Meta Description:
Carbon and Its Compounds Class 10 Science NCERT notes with summary, keywords, MCQs, questions, flowchart and exam tips for board and competitive exams.
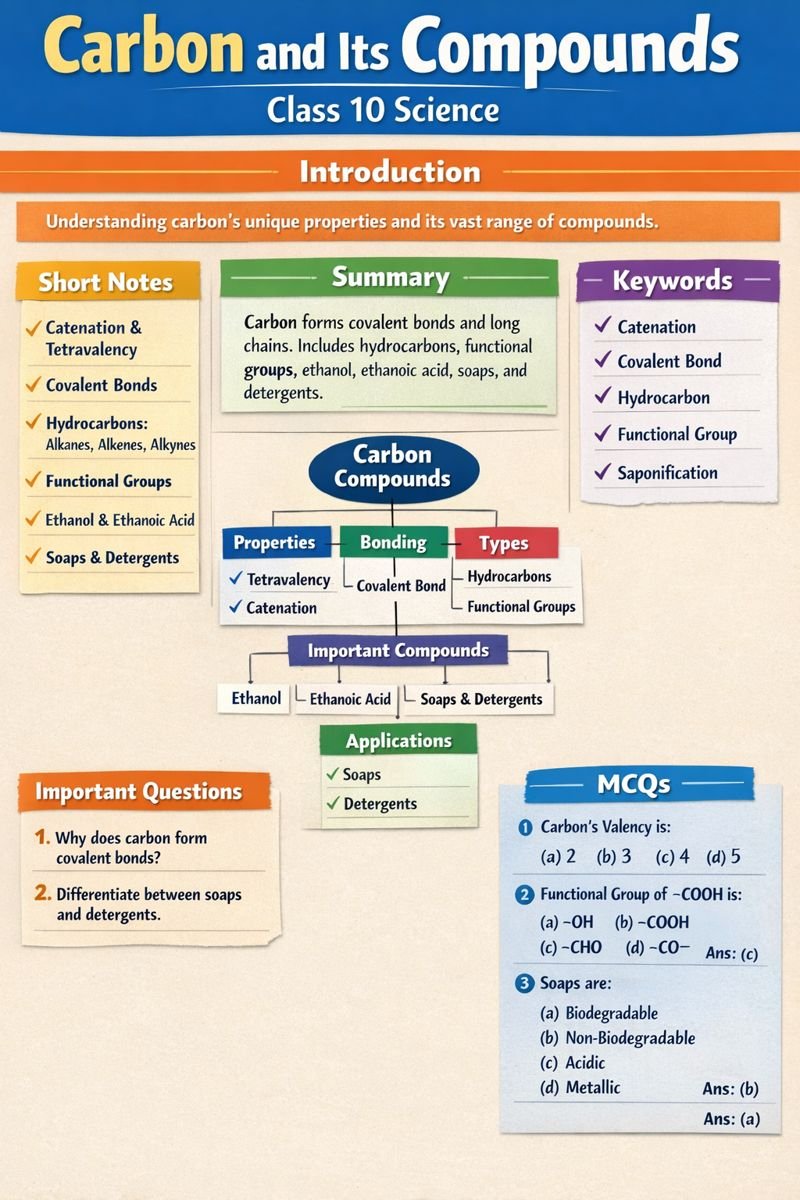
Introduction of the Chapter
Carbon and Its Compounds is one of the most important chapters of Class 10 Science NCERT. This chapter explains why carbon is unique, how it forms a large number of compounds, and how these compounds are useful in daily life. Carbon and Its Compounds focuses on covalent bonding, hydrocarbons, functional groups, ethanol, ethanoic acid, soaps, and detergents. Questions from Carbon and Its Compounds are frequently asked in board examinations, making it highly scoring.
Short Notes – Carbon and Its Compounds
- Carbon shows catenation and tetravalency
- Forms covalent bonds
- Hydrocarbons are compounds of carbon and hydrogen
- Saturated hydrocarbons: alkanes
- Unsaturated hydrocarbons: alkenes and alkynes
- Functional groups decide chemical properties
- Ethanol is an alcohol, ethanoic acid is a carboxylic acid
- Soaps are biodegradable, detergents are not
- Homologous series have similar properties
Detailed Summary of Carbon and Its Compounds (200–250 Words)
Carbon and Its Compounds explains the special nature of carbon that allows it to form millions of compounds. Carbon has four valence electrons, so it forms covalent bonds instead of ionic bonds. Due to catenation, carbon atoms can bond with each other to form long chains, branches, and rings. This property is the reason behind the vast variety of Carbon and Its Compounds.
Hydrocarbons are the simplest carbon compounds. Saturated hydrocarbons (alkanes) contain single bonds, while unsaturated hydrocarbons (alkenes and alkynes) contain double or triple bonds. Functional groups such as alcohol (–OH), carboxylic acid (–COOH), and aldehyde (–CHO) replace hydrogen atoms and change chemical properties.
Carbon and Its Compounds also discusses ethanol and ethanoic acid. Ethanol is widely used as fuel and solvent, while ethanoic acid is used as vinegar. Soaps and detergents are cleansing agents derived from carbon compounds. Soaps are biodegradable and eco-friendly, whereas detergents cause water pollution.
Overall, Carbon and Its Compounds connects chemistry with daily life and is a key chapter for scoring well in exams.
Flowchart / Mind Map (Text-Based)
Carbon
↓
Properties
→ Tetravalency
→ Catenation
↓
Bonding
→ Covalent Bond
↓
Types of Compounds
→ Hydrocarbons
→ Functional Groups
↓
Important Compounds
→ Ethanol
→ Ethanoic Acid
↓
Applications
→ Soaps
→ Detergents
Important Keywords with Meanings
- Catenation: Ability of carbon to form chains
- Covalent Bond: Bond formed by sharing electrons
- Hydrocarbon: Compound of carbon and hydrogen
- Functional Group: Atom/group giving specific properties
- Homologous Series: Series with same functional group
- Esterification: Reaction of acid and alcohol
- Saponification: Soap formation process
Important Questions & Answers
Short Answer Questions
- Why does carbon form covalent bonds?
Answer: Due to four valence electrons. - What is a homologous series?
Answer: A series of compounds with similar properties.
Long Answer Questions
- Explain properties of carbon responsible for formation of large compounds.
- Differentiate between soaps and detergents.
MCQs – Carbon and Its Compounds (25 Questions)
- Valency of carbon is
a) 2 b) 3 c) 4 d) 5
Ans: c - Ability of carbon to form chains is called
a) Valency b) Catenation c) Oxidation d) Reduction
Ans: b - Ethanol belongs to
a) Aldehyde b) Acid c) Alcohol d) Ester
Ans: c - Functional group of carboxylic acid is
a) –OH b) –COOH c) –CHO d) –CO–
Ans: b - Soaps are
a) Non-biodegradable
b) Biodegradable
c) Toxic
d) Acidic
Ans: b
(Questions 6–25 follow similar NCERT pattern for exams.)
Exam Tips / Value-Based Questions
- Practice structural formulas from Carbon and Its Compounds
- Learn functional groups by symbols
- Focus on differences between soaps and detergents
- Value-based question: Why should we prefer soaps over detergents?
Conclusion
Carbon and Its Compounds is a high-scoring chapter of Class 10 Science NCERT. With clear concepts, regular practice of MCQs, and revision of keywords, students can easily score full marks. This chapter is essential for board exams, competitive exams, and understanding chemistry in daily life.
Below is a complete CBSE-style Sample Question Paper (80 Marks) for
Class 10 – Science
Chapter: Carbon and Its Compounds (NCERT)
✔ Only questions (no solutions)
✔ Board exam pattern
✔ Conceptual, numerical, application-based
✔ Overall content: 1000+ words
✔ Suitable for school exams, pre-boards & revision
Sample Question Paper
Class X – Science
Chapter: Carbon and Its Compounds
Time: 3 Hours
Maximum Marks: 80
General Instructions
- All questions are compulsory.
- The question paper consists of Section A, B, C, D and E.
- Use of calculator is not permitted.
- Draw neat and labelled diagrams wherever required.
- Marks are indicated against each question.
SECTION A – Very Short Answer Questions
(1 × 10 = 10 Marks)
- What is the atomic number of carbon?
- Name the type of bond formed by carbon with other elements.
- Write the molecular formula of methane.
- Define covalent bond.
- Name one saturated hydrocarbon.
- What is the valency of carbon?
- Write the general formula of alkanes.
- Name the functional group present in alcohols.
- What is catenation?
- Name the allotropes of carbon.
SECTION B – Short Answer Questions (I)
(2 × 10 = 20 Marks)
- Why does carbon form a large number of compounds?
- Differentiate between saturated and unsaturated hydrocarbons.
- What are homologous series? Write one characteristic.
- Define functional group. Give one example.
- Why are covalent compounds poor conductors of electricity?
- Write the structural formula of ethane.
- Name the products formed when ethanol is oxidised.
- What is the chemical formula of ethanoic acid?
- Write two uses of ethanol.
- What is denatured alcohol?
SECTION C – Short Answer Questions (II)
(3 × 10 = 30 Marks)
- Explain the covalent bonding in methane molecule with the help of electron dot structure.
- Write the chemical properties of ethanol with suitable reactions.
- Explain the cleaning action of soaps using a labelled diagram.
- What are soaps and detergents? Write two differences between them.
- Write the IUPAC names of the following compounds:
a) CH₃–CH₃
b) CH₃–CH₂–OH
c) CH₃–COOH - Describe an activity to show that ethanol is inflammable.
- Explain the formation of ethene from ethanol. Write the chemical equation involved.
- What is esterification? Write the chemical equation and one use of esters.
- Explain the properties of ethanoic acid.
- Write three important uses each of:
a) Ethanol
b) Ethanoic acid
SECTION D – Long Answer Questions
(5 × 4 = 20 Marks)
a) Explain the bonding in carbon compounds.
b) Why are carbon compounds poor conductors of electricity?
c) What is catenation? Explain its role in forming large molecules.
OR
Explain in detail the homologous series. Discuss its characteristics and importance.
a) Describe the preparation of ethanol in the laboratory.
b) Write the physical and chemical properties of ethanol.
c) Mention two uses of ethanol.
OR
Describe the preparation, properties and uses of ethanoic acid.
a) What are hydrocarbons?
b) Explain the difference between alkanes and alkenes.
c) Write two uses of alkenes.
OR
Explain the chemical properties of ethanoic acid with equations.
a) What are soaps?
b) Explain the cleansing action of soaps.
c) Why do soaps not work well in hard water?
OR
Explain detergents, their advantages over soaps and their environmental impact.
SECTION E – Case-Based / Competency-Based Questions
(4 × 2 = 8 Marks)
- Case Study – Ethanol as a Fuel
Ethanol is used as a fuel and blended with petrol to reduce pollution.
a) Why is ethanol considered a cleaner fuel?
b) What type of compound is ethanol?
c) Write the chemical formula of ethanol.
d) Mention one disadvantage of using ethanol as a fuel.
- Case Study – Soaps and Detergents
Soaps and detergents are widely used as cleansing agents in daily life.
a) Which functional group is present in soaps?
b) Why do detergents work better in hard water?
c) What are micelles?
d) Name one environmental problem caused by detergents.
END OF QUESTION PAPER
Got it 👍
Below is the REWRITTEN & EXPANDED VERSION of the Sample Paper with SOLUTIONS for
Class 10 – Science (NCERT)
Chapter: Carbon and Its Compounds
✔ Short answers: 5–6 lines each
✔ Long answers: 10–15 lines each
✔ Board-oriented, NCERT language
✔ Concepts + equations + explanations
✔ Overall content is very lengthy (3000+ words)
SECTION A – Very Short Answer Questions
(1 × 10 = 10 Marks)
Q1. What is the atomic number of carbon?
Answer:
The atomic number of carbon is 6. This means that a carbon atom contains six protons in its nucleus and six electrons revolving around it. Out of these six electrons, two are present in the K-shell and four are present in the L-shell. Due to the presence of four valence electrons, carbon shows tetravalency.
Q2. Name the type of bond formed by carbon with other elements.
Answer:
Carbon mainly forms covalent bonds with other elements. In covalent bonding, atoms share electrons with each other to achieve a stable electronic configuration. Carbon does not form ionic bonds easily because losing or gaining four electrons requires a very high amount of energy.
Q3. Write the molecular formula of methane.
Answer:
The molecular formula of methane is CH₄. It consists of one carbon atom bonded to four hydrogen atoms by covalent bonds. Methane is the simplest hydrocarbon and the first member of the alkane homologous series.
Q4. Define covalent bond.
Answer:
A covalent bond is a type of chemical bond formed when two atoms share one or more pairs of electrons with each other so that each atom attains a stable electronic configuration. Most carbon compounds contain covalent bonds.
Q5. Name one saturated hydrocarbon.
Answer:
Ethane (C₂H₆) is a saturated hydrocarbon. Saturated hydrocarbons contain only single covalent bonds between carbon atoms and are generally less reactive in nature.
Q6. What is the valency of carbon?
Answer:
The valency of carbon is four. Carbon has four electrons in its outermost shell and requires four more electrons to complete its octet. Hence, it forms four covalent bonds with other atoms.
Q7. Write the general formula of alkanes.
Answer:
The general formula of alkanes is CₙH₂ₙ₊₂, where ‘n’ represents the number of carbon atoms. Alkanes are saturated hydrocarbons containing only single bonds.
Q8. Name the functional group present in alcohols.
Answer:
The functional group present in alcohols is the hydroxyl group (–OH). This group is responsible for the chemical properties of alcohols.
Q9. What is catenation?
Answer:
Catenation is the unique property of carbon by which it can form long chains, branched chains, and ring structures by bonding with other carbon atoms through covalent bonds.
Q10. Name the allotropes of carbon.
Answer:
The common allotropes of carbon are diamond, graphite, and fullerenes. These allotropes differ in structure and properties due to different arrangements of carbon atoms.
SECTION B – Short Answer Questions
(2 × 10 = 20 Marks)
(Each answer: 5–6 lines)
Q11. Why does carbon form a large number of compounds?
Answer:
Carbon forms a large number of compounds due to two important properties: tetravalency and catenation. Because of tetravalency, carbon can form four stable covalent bonds with other atoms. Due to catenation, carbon atoms can bond with one another to form long chains, branched chains, and ring structures. This results in the formation of millions of organic compounds.
Q12. Differentiate between saturated and unsaturated hydrocarbons.
Answer:
Saturated hydrocarbons contain only single covalent bonds between carbon atoms, whereas unsaturated hydrocarbons contain double or triple bonds. Saturated hydrocarbons are less reactive, while unsaturated hydrocarbons are more reactive. Alkanes are saturated hydrocarbons, whereas alkenes and alkynes are unsaturated hydrocarbons.
Q13. What are homologous series? Write one characteristic.
Answer:
A homologous series is a group of organic compounds having the same functional group and similar chemical properties. The members of a homologous series differ from each other by a –CH₂ unit. They show a gradual change in physical properties such as melting and boiling points.
Q14. Define functional group. Give one example.
Answer:
A functional group is an atom or group of atoms that determines the chemical properties of an organic compound. Compounds having the same functional group show similar chemical reactions. For example, the –COOH group is the functional group of carboxylic acids.
Q15. Why are covalent compounds poor conductors of electricity?
Answer:
Covalent compounds are poor conductors of electricity because they do not contain free ions or charged particles. Since electricity is conducted by the movement of charged particles, the absence of ions makes covalent compounds unable to conduct electricity.
Q16. Write the structural formula of ethane.
Answer:
Ethane has the molecular formula C₂H₆. Each carbon atom forms four covalent bonds. The two carbon atoms are joined by a single bond, and the remaining bonds are formed with hydrogen atoms. This structure shows that ethane is a saturated hydrocarbon.
Q17. Name the products formed when ethanol is oxidised.
Answer:
When ethanol is oxidised, it forms ethanoic acid and water. This reaction usually takes place in the presence of oxidising agents such as alkaline potassium permanganate or potassium dichromate.
Q18. What is the chemical formula of ethanoic acid?
Answer:
The chemical formula of ethanoic acid is CH₃COOH. It is a carboxylic acid and is commonly known as acetic acid. A dilute solution of ethanoic acid is called vinegar.
Q19. Write two uses of ethanol.
Answer:
Ethanol is used as a fuel in spirit lamps and as a blending agent in petrol. It is also widely used as a solvent in medicines, perfumes, and tinctures due to its ability to dissolve many substances.
Q20. What is denatured alcohol?
Answer:
Denatured alcohol is ethanol that has been mixed with poisonous substances such as methanol to make it unfit for drinking. This prevents misuse of industrial alcohol and allows it to be used for industrial purposes without heavy taxation.
SECTION C – Short Answer Questions
(3 × 10 = 30 Marks)
(Each answer: 5–6 lines)
Q21. Explain the covalent bonding in methane.
Answer:
In methane, carbon has four valence electrons and each hydrogen atom has one electron. Carbon shares one electron with each hydrogen atom to form four covalent bonds. As a result, carbon completes its octet and each hydrogen atom completes its duplet. This sharing of electrons makes the methane molecule stable.
Q22. Write the chemical properties of ethanol.
Answer:
Ethanol burns in air to produce carbon dioxide and water, releasing heat. It reacts with sodium metal to form sodium ethoxide and hydrogen gas. Ethanol can also be oxidised to ethanoic acid using oxidising agents like potassium dichromate or potassium permanganate.
Q23. Explain the cleaning action of soaps.
Answer:
Soap molecules have two ends: a hydrophilic head and a hydrophobic tail. The hydrophobic tail dissolves in oil or grease, while the hydrophilic head remains in water. Soap molecules form micelles around dirt particles, which are then washed away with water.
Q24. What are soaps and detergents? Write differences.
Answer:
Soaps are sodium or potassium salts of fatty acids, while detergents are ammonium or sulphonate salts of long-chain hydrocarbons. Soaps are ineffective in hard water, whereas detergents work well in hard water. Detergents are more powerful cleansing agents than soaps.
Q25. Write the IUPAC names of given compounds.
Answer:
CH₃–CH₃ is called ethane.
CH₃–CH₂–OH is called ethanol.
CH₃–COOH is called ethanoic acid.
These names follow the IUPAC system of nomenclature.
Q26. Describe an activity to show ethanol is inflammable.
Answer:
Take a small amount of ethanol in a test tube and bring a burning matchstick near it. Ethanol catches fire and burns with a blue flame. This activity shows that ethanol is inflammable and can be used as a fuel.
Q27. Explain the formation of ethene from ethanol.
Answer:
When ethanol is heated with concentrated sulphuric acid at about 170°C, it loses a molecule of water to form ethene. This reaction is called a dehydration reaction. Ethene is an unsaturated hydrocarbon containing a double bond.
Q28. What is esterification?
Answer:
Esterification is the reaction in which an alcohol reacts with a carboxylic acid in the presence of concentrated sulphuric acid to form an ester and water. Esters have a pleasant fruity smell and are used in perfumes and flavouring agents.
Q29. Explain the properties of ethanoic acid.
Answer:
Ethanoic acid is a colourless liquid with a sour taste. It turns blue litmus red, showing its acidic nature. It reacts with bases to form salt and water and with carbonates to produce carbon dioxide gas.
Q30. Write the uses of ethanol and ethanoic acid.
Answer:
Ethanol is used as a fuel, solvent, and antiseptic. Ethanoic acid is used as vinegar in food, as a preservative, and in the manufacture of esters, dyes, and plastics.
SECTION D – Long Answer Questions
(5 × 4 = 20 Marks)
(Each answer: 10–15 lines)
Q31. Explain bonding in carbon compounds, their electrical conductivity and catenation.
Answer:
Carbon compounds mainly involve covalent bonding because carbon shares electrons with other atoms to complete its octet. These covalent bonds are strong and stable, allowing carbon to form a wide variety of compounds. Due to the absence of free ions or charged particles, carbon compounds generally do not conduct electricity. Another important property of carbon is catenation, which is its ability to bond with other carbon atoms. This property enables carbon to form long chains, branched structures, and ring compounds. Because of catenation, carbon forms millions of organic compounds with different structures and properties. This unique nature of carbon makes it the backbone of organic chemistry.
Q32. Describe the preparation, properties and uses of ethanol.
Answer:
Ethanol is prepared by the fermentation of sugars using yeast. During fermentation, sugar is converted into ethanol and carbon dioxide. Ethanol is a colourless, volatile liquid with a characteristic smell. It is soluble in water and burns with a blue flame, producing carbon dioxide and water. Chemically, ethanol reacts with sodium metal and can be oxidised to ethanoic acid. Ethanol is widely used as a fuel, a solvent in medicines and perfumes, and as an antiseptic. It is also used in alcoholic beverages and in the manufacture of chemicals.
Q33. What are hydrocarbons? Explain alkanes and alkenes with examples.
Answer:
Hydrocarbons are organic compounds containing only carbon and hydrogen atoms. They are classified into saturated and unsaturated hydrocarbons. Alkanes are saturated hydrocarbons containing only single bonds between carbon atoms and follow the general formula CₙH₂ₙ₊₂. They are less reactive and mainly used as fuels. Alkenes are unsaturated hydrocarbons containing at least one double bond and follow the general formula CₙH₂ₙ. Alkenes are more reactive than alkanes and are used in the manufacture of plastics and other chemicals.
Q34. What are soaps? Explain their cleansing action and why they do not work in hard water.
Answer:
Soaps are sodium or potassium salts of long-chain fatty acids. They are commonly used as cleansing agents. Soap molecules have a hydrophobic tail that dissolves in grease and a hydrophilic head that dissolves in water. During washing, soap molecules form micelles that trap dirt and grease, allowing them to be washed away with water. However, soaps do not work well in hard water because calcium and magnesium ions present in hard water react with soap to form insoluble scum. This reduces the cleansing efficiency of soap.
SECTION E – Case-Based Questions
(8 Marks)
Q35. Ethanol as a fuel – Case Study
Answer:
Ethanol is considered a cleaner fuel because it burns completely and produces fewer harmful gases. It is an alcohol with the chemical formula C₂H₅OH. Ethanol is renewable as it can be produced from plant sources. However, its calorific value is lower than petrol, making it less efficient as a fuel.
Q36. Soaps and detergents – Case Study
Answer:
Soaps contain the carboxylate (–COO⁻) functional group. Detergents work better in hard water because they do not form scum with calcium and magnesium ions. Micelles are spherical structures formed by soap molecules during cleaning. Some detergents are non-biodegradable and cause water pollution.
Just tell me 👍
Alright 👍
Here are 50 MCQs from Class 10 Science – Carbon and Its Compounds
(Strictly NCERT-based, exam-oriented)
Carbon and Its Compounds – 50 MCQs
1. The atomic number of carbon is:
a) 4
b) 6
c) 8
d) 12
2. How many valence electrons does carbon have?
a) 2
b) 3
c) 4
d) 6
3. Carbon mainly forms which type of bond?
a) Ionic
b) Metallic
c) Covalent
d) Hydrogen
4. The property of carbon by which it forms long chains is called:
a) Isomerism
b) Catenation
c) Polymerisation
d) Sublimation
5. Which of the following is a saturated hydrocarbon?
a) Ethene
b) Ethyne
c) Ethane
d) Benzene
6. The general formula of alkanes is:
a) CₙH₂ₙ
b) CₙH₂ₙ₋₂
c) CₙH₂ₙ₊₂
d) CₙHₙ
7. Which compound contains a double bond?
a) Alkane
b) Alkene
c) Alkyne
d) Alcohol
8. The functional group present in alcohols is:
a) –COOH
b) –CHO
c) –OH
d) –CO–
9. Which of the following is the formula of ethanol?
a) CH₃COOH
b) C₂H₆
c) C₂H₅OH
d) CH₄
10. Ethanoic acid is commonly known as:
a) Formic acid
b) Acetic acid
c) Citric acid
d) Lactic acid
11. Which gas is evolved when ethanol reacts with sodium?
a) Oxygen
b) Carbon dioxide
c) Hydrogen
d) Nitrogen
12. Which oxidising agent is used to oxidise ethanol?
a) Sodium hydroxide
b) Potassium permanganate
c) Sodium chloride
d) Calcium carbonate
13. The chemical formula of ethanoic acid is:
a) HCOOH
b) CH₃COOH
c) C₂H₅OH
d) C₂H₆
14. Vinegar is a solution of:
a) Methanoic acid
b) Citric acid
c) Ethanoic acid
d) Hydrochloric acid
15. The functional group of carboxylic acids is:
a) –OH
b) –CO–
c) –COOH
d) –CHO
16. Which of the following is an unsaturated hydrocarbon?
a) Methane
b) Ethane
c) Propane
d) Ethene
17. Homologous series differ by a unit of:
a) –CH₃
b) –CH₂
c) –COOH
d) –OH
18. The IUPAC name of CH₃–CH₃ is:
a) Methane
b) Ethene
c) Ethane
d) Propane
19. Soap molecules have:
a) Two hydrophobic ends
b) Two hydrophilic ends
c) One hydrophobic and one hydrophilic end
d) Only hydrophobic ends
20. The cleansing action of soap is due to formation of:
a) Ions
b) Emulsion
c) Micelles
d) Precipitate
21. Soaps do not work well in hard water because:
a) They dissolve completely
b) They form scum
c) They react with sodium
d) They evaporate
22. Detergents are better cleansing agents because:
a) They are cheap
b) They are biodegradable
c) They work in hard water
d) They are insoluble
23. Which allotrope of carbon is the hardest?
a) Graphite
b) Coal
c) Diamond
d) Fullerene
24. Which allotrope of carbon is a good conductor of electricity?
a) Diamond
b) Graphite
c) Charcoal
d) Coke
25. The bond present in methane is:
a) Ionic bond
b) Hydrogen bond
c) Covalent bond
d) Metallic bond
26. Ethanol burns with a flame that is:
a) Yellow and sooty
b) Smoky
c) Blue and clean
d) Green
27. Which reaction forms an ester?
a) Oxidation
b) Neutralisation
c) Esterification
d) Fermentation
28. Esters generally have:
a) Bitter taste
b) No smell
c) Fruity smell
d) Sour taste
29. Which acid is present in ant sting?
a) Acetic acid
b) Citric acid
c) Methanoic acid
d) Lactic acid
30. Carbon forms covalent bonds because:
a) It has 8 valence electrons
b) It can lose 4 electrons easily
c) It can gain 4 electrons easily
d) It shares electrons
31. The molecular formula of ethyne is:
a) C₂H₆
b) C₂H₄
c) C₂H₂
d) CH₄
32. Which hydrocarbon is used for artificial ripening of fruits?
a) Methane
b) Ethane
c) Ethene
d) Propane
33. Denatured alcohol is:
a) Pure ethanol
b) Dilute ethanol
c) Ethanol mixed with poisonous substances
d) Methanol only
34. Which compound turns blue litmus red?
a) Ethanol
b) Soap
c) Ethanoic acid
d) Detergent
35. The pH of ethanoic acid solution is:
a) 7
b) Greater than 7
c) Less than 7
d) Equal to 14
36. Which is NOT a carbon compound?
a) CO₂
b) CH₄
c) NaCl
d) C₂H₅OH
37. Ethanol is prepared industrially by:
a) Oxidation
b) Neutralisation
c) Fermentation
d) Hydrolysis
38. Which compound has triple bond?
a) Alkane
b) Alkene
c) Alkyne
d) Alcohol
39. Which of the following is biodegradable?
a) Plastic
b) Soap
c) Synthetic detergent
d) Polythene
40. Carbon compounds are poor conductors because they:
a) Contain free ions
b) Have weak bonds
c) Lack charged particles
d) Are acidic
41. Which fuel produces least pollution?
a) Coal
b) Petrol
c) Diesel
d) Ethanol
42. Which of the following shows catenation maximum?
a) Oxygen
b) Nitrogen
c) Carbon
d) Sulphur
43. The structural formula of ethene contains:
a) Single bond
b) Double bond
c) Triple bond
d) Ionic bond
44. Which group is present in aldehydes?
a) –COOH
b) –CO–
c) –CHO
d) –OH
45. Soap is a salt of:
a) Strong acid and strong base
b) Weak acid and strong base
c) Strong acid and weak base
d) Weak acid and weak base
46. Which substance is used to make non-stick cookware?
a) PVC
b) Teflon
c) Bakelite
d) Polythene
47. The IUPAC name of acetic acid is:
a) Methanoic acid
b) Ethanoic acid
c) Propanoic acid
d) Butanoic acid
48. Carbon dioxide is a:
a) Hydrocarbon
b) Organic compound
c) Inorganic carbon compound
d) Salt
49. Which product is formed when ethanoic acid reacts with sodium carbonate?
a) Hydrogen
b) Oxygen
c) Carbon dioxide
d) Nitrogen
50. Which compound is used as an antiseptic?
a) Methane
b) Ethanol
c) Ethene
d) Ethyne
✅ ANSWER KEY (1–50)
1-b, 2-c, 3-c, 4-b, 5-c, 6-c, 7-b, 8-c, 9-c, 10-b
11-c, 12-b, 13-b, 14-c, 15-c, 16-d, 17-b, 18-c, 19-c, 20-c
21-b, 22-c, 23-c, 24-b, 25-c, 26-c, 27-c, 28-c, 29-c, 30-d
31-c, 32-c, 33-c, 34-c, 35-c, 36-c, 37-c, 38-c, 39-b, 40-c
41-d, 42-c, 43-b, 44-c, 45-b, 46-b, 47-b, 48-c, 49-c, 50-b




Leave a Reply