jewellery?
Answer: It is non-reactive and lustrous. - What is catenation?
Answer: Ability of carbon to form long chains. - Which metal is used for making statues?
Answer: Bronze.
🔹 Higher Order Quick Facts
- Which metal is least reactive?
Answer: Gold. - Which metal is highly reactive?
Answer: Potassium. - What happens when metal carbonates are heated?
Answer: They give metal oxide and CO₂. - What is the formula of rust?
Answer: Fe₂O₃·xH₂O. - What is electrolysis?
Answer: Process using electricity to extract metal. - Which bond involves sharing of electrons?
Answer: Covalent bond. - Which bond involves transfer of electrons?
Answer: Ionic bond. - Which non-metal is a gas at room temperature?
Answer: Oxygen. - Which metal is a liquid at room temperature?
Answer: Mercury. - What type of ions do metals form?
Answer: Positive ions (cations). - What type of ions do non-metals form?
Answer: Negative ions (anions). - Why is stainless steel rust-resistant?
Answer: Due to chromium content. - What protects aluminium from corrosion?
Answer: Thin oxide layer. - Which metal is used in making utensils?
Answer: Aluminium. - Which non-metal is used in matchsticks?
Answer: Phosphorus. - What is the valency of magnesium?
Answer: 2. - What is the valency of chlorine?
Answer: 1. - What happens when sodium reacts with chlorine?
Answer: Forms sodium chloride. - What is the nature of CO₂?
Answer: Acidic. - What is the nature of MgO?
Answer: Basic.
🔹 Physical Properties
- Describe the physical properties of metals.
Metals are generally hard, lustrous, malleable, ductile, and sonorous. They conduct heat and electricity efficiently. Most metals have high melting and boiling points and are solid at room temperature, except mercury. These properties make them suitable for construction and electrical uses. - Why are metals good conductors of electricity?
Metals contain free-moving electrons in their outer shell. These electrons move easily when electric current passes, allowing the flow of electricity. This property makes metals like copper and aluminium useful in electrical wiring and appliances. - Explain malleability with example.
Malleability is the ability of a metal to be beaten into thin sheets without breaking. Gold and aluminium are highly malleable metals. This property is useful in making aluminium foil and gold leaves for decoration. - Explain ductility with example.
Ductility is the property of metals to be drawn into thin wires. Copper is highly ductile and is used in electrical wiring. This property helps in making cables and transmission lines. - Why are non-metals brittle?
Non-metals do not have free electrons and lack metallic bonding. Therefore, they cannot withstand hammering or stretching. When force is applied, they break easily. Sulphur and phosphorus are common examples of brittle non-metals.
🔹 Chemical Properties
- Explain reaction of metals with oxygen.
Metals react with oxygen to form metal oxides. For example, magnesium burns in air to form magnesium oxide. These oxides are generally basic in nature and react with acids to form salts and water. - Explain reaction of metals with water.
Highly reactive metals like sodium react vigorously with cold water to form metal hydroxide and hydrogen gas. Less reactive metals like iron react with steam to produce metal oxide and hydrogen gas. - Describe reaction of metals with acids.
Metals react with dilute acids to form salt and hydrogen gas. For example, zinc reacts with hydrochloric acid to produce zinc chloride and hydrogen. This reaction shows metals are electropositive in nature. - Explain reaction of non-metals with oxygen.
Non-metals react with oxygen to form non-metal oxides, which are generally acidic. For example, carbon burns in oxygen to form carbon dioxide, which dissolves in water to form carbonic acid. - What is displacement reaction? Give example.
A displacement reaction occurs when a more reactive metal displaces a less reactive metal from its salt solution. For example, zinc displaces copper from copper sulphate solution.
🔹 Reactivity Series
- What is reactivity series? Why is it important?
The reactivity series is the arrangement of metals in decreasing order of their reactivity. It helps predict displacement reactions and methods of extraction of metals. - Why is potassium more reactive than copper?
Potassium has only one valence electron and loses it easily to form a positive ion. Copper holds its electrons more tightly, so it is less reactive compared to potassium. - Why cannot copper displace zinc from zinc sulphate?
Copper is less reactive than zinc. According to the reactivity series, only a more reactive metal can displace a less reactive metal from its compound. - Why are gold and silver found in free state?
Gold and silver are least reactive metals. They do not react easily with air, water, or acids, so they are found in native or free state in nature. - Why are sodium and potassium stored in kerosene?
Sodium and potassium are highly reactive metals. They react vigorously with air and moisture. To prevent accidental reactions, they are stored under kerosene.
🔹 Extraction of Metals
- What is metallurgy?
Metallurgy is the process of extracting metals from their ores and refining them for use. It includes concentration, roasting, reduction, and refining. - What is concentration of ore?
Concentration of ore is the removal of impurities (gangue) from the ore before extraction. It increases the percentage of metal content in the ore. - Explain roasting and calcination.
Roasting is heating the ore in presence of air, while calcination is heating in absence of air. These processes remove volatile impurities and convert ore into oxide form. - How are highly reactive metals extracted?
Highly reactive metals like sodium and aluminium are extracted using electrolysis because they cannot be reduced by carbon. - Explain extraction of iron briefly.
Iron is extracted from haematite in a blast furnace. The ore is reduced using carbon monoxide to form molten iron.
(Continuing 21–100 in same format)
🔹 Alloys
- What is an alloy?
An alloy is a homogeneous mixture of two or more metals or a metal and a non-metal. Alloys are made to improve strength, hardness, and resistance to corrosion compared to pure metals. - Why is steel stronger than iron?
Steel contains iron mixed with carbon. Carbon increases hardness and strength, making steel stronger and more durable than pure iron. - Why is stainless steel rust-resistant?
Stainless steel contains chromium, which forms a thin protective oxide layer on the surface. This layer prevents corrosion and rusting. - Why is brass used for making utensils?
Brass is an alloy of copper and zinc. It is corrosion-resistant, strong, and easy to shape, making it suitable for utensils and decorative items. - Why are alloys preferred over pure metals?
Alloys are generally stronger, harder, and more resistant to corrosion than pure metals. They also have improved durability and practical applications.
🔹 Ionic Compounds
- What is an ionic bond?
An ionic bond is formed by the transfer of electrons from a metal to a non-metal. It results in the formation of oppositely charged ions that attract each other. - Why do ionic compounds have high melting points?
Ionic compounds have strong electrostatic forces between oppositely charged ions. Large energy is required to break these forces, so they have high melting points. - Why do ionic compounds conduct electricity in molten state?
In molten state, ions are free to move and carry electric charge. In solid state, ions are fixed and cannot conduct electricity. - Explain formation of NaCl.
Sodium loses one electron to form Na⁺, and chlorine gains one electron to form Cl⁻. The oppositely charged ions attract, forming ionic compound NaCl. - Difference between ionic and covalent bond.
Ionic bond involves transfer of electrons, while covalent bond involves sharing of electrons between atoms.
🔹 Uses and Properties of Metals
- Explain the properties and uses of iron.
Iron is a strong, hard metal with high tensile strength. It reacts with moisture to form rust. Iron is widely used in construction, making tools, bridges, railway tracks, and machinery because of its strength and durability. - Describe properties and uses of aluminium.
Aluminium is lightweight, corrosion-resistant, and a good conductor of heat and electricity. It forms a protective oxide layer. It is used in aircraft bodies, utensils, electrical cables, and packaging materials due to its light weight and strength. - Explain properties and uses of copper.
Copper is ductile, malleable, and an excellent conductor of electricity. It resists corrosion and has a reddish-brown color. It is widely used in electrical wiring, motors, coins, and plumbing materials. - Why is gold used in jewellery?
Gold is lustrous, highly malleable, ductile, and non-reactive. It does not corrode or tarnish easily. These properties make it ideal for making jewellery and decorative items. - Why is silver used in electrical contacts?
Silver is the best conductor of electricity among metals. It also has good thermal conductivity and resistance to corrosion, making it suitable for electrical switches and contacts.
🔹 Properties and Uses of Non-Metals
- Explain properties and uses of carbon.
Carbon exists in different forms like diamond and graphite. Diamond is hard, while graphite is soft and conducts electricity. Carbon is used in fuels, pencils, cutting tools, and in making organic compounds. - Describe properties and uses of oxygen.
Oxygen is a colorless, odorless gas essential for respiration and combustion. It supports burning and is used in hospitals for patients and in welding processes. - Explain importance of nitrogen.
Nitrogen is a colorless, inert gas that forms about 78% of the atmosphere. It is used in fertilizers, food preservation, and manufacturing ammonia. - Describe properties and uses of sulphur.
Sulphur is a yellow, brittle non-metal. It is used in making sulphuric acid, medicines, rubber vulcanization, and fertilizers. - Why is chlorine used for water purification?
Chlorine is a reactive non-metal that kills harmful bacteria and microorganisms in water. It is widely used to disinfect drinking water and swimming pools.
🔹 Corrosion and Prevention
- What is rusting of iron?
Rusting is the formation of reddish-brown hydrated iron oxide when iron reacts with oxygen and moisture. It weakens the metal and damages structures. - What conditions are necessary for rusting?
Rusting requires both oxygen and water (moisture). In the absence of either, rusting does not occur. - How does painting prevent rusting?
Painting forms a protective layer over iron, preventing contact with air and moisture. This stops the oxidation process and prevents rust formation. - What is galvanization?
Galvanization is coating iron with a thin layer of zinc. Zinc protects iron by preventing exposure to air and moisture, reducing corrosion. - Why is aluminium resistant to corrosion?
Aluminium forms a thin protective oxide layer on its surface when exposed to air. This layer prevents further reaction with oxygen and protects it from corrosion.
🔹 Chemical Concepts
- Explain oxidation in terms of electrons.
Oxidation is the loss of electrons by an atom or ion during a chemical reaction. For example, sodium loses one electron to form Na⁺. - Explain reduction in terms of electrons.
Reduction is the gain of electrons by an atom or ion. For example, chlorine gains one electron to form Cl⁻. - What is an amphoteric oxide?
An amphoteric oxide reacts with both acids and bases. Aluminium oxide is an example as it reacts with hydrochloric acid and sodium hydroxide. - Why do metals form positive ions?
Metals have fewer valence electrons and tend to lose them easily to achieve stable configuration. Losing electrons makes them positively charged ions. - Why do non-metals form negative ions?
Non-metals have nearly full outer shells and tend to gain electrons to complete their octet, forming negatively charged ions.
🔹 Higher Understanding
- Why are metals generally solid at room temperature?
Metals have strong metallic bonds due to attraction between positive ions and free electrons. This strong bonding keeps them solid at room temperature. - Why are non-metals poor conductors of heat?
Non-metals lack free electrons to transfer energy. Therefore, heat cannot pass through them easily, making them poor conductors. - Why is graphite soft despite being a form of carbon?
In graphite, carbon atoms are arranged in layers held together by weak forces. These layers slide over each other easily, making graphite soft. - Why does diamond have high melting point?
Diamond has a strong three-dimensional network of covalent bonds. A large amount of energy is needed to break these bonds, giving it a high melting point. - Why do ionic compounds dissolve in water?
Water molecules attract and surround ions, separating them from the crystal lattice. This process helps ionic compounds dissolve in water. - Why do covalent compounds have low melting points?
Covalent compounds have weak intermolecular forces between molecules. Less energy is required to break these forces, so they have low melting points. - Explain displacement reaction with example.
In displacement reaction, a more reactive metal displaces a less reactive metal from its compound. Example: Zn + CuSO₄ → ZnSO₄ + Cu. - Why is potassium more reactive than magnesium?
Potassium has one valence electron and loses it easily. Magnesium has two valence electrons and holds them more strongly, so it is less reactive. - Why is copper less reactive than iron?
Copper lies below iron in the reactivity series. It does not react easily with acids or water compared to iron. - Why is electrolysis used for aluminium extraction?
Aluminium is highly reactive and cannot be reduced by carbon. Therefore, electrolysis is used to extract aluminium from its ore.
🔹 Reactions & Chemical Behavior
- Explain why sodium reacts vigorously with water.
Sodium is a highly reactive metal with one valence electron. It loses this electron easily and reacts rapidly with water to form sodium hydroxide and hydrogen gas. The reaction releases a large amount of heat, making it vigorous. - Why does magnesium react slowly with cold water but faster with hot water?
Magnesium is less reactive than sodium. With cold water, it reacts very slowly, but when heated, the reaction speeds up. Higher temperature increases kinetic energy, making magnesium react more quickly. - Explain the reaction of iron with steam.
Iron does not react with cold water but reacts with steam to form iron oxide and hydrogen gas. The reaction occurs at high temperature and demonstrates moderate reactivity of iron. - Why are non-metals generally electronegative?
Non-metals have nearly complete valence shells. They tend to gain electrons to achieve stable configuration. This ability to attract electrons makes them electronegative. - Why are metals electropositive?
Metals have fewer valence electrons and lose them easily to form positive ions. This tendency to donate electrons makes them electropositive.
🔹 Extraction & Metallurgy
- Why is carbon used in reduction of some metals?
Carbon is a good reducing agent. It removes oxygen from metal oxides to produce pure metal. This method is suitable for moderately reactive metals like iron and zinc. - Why cannot sodium be extracted by reduction with carbon?
Sodium is highly reactive and forms very stable compounds. Carbon cannot reduce sodium oxide effectively. Therefore, electrolysis is used to extract sodium. - What is electrolytic refining?
Electrolytic refining is a purification process where impure metal is made anode and pure metal is cathode. Electric current is passed to obtain pure metal deposited at cathode. - Why is aluminium extraction costly?
Aluminium extraction requires electrolysis, which consumes large amounts of electricity. Hence, the process is energy-intensive and expensive. - Explain the role of flux in metallurgy.
Flux is added during smelting to remove impurities. It reacts with gangue to form slag, which can be easily separated from molten metal.
🔹 Alloys & Applications
- Why is stainless steel preferred over iron?
Stainless steel contains chromium and nickel, which make it corrosion-resistant and stronger. It does not rust easily, so it is widely used in utensils and medical instruments. - Why is bronze used for statues?
Bronze is hard, corrosion-resistant, and durable. It can be easily cast into shapes, making it ideal for statues and sculptures. - Why is aluminium used in aircraft?
Aluminium is lightweight and corrosion-resistant. Despite being light, it is strong when alloyed. These properties make it suitable for aircraft manufacturing. - Why are copper wires used in electricity transmission?
Copper has high electrical conductivity and ductility. It allows efficient transmission of electricity with minimal energy loss. - Why is gold mixed with copper for jewellery?
Pure gold is soft. Mixing copper increases hardness and durability while maintaining lustre, making it suitable for jewellery.
🔹 Corrosion & Protection
- How does oiling prevent rusting?
Oiling forms a protective layer over metal, preventing contact with air and moisture. This stops oxidation and prevents rust formation. - How does electroplating protect metals?
Electroplating coats a metal with another metal layer using electricity. This layer protects the base metal from corrosion and improves appearance. - Why does silver tarnish?
Silver reacts with sulphur compounds in air to form silver sulphide, which appears as a black coating on its surface. - Why does copper turn green over time?
Copper reacts with carbon dioxide, moisture, and oxygen forming basic copper carbonate, which appears green in color. - Why is rusting harmful?
Rusting weakens iron structures, reducing strength and durability. It can cause structural damage and economic loss.
🔹 Bonding & Compounds
- Why do ionic compounds have high boiling points?
Strong electrostatic forces exist between oppositely charged ions. A large amount of energy is required to separate them, resulting in high boiling points. - Why do ionic compounds conduct electricity in solution?
When dissolved in water, ionic compounds dissociate into free ions. These ions move freely and carry electric charge. - Why are covalent compounds poor conductors?
Covalent compounds do not have free ions or electrons. Therefore, they cannot conduct electricity effectively. - Why is NaCl stable?
Sodium achieves stable configuration by losing one electron, and chlorine gains one. The electrostatic attraction between Na⁺ and Cl⁻ makes NaCl stable. - Explain the formation of magnesium oxide.
Magnesium loses two electrons to form Mg²⁺, and oxygen gains two electrons to form O²⁻. The oppositely charged ions attract, forming MgO.
🔹 Comparison & Reasoning
- Why are metals generally harder than non-metals?
Metals have strong metallic bonding due to free electrons. Non-metals lack this bonding, making them softer and brittle. - Why is diamond harder than graphite?
Diamond has a strong three-dimensional network of covalent bonds. Graphite has layered structure with weak forces between layers. - Why do metals expand on heating?
Heat increases kinetic energy of particles, causing atoms to move apart slightly, leading to expansion. - Why are non-metals often gases?
Non-metals have weak intermolecular forces. Therefore, many exist as gases at room temperature. - Why are metals shiny?
Metals reflect light due to free electrons on their surface, giving them a lustrous appearance.
🔹 Environmental & Daily Life
- How does mining affect environment?
Mining causes deforestation, soil erosion, and pollution. It disturbs ecological balance and damages habitats. - Why is recycling metals important?
Recycling reduces mining, saves energy, conserves resources, and decreases environmental pollution. - How are metals important in daily life?
Metals are used in buildings, vehicles, appliances, and tools due to their strength and conductivity. - How are non-metals important in daily life?
Non-metals like oxygen, nitrogen, and carbon are essential for life, fertilizers, and industrial processes. - Why is oxygen necessary for combustion?
Oxygen supports burning by reacting with substances to release energy in the form of heat and light.
🔹 Advanced Concepts
- What is catenation and why is carbon special?
Catenation is the ability of an element to form long chains with itself. Carbon shows strong catenation, forming millions of compounds. - Why does aluminium not rust like iron?
Aluminium forms a thin oxide layer that protects it from further oxidation, unlike iron which forms porous rust. - Why is mercury liquid at room temperature?
Mercury has weak metallic bonding compared to other metals, resulting in low melting point. - Why is graphite used as lubricant?
Graphite layers slide easily over one another due to weak interlayer forces, making it a good lubricant. - Why are metals essential for industrial development?
Metals provide strength, conductivity, and durability required for machines, infrastructure, transportation, and technology, making them vital for industrial growth.
1. Describe the physical and chemical properties of metals.
Answer:
Metals are generally hard, shiny, malleable, ductile, and good conductors of heat and electricity. They have high melting and boiling points and are sonorous in nature. Most metals are solids at room temperature except mercury. Chemically, metals tend to lose electrons and form positive ions (cations). They react with oxygen to form metal oxides, which are mostly basic in nature. Metals react with water to produce metal hydroxides and hydrogen gas. They also react with dilute acids to form salts and hydrogen gas. Some metals, like sodium and potassium, are highly reactive and stored in kerosene. Less reactive metals like gold and platinum are found in native form. Metals also form ionic compounds by transferring electrons to non-metals.
2. Explain the reactivity series of metals and its importance.
Answer:
The reactivity series is a list of metals arranged in decreasing order of their reactivity. It includes metals like potassium, sodium, calcium, magnesium, aluminum, zinc, iron, lead, copper, mercury, silver, gold, and platinum. Highly reactive metals are placed at the top, while least reactive metals are at the bottom. This series helps predict the outcome of displacement reactions. A metal higher in the series can displace a metal lower in the series from its compound. It also helps in understanding extraction methods of metals. Highly reactive metals are extracted by electrolysis, moderately reactive metals by reduction with carbon, and less reactive metals by heating their ores. Thus, the reactivity series is important in predicting chemical behavior and industrial extraction processes.
3. Describe the extraction of metals from their ores.
Answer:
The extraction of metals involves three main steps: concentration of ore, conversion to oxide, and reduction. First, impurities are removed by methods like handpicking, washing, or froth flotation. Next, ores are converted into metal oxides by roasting (heating in air) or calcination (heating in absence of air). Finally, the metal oxide is reduced to metal. Highly reactive metals such as sodium and aluminum are extracted by electrolysis of molten compounds. Moderately reactive metals like zinc and iron are reduced using carbon or coke. Less reactive metals like mercury and copper are obtained by heating their ores directly. After extraction, metals are purified by methods such as electrolytic refining. This process ensures pure metal for industrial use.
4. Explain corrosion and its prevention methods.
Answer:
Corrosion is the slow destruction of metals due to reaction with air, moisture, or other chemicals. Rusting of iron is a common example, where iron reacts with oxygen and water to form hydrated iron oxide. Corrosion weakens metals and causes economic loss. To prevent corrosion, several methods are used. Painting, greasing, and oiling create a protective layer that prevents contact with air and moisture. Galvanization involves coating iron with zinc to prevent rusting. Alloying is another method where metals are mixed to improve resistance, such as stainless steel. Electroplating coats one metal with another using electricity. Proper storage and regular maintenance also help reduce corrosion. Prevention methods increase the life and durability of metal objects.
5. Differentiate between metals and non-metals.
Answer:
Metals are generally hard, shiny, malleable, ductile, and good conductors of heat and electricity. They lose electrons to form positive ions and usually form basic oxides. Metals react with acids to release hydrogen gas. Examples include iron, copper, and aluminum. Non-metals, on the other hand, are usually dull, brittle (if solid), and poor conductors of heat and electricity, except graphite. They gain or share electrons and form negative ions or covalent bonds. Non-metal oxides are usually acidic or neutral. Non-metals do not react with dilute acids to produce hydrogen gas. Examples include oxygen, sulfur, and chlorine. Thus, metals and non-metals differ in physical properties, chemical behavior, and electron transfer tendencies.




Leave a Reply