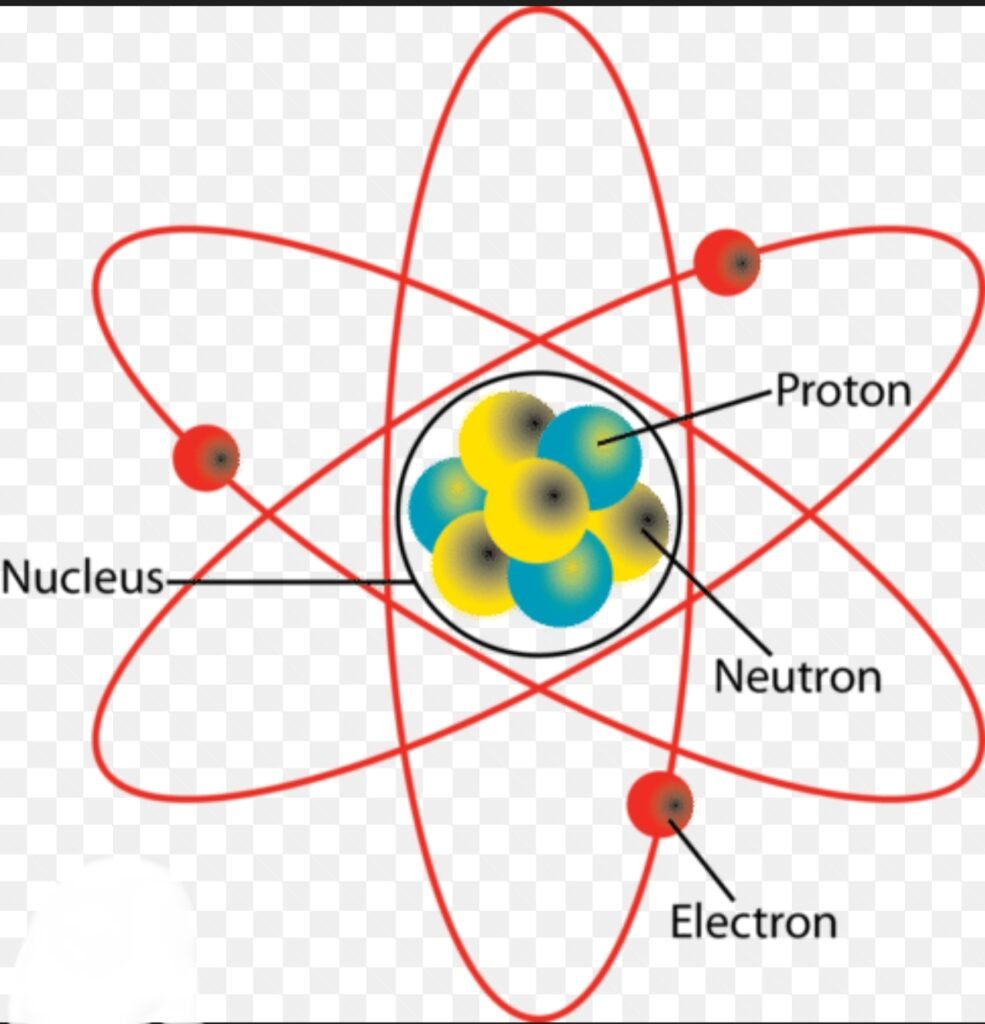
Atoms and Molecules: Class 9 Chemistry Complete Notes, Summary, and MCQs
Meta Description:
Explore Class 9 Chemistry chapter “Atoms and Molecules” with detailed summary, notes, MCQs, and important questions for exam preparation.
Introduction to Atoms and Molecules
The chapter Atoms and Molecules is a fundamental concept in Chemistry that helps us understand the building blocks of matter. Everything around us — from the air we breathe to the water we drink — is made up of atoms. Atoms combine in specific ways to form molecules, which constitute the substances we observe daily. This chapter lays the foundation for chemical reactions, stoichiometry, and the study of elements and compounds.
Understanding atoms and molecules is essential not only for scoring in exams but also for developing a scientific perspective of the world. The NCERT syllabus emphasizes simple explanations, real-life examples, and problem-solving approaches.
Short Notes: Atoms and Molecules
- Atom: Smallest particle of an element that retains its chemical properties.
- Molecule: Combination of two or more atoms bonded together.
- Law of Conservation of Mass: Mass is neither created nor destroyed in a chemical reaction.
- Law of Definite Proportions: A chemical compound always contains the same elements in a fixed ratio.
- Atomic Mass: Mass of an atom relative to carbon-12 standard.
- Molecular Mass: Sum of the atomic masses of all atoms in a molecule.
- Formula Unit: Simplest ratio of ions in an ionic compound.
- Chemical Reaction: Rearrangement of atoms to form new substances.
- Subatomic Particles: Electrons, protons, and neutrons make up an atom.
Detailed Summary of Atoms and Molecules (1000–1200 words)
The chapter Atoms and Molecules starts with the historical development of the concept of the atom. Ancient Greek philosopher Democritus first proposed that matter is made up of indivisible particles called “atoms.” This idea was later developed with experimental proof by scientists like Dalton, Thomson, Rutherford, and others.
Dalton’s Atomic Theory
John Dalton (1803) formulated the Atomic Theory:
- All matter is made of tiny indivisible particles called atoms.
- Atoms of the same element are identical in mass and properties.
- Atoms of different elements have different masses and properties.
- Atoms combine in simple, whole-number ratios to form compounds.
- Chemical reactions involve rearrangement of atoms, not their creation or destruction.
Dalton’s theory helped explain laws of chemical combination, including:
- Law of Conservation of Mass
- Law of Definite Proportions
- Law of Multiple Proportions
Molecules and Compounds
A molecule is formed when two or more atoms chemically combine. For example, a water molecule (H₂O) consists of two hydrogen atoms and one oxygen atom. Molecules can be elementary (like O₂, N₂) or compound molecules (like CO₂, H₂O).
A compound is a substance made from two or more different elements in a definite ratio. Compounds have fixed chemical formulas, for example:
- NaCl → Sodium chloride
- H₂O → Water
- CO₂ → Carbon dioxide
Chemical Formulas
Chemical formulas represent the composition of a molecule or compound. There are different types:
- Empirical Formula: Simplest ratio of atoms (e.g., CH₂O for glucose).
- Molecular Formula: Actual number of atoms in a molecule (e.g., C₆H₁₂O₆ for glucose).
- Structural Formula: Shows how atoms are bonded (e.g., H–O–H for water).
Atomic and Molecular Mass
- Atomic Mass: Weighted average mass of an atom compared to 1/12th of carbon-12.
- Molecular Mass: Sum of atomic masses of atoms in a molecule.
- Mole Concept: One mole contains 6.022 × 10²³ particles (Avogadro’s number).
Laws of Chemical Combination
- Law of Conservation of Mass: Antoine Lavoisier proved that total mass of reactants equals total mass of products.
- Law of Definite Proportions: Joseph Proust showed that elements combine in a fixed ratio by mass.
- Law of Multiple Proportions: Dalton observed that if elements combine in more than one ratio, the masses are multiples of each other.
Importance of Atoms and Molecules
Understanding atoms and molecules is crucial for:
- Predicting the outcome of chemical reactions.
- Calculating molecular masses and chemical formulas.
- Studying physical and chemical properties of matter.
- Applying in real-world situations like pharmaceuticals, material science, and nanotechnology.
This chapter also emphasizes problem-solving in topics like:
- Calculating molecular mass
- Determining the ratio of elements in compounds
- Using moles in chemical reactions
Modern Atomic Theory
- Electron Cloud Model: Electrons move around the nucleus in regions called orbitals.
- Protons and Neutrons: Nucleus contains protons (+ charge) and neutrons (neutral).
- Electrons: Negatively charged particles in orbitals determine chemical behavior.
Flowchart / Mind Map (Text-Based)
Atoms and Molecules
│
├─ Atoms
│ ├─ Subatomic Particles
│ │ ├─ Proton
│ │ ├─ Neutron
│ │ └─ Electron
│ ├─ Atomic Mass
│ └─ Dalton’s Theory
│
├─ Molecules
│ ├─ Elementary Molecules (O₂, N₂)
│ └─ Compound Molecules (H₂O, CO₂)
│
├─ Laws of Chemical Combination
│ ├─ Law of Conservation of Mass
│ ├─ Law of Definite Proportions
│ └─ Law of Multiple Proportions
│
├─ Chemical Formulas
│ ├─ Empirical Formula
│ ├─ Molecular Formula
│ └─ Structural Formula
│
└─ Applications
├─ Stoichiometry
├─ Material Science
└─ Pharmaceuticals
Important Keywords with Meanings
| Keyword | Meaning |
|---|---|
| Atom | Smallest unit of an element |
| Molecule | Combination of two or more atoms |
| Compound | Substance of two or more elements in fixed ratio |
| Empirical Formula | Simplest ratio of atoms in a compound |
| Molecular Mass | Total mass of atoms in a molecule |
| Subatomic Particles | Proton, Neutron, Electron |
| Avogadro’s Number | 6.022 × 10²³ particles per mole |
| Chemical Reaction | Rearrangement of atoms to form new substance |
| Law of Conservation | Mass is constant in chemical reactions |
| Law of Definite Proportions | Element ratio in a compound is fixed |
Important Questions & Answers
Very Short Answer Questions (1–3 Marks)
- Define atom.
Answer: The smallest particle of an element that retains its chemical properties. - What is a molecule?
Answer: A combination of two or more atoms bonded together. - State the law of conservation of mass.
Answer: Mass is neither created nor destroyed in a chemical reaction. - Give an example of a compound molecule.
Answer: Water (H₂O) - What is an empirical formula?
Answer: Simplest ratio of atoms in a compound. - What is atomic mass?
Answer: Weighted average mass of an atom compared to 1/12th of carbon-12. - Name the subatomic particles.
Answer: Proton, Neutron, Electron - What is molecular mass?
Answer: Sum of atomic masses of atoms in a molecule. - Give an example of a law explained by Dalton.
Answer: Law of Multiple Proportions - Define a chemical reaction.
Answer: Rearrangement of atoms to form new substances.
Short Answer Questions (5 Marks)
- Explain Dalton’s Atomic Theory with examples.
Answer: Dalton’s Atomic Theory states that all matter is made of atoms, atoms of same element are identical, different elements have different atoms, and atoms combine in simple ratios to form compounds. Example: H₂O → 2H + 1O. - Explain the difference between atom and molecule.
Answer: Atom is the smallest particle of an element, while a molecule is a combination of two or more atoms chemically bonded. Example: O (atom), O₂ (molecule). - Describe the law of definite proportions with example.
Answer: A compound always contains elements in a fixed ratio. Example: Water always has 2H:1O by atoms or 11.1% H and 88.9% O by mass. - Write a note on subatomic particles.
Answer: Atoms contain protons (+1), neutrons (0), and electrons (−1). Protons and neutrons form the nucleus, electrons orbit in shells. - Explain molecular and empirical formula difference with example.
Answer: Empirical formula shows simplest ratio (CH₂O), molecular formula shows actual number of atoms (C₆H₁₂O₆). - Describe the law of conservation of mass.
Answer: Mass of reactants = Mass of products. Example: H₂ + O₂ → H₂O. Mass of H₂ and O₂ = Mass of H₂O. - Define and give example of compound molecule.
Answer: Molecule made of different elements. Example: CO₂, H₂O. - Explain importance of atoms and molecules in daily life.
Answer: They explain chemical reactions, formation of compounds, material properties, and pharmaceuticals. - Describe Avogadro’s number and mole concept.
Answer: One mole = 6.022 × 10²³ particles. Used to count atoms and molecules in reactions. - Explain types of chemical formulas.
Answer: Empirical (simplest ratio), Molecular (actual number of atoms), Structural (bonding arrangement).
Multiple Choice Questions (MCQs)
- Smallest particle of an element retaining chemical properties is:
a) Molecule
b) Atom ✅
c) Compound
d) Electron - Water molecule contains:
a) 1 H and 2 O
b) 2 H and 1 O ✅
c) 2 O and 1 H
d) 1 O and 1 H - Law stating mass remains constant in reactions:
a) Law of Multiple Proportions
b) Law of Definite Proportions
c) Law of Conservation of Mass ✅
d) Law of Chemical Combination
Exam Tips & Value-Based Questions
- Tip 1: Always write chemical formulas correctly.
- Tip 2: Use atomic and molecular masses in calculations carefully.
- Tip 3: Revise Dalton’s atomic theory thoroughly.
- Tip 4: Remember examples for laws of chemical combination.
- Tip 5: Solve NCERT exemplar questions for practice.
Value-Based Questions:
- Why is understanding atoms important for environmental protection?
Answer: It helps in pollution control and chemical waste management. - How can molecules improve healthcare?
Answer: Molecules form drugs and medicines. - Importance of accurate measurement in chemical reactions?
Answer: Prevents wastage, ensures safety. - Role of atoms in material science?
Answer: Understanding atomic structure aids in designing stronger materials. - How does knowledge of molecules promote sustainable chemistry?
Answer: Helps in developing eco-friendly compounds and reactions.
Conclusion 💫
The chapter Atoms and Molecules is a cornerstone of Chemistry, forming the basis of chemical knowledge and problem-solving skills. Understanding atoms — the indivisible units of elements — and molecules — combinations of atoms — enables students to explain physical and chemical changes in everyday life. From Dalton’s atomic theory to modern models, the study of atoms and molecules integrates historical discoveries, laws of chemical combination, and practical applications in daily life.
Mastery of this chapter helps students in exam preparation, competitive exams, and building a strong foundation for higher studies in Chemistry. It also encourages analytical thinking, observation, and quantitative reasoning. Using short notes, flowcharts, MCQs, and example problems ensures students can grasp concepts effectively and retain information for exams.
Whether it is calculating molecular masses, understanding chemical formulas, or explaining conservation laws, the chapter emphasizes clarity and precision. Furthermore, knowledge of atoms and molecules is crucial for innovations in pharmaceuticals, material science, nanotechnology, and environmental chemistry.
By studying Atoms and Molecules, students not only achieve academic success but also develop scientific reasoning, problem-solving skills, and an understanding of matter at a microscopic level. Regular practice, revision of important questions, and familiarity with keywords ensure long-term retention and better performance in both school exams and competitive tests.
In conclusion, Atoms and Molecules is not just a chapter; it is a gateway to understanding the microscopic world that governs all chemical and physical processes. Mastery of this topic will pave the way for excellence in Chemistry and related sciences.
Long Question Answer ✨
1. Explain Dalton’s Atomic Theory with examples.
Answer:
Dalton’s Atomic Theory (1803) is the foundation of modern chemistry. Its main points are:
- Matter is made of indivisible atoms.
- Atoms of the same element are identical in mass and properties.
- Atoms of different elements have different masses and properties.
- Atoms combine in simple, whole-number ratios to form compounds.
- In chemical reactions, atoms are rearranged but not created or destroyed.
Example: Water (H₂O) is formed when two hydrogen atoms combine with one oxygen atom.
2. Distinguish between atom and molecule with examples.
Answer:
| Feature | Atom | Molecule |
|---|---|---|
| Definition | Smallest particle of an element | Combination of two or more atoms |
| Chemical property | Retains properties of element | May show new properties |
| Example | O, H, Na | H₂, O₂, H₂O |
3. Describe the law of conservation of mass with a suitable example.
Answer:
The Law of Conservation of Mass states that in a chemical reaction, mass is neither created nor destroyed.
Example:
2H₂ + O₂ → 2H₂O
- Mass of reactants = Mass of products
- Mass of H₂ (4 g) + Mass of O₂ (32 g) = Mass of H₂O (36 g)
This law was proven by Antoine Lavoisier.
4. Explain the law of definite proportions with example.
Answer:
The Law of Definite Proportions states that a chemical compound always contains the same elements in a fixed proportion by mass.
Example: Water (H₂O) always contains:
- Hydrogen = 11.1% by mass
- Oxygen = 88.9% by mass
No matter how water is obtained, the ratio remains the same.
5. State and explain the law of multiple proportions.
Answer:
The Law of Multiple Proportions (Dalton) states:
If two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in small whole-number ratios.
Example: Carbon and oxygen form:
- CO → 12 g C combines with 16 g O
- CO₂ → 12 g C combines with 32 g O
- Ratio of O in CO₂ : CO = 32 : 16 = 2 : 1
6. Define molecule. Explain types of molecules with examples.
Answer:
A molecule is a combination of two or more atoms chemically bonded together.
Types of Molecules:
- Elementary Molecules – made of the same type of atoms (O₂, N₂, H₂).
- Compound Molecules – made of different types of atoms (H₂O, CO₂, NaCl).
7. Explain atomic mass and molecular mass with examples.
Answer:
- Atomic Mass: Relative mass of an atom compared to 1/12th of carbon-12.
Example: Hydrogen = 1, Oxygen = 16 - Molecular Mass: Sum of atomic masses in a molecule.
Example: H₂O = 2(1) + 16 = 18
8. Describe empirical formula, molecular formula, and structural formula.
Answer:
- Empirical Formula: Simplest ratio of atoms in a compound (CH₂O for glucose).
- Molecular Formula: Actual number of atoms (C₆H₁₂O₆ for glucose).
- Structural Formula: Shows how atoms are bonded (H–O–H for water).
9. Explain the subatomic particles of an atom.
Answer:
Atoms are made of subatomic particles:
- Proton (p⁺): Positive charge, mass ≈ 1 amu, found in nucleus.
- Neutron (n⁰): Neutral, mass ≈ 1 amu, in nucleus.
- Electron (e⁻): Negative charge, negligible mass, orbits nucleus.
Example: Oxygen atom → 8 protons, 8 neutrons, 8 electrons.
10. What is Avogadro’s number? Explain the mole concept.
Answer:
- Avogadro’s Number: 6.022 × 10²³ particles per mole.
- Mole Concept: One mole of a substance contains Avogadro’s number of particles.
Example: 1 mole of H₂O = 6.022 × 10²³ molecules = 18 g H₂O
11. Explain the importance of atoms and molecules in daily life.
Answer:
Atoms and molecules help explain:
- Composition of matter (air, water, food).
- Chemical reactions (digestion, combustion, rusting).
- Pharmaceutical drugs (molecular structure determines effectiveness).
- Materials (plastic, metal alloys).
- Environmental chemistry (pollution, recycling).
Understanding atoms and molecules builds a foundation for advanced chemistry and real-world applications.
12. Calculate molecular mass and write empirical formula:
Problem: A compound contains 40% carbon, 6.7% hydrogen, 53.3% oxygen. Molecular mass = 180 g/mol.
Answer:
- Convert % to grams (assume 100 g sample):
- C = 40 g, H = 6.7 g, O = 53.3 g
- Convert grams to moles:
- C = 40 ÷ 12 = 3.33 mol
- H = 6.7 ÷ 1 = 6.7 mol
- O = 53.3 ÷ 16 = 3.33 mol
- Simplest ratio (divide by smallest = 3.33):
- C : H : O = 1 : 2 : 1 → Empirical formula = CH₂O
- Molecular formula = (Molecular mass ÷ Empirical mass) × Empirical formula
- Empirical mass = 12 + 2 + 16 = 30
- Molecular mass = 180 → 180 ÷ 30 = 6
- Molecular formula = C₆H₁₂O₆
Assertion Reason 💫
Instructions:
- Assertion (A): A statement that may be true or false.
- Reason (R): A statement that explains the assertion.
- Choose the correct option:
- Both A and R are true, and R is the correct explanation of A
- Both A and R are true, but R is not the correct explanation of A
- A is true, R is false
- A is false, R is true
Questions
1.
- A: Water is always composed of 2 hydrogen atoms and 1 oxygen atom.
- R: Law of Definite Proportions states that elements combine in fixed mass ratios.
Answer: 1 (Both A and R are true, and R explains A)
2.
- A: Mass is neither created nor destroyed in a chemical reaction.
- R: Atoms are rearranged during a chemical reaction without any change in mass.
Answer: 1 (Both A and R are true, R explains A)
3.
- A: A molecule of oxygen contains 2 oxygen atoms.
- R: Oxygen is a compound.
Answer: 3 (A is true, R is false; oxygen is an element)
4.
- A: Dalton’s Atomic Theory states that atoms are indivisible.
- R: Atoms are made of protons, neutrons, and electrons.
Answer: 3 (A is true according to Dalton, R is false in Dalton’s context)
5.
- A: CO and CO₂ are two different compounds of carbon and oxygen.
- R: The masses of oxygen combining with fixed carbon in CO and CO₂ are in small whole numbers.
Answer: 1 (Both A and R are true, R explains A)
6.
- A: The molecular mass of H₂O is 18 u.
- R: Molecular mass is the sum of atomic masses of atoms in a molecule.
Answer: 1 (Both A and R are true, R explains A)
7.
- A: Hydrogen and oxygen form water in 2:1 atomic ratio.
- R: Law of Multiple Proportions explains combination of elements in multiple ratios.
Answer: 2 (Both A and R are true, but R does not explain A)
8.
- A: Atomic mass of hydrogen is 1 u.
- R: Atomic mass is measured relative to carbon-12.
Answer: 1 (Both A and R are true, R explains A)
9.
- A: H₂O has the same composition irrespective of the source.
- R: Compounds are made of elements in a definite ratio.
Answer: 1 (Both A and R are true, R explains A)
10.
- A: Molecules can be elementary or compound.
- R: Elementary molecules consist of different types of atoms.
Answer: 3 (A is true, R is false; elementary molecules consist of same type of atoms)
11.
- A: One mole of any substance contains 6.022 × 10²³ particles.
- R: Avogadro’s number is used to count large numbers of atoms and molecules.
Answer: 1 (Both A and R are true, R explains A)
12.
- A: Empirical formula of glucose is CH₂O.
- R: Molecular formula shows the simplest ratio of atoms.
Answer: 3 (A is true, R is false; molecular formula shows actual number of atoms)
13.
- A: Protons are positively charged subatomic particles.
- R: Neutrons carry no charge and have same mass as protons.
Answer: 2 (Both A and R are true, but R does not explain A)
14.
- A: Chemical reactions involve rearrangement of atoms.
- R: The total number of atoms remains unchanged before and after reaction.
Answer: 1 (Both A and R are true, R explains A)
15.
- A: Molecular mass of CO₂ = 44 u.
- R: CO₂ contains 1 carbon atom and 2 oxygen atoms, with atomic masses 12 and 16 u respectively.
Answer: 1 (Both A and R are true, R explains A)
10 Case-Based Questions – Atoms and Molecules
Case 1: Law of Conservation of Mass
Case:
A student mixes 4 g of hydrogen with 32 g of oxygen to form water. He measures the mass of water produced and finds it to be 36 g.
Questions:
- Which law does this experiment verify?
- Calculate the mass of water formed.
Answer:
- Law of Conservation of Mass – Mass of reactants = Mass of products.
- Mass of water = Mass of H + Mass of O = 4 g + 32 g = 36 g.
Case 2: Empirical and Molecular Formula
Case:
A compound contains 40% carbon, 6.7% hydrogen, and 53.3% oxygen. Its molecular mass is 180 g/mol.
Questions:
- Determine the empirical formula.
- Find the molecular formula.
Answer:
- Convert % to grams → C:40 g, H:6.7 g, O:53.3 g
- Moles: C = 3.33, H = 6.7, O = 3.33 → Ratio = 1:2:1 → CH₂O
- Empirical mass = 12+2+16=30, Molecular mass = 180 → 180 ÷ 30 = 6 → C₆H₁₂O₆
Case 3: Law of Definite Proportions
Case:
Water from a river and water from a lab are analyzed. Both contain 11.1% hydrogen and 88.9% oxygen by mass.
Questions:
- Which law is illustrated here?
- Explain why the composition is identical.
Answer:
- Law of Definite Proportions
- Elements combine in a fixed ratio by mass to form compounds; hence, water always has same ratio of H and O.
Case 4: Mole Concept
Case:
1 mole of NaCl contains 6.022 × 10²³ formula units.
Questions:
- How many moles are present in 1.204 × 10²⁴ formula units?
- Explain the significance of Avogadro’s number.
Answer:
- Moles = 1.204 × 10²⁴ ÷ 6.022 × 10²³ = 2 moles
- Avogadro’s number helps count atoms, molecules, or ions in a substance.
Case 5: Molecular Mass
Case:
A student calculates the molecular mass of CO₂. Atomic masses: C = 12, O = 16.
Questions:
- Find molecular mass of CO₂.
- How does molecular mass differ from atomic mass?
Answer:
- Molecular mass = 12 + 2×16 = 44 u
- Atomic mass = mass of single atom, Molecular mass = sum of atomic masses in molecule.
Case 6: Law of Multiple Proportions
Case:
Carbon and oxygen form CO and CO₂. 12 g of C combines with 16 g O → CO, 12 g C combines with 32 g O → CO₂.
Questions:
- Find the ratio of oxygen in CO₂ to CO.
- Which law is illustrated?
Answer:
- Ratio = 32 : 16 = 2:1
- Law of Multiple Proportions – Masses of one element combine in small whole numbers with fixed mass of another.
Case 7: Subatomic Particles
Case:
An atom has 11 protons, 12 neutrons, and 11 electrons.
Questions:
- Identify the element.
- Calculate its mass number.
Answer:
- Atomic number = Number of protons = 11 → Sodium (Na)
- Mass number = Protons + Neutrons = 11 + 12 = 23
Case 8: Molecules of Elements
Case:
Oxygen gas contains O₂ molecules, while hydrogen gas contains H₂ molecules.
Questions:
- Define elementary molecules.
- Give one example of a compound molecule.
Answer:
- Elementary molecules are made of the same type of atoms (O₂, H₂)
- Compound molecule example → H₂O
Case 9: Empirical Formula Problem
Case:
A compound contains 20 g nitrogen and 80 g oxygen.
Questions:
- Find the empirical formula.
- Explain the steps.
Answer:
- Moles: N = 20 ÷ 14 ≈ 1.43, O = 80 ÷ 16 = 5 → ratio ≈ 1:3.5 → multiply by 2 → 2:7 → N₂O₇
- Steps: Convert mass → moles → ratio → simplest whole number formula.
Case 10: Chemical Reaction Analysis
Case:
2 moles of H₂ react with 1 mole of O₂ to form water.
Questions:
- Write the balanced chemical equation.
- Verify if it obeys the law of conservation of mass.
Answer:
- 2H₂ + O₂ → 2H₂O
- Mass of reactants = Mass of products → 4 g H + 32 g O = 36 g H₂O ✅
These 10 case-based questions cover:
- Laws of chemical combination
- Mole concept & Avogadro’s number
- Molecular and empirical formulas
- Subatomic particles
- Chemical reactions
Multiple choice questions
50 MCQs – Atoms and Molecules
1–10: Basic Concepts
- Smallest particle of an element retaining its chemical properties is:
a) Molecule
b) Atom ✅
c) Compound
d) Electron - A molecule is:
a) Single atom of an element
b) Two or more atoms chemically bonded ✅
c) Always made of different elements
d) Negatively charged particle - Which of the following is an elementary molecule?
a) H₂ ✅
b) H₂O
c) CO₂
d) NaCl - CO₂ is a:
a) Element
b) Compound ✅
c) Mixture
d) Molecule - The atomic mass of an element is measured relative to:
a) Hydrogen-1
b) Oxygen-16
c) Carbon-12 ✅
d) Nitrogen-14 - Which particle has negligible mass?
a) Proton
b) Neutron
c) Electron ✅
d) Nucleus - The nucleus of an atom contains:
a) Only electrons
b) Protons and neutrons ✅
c) Electrons and neutrons
d) Only protons - The chemical formula H₂O represents:
a) 1 hydrogen + 2 oxygen atoms
b) 2 hydrogen + 1 oxygen atom ✅
c) 2 hydrogen + 2 oxygen atoms
d) 1 hydrogen + 1 oxygen atom - Atomic number of an element is determined by:
a) Number of electrons
b) Number of protons ✅
c) Number of neutrons
d) Mass number - Mass number of an atom =
a) Protons + Neutrons ✅
b) Protons – Neutrons
c) Electrons + Neutrons
d) Electrons × Protons
11–20: Laws of Chemical Combination
- Law of Conservation of Mass was proposed by:
a) Dalton
b) Lavoisier ✅
c) Proust
d) Rutherford - Law of Definite Proportions is also known as:
a) Proust’s Law ✅
b) Dalton’s Law
c) Avogadro’s Law
d) Lavoisier’s Law - Law of Multiple Proportions was given by:
a) Lavoisier
b) Dalton ✅
c) Proust
d) Rutherford - In chemical reactions:
a) Atoms are created
b) Atoms are destroyed
c) Atoms are rearranged ✅
d) Mass decreases - Which law is illustrated by water from river and water from lab having same composition?
a) Law of Multiple Proportions
b) Law of Definite Proportions ✅
c) Law of Conservation of Mass
d) Dalton’s Law - A compound always contains:
a) Elements in variable proportion
b) Elements in fixed proportion ✅
c) Single element only
d) No elements - In CO and CO₂, ratio of oxygen masses combining with 12 g of carbon is:
a) 1:1
b) 1:2 ✅
c) 2:1
d) 1:3 - Mass of reactants = Mass of products refers to:
a) Law of Definite Proportions
b) Law of Conservation of Mass ✅
c) Law of Multiple Proportions
d) Dalton’s Law - Which scientist introduced the concept of atoms?
a) Lavoisier
b) Dalton ✅
c) Thomson
d) Rutherford - Atoms of the same element are:
a) Different in mass
b) Identical in mass and properties ✅
c) Negatively charged
d) Always reactive
21–30: Molecules & Compounds
- Which of the following is a compound molecule?
a) O₂
b) H₂O ✅
c) N₂
d) Cl₂ - Elementary molecules contain:
a) Same type of atoms ✅
b) Different atoms
c) Only neutrons
d) Electrons only - Which of the following is NOT a molecule?
a) H₂ ✅
b) O₂
c) H₂O
d) CO₂ - Empirical formula shows:
a) Actual number of atoms
b) Simplest ratio of atoms ✅
c) Structural bonds
d) Mass number - Molecular formula shows:
a) Actual number of atoms ✅
b) Simplest ratio
c) Atomic number
d) Mass ratio - Structural formula shows:
a) Atomic masses
b) Bonding arrangement ✅
c) Empirical formula
d) Molecular mass - NaCl is represented by:
a) Molecular formula
b) Formula unit ✅
c) Empirical formula
d) Structural formula - H₂O contains:
a) 1 H + 2 O
b) 2 H + 1 O ✅
c) 2 H + 2 O
d) 1 H + 1 O - Molecules of elements:
a) Always compounds
b) Can be elementary ✅
c) Always ions
d) All radioactive - Which is a diatomic molecule?
a) H₂ ✅
b) H₂O
c) CO₂
d) CH₄
31–40: Atomic Mass, Molecular Mass, & Mole Concept
- Atomic mass of hydrogen is:
a) 1 u ✅
b) 2 u
c) 12 u
d) 16 u - Molecular mass of H₂O =
a) 16 u
b) 18 u ✅
c) 20 u
d) 10 u - Molecular mass of CO₂ =
a) 28 u
b) 44 u ✅
c) 32 u
d) 12 u - 1 mole contains:
a) 6.022 × 10²³ particles ✅
b) 10²³ particles
c) 6.022 × 10²⁴ particles
d) 1 g of substance - Number of moles =
a) Mass ÷ Atomic mass ✅
b) Mass × Atomic mass
c) Atomic mass ÷ Mass
d) Mass + Atomic mass - Mass of 2 moles of H₂O =
a) 18 g
b) 36 g ✅
c) 9 g
d) 24 g - Avogadro’s number is used to:
a) Count molecules and atoms ✅
b) Measure mass
c) Determine density
d) Measure volume - Atomic number of sodium = 11, number of neutrons = 12, mass number = ?
a) 11
b) 12
c) 23 ✅
d) 24 - Mass of 1 mole of O₂ =
a) 16 g
b) 32 g ✅
c) 64 g
d) 8 g - Moles of 18 g H₂O =
a) 1 ✅
b) 2
c) 0.5
d) 3
41–50: Applied & Conceptual MCQs
- Which is true for a chemical reaction?
a) Atoms are destroyed
b) Atoms are created
c) Atoms are rearranged ✅
d) Mass decreases - 12 g of carbon combines with 32 g of oxygen → compound. Mass of compound = ?
a) 32 g
b) 44 g ✅
c) 12 g
d) 24 g - Water always contains hydrogen and oxygen in ratio:
a) 1:1
b) 2:1 atoms ✅
c) 3:1
d) 1:2 - A compound has 2:1 atomic ratio → type of formula?
a) Empirical ✅
b) Molecular
c) Structural
d) Formula unit - Subatomic particle with +1 charge is:
a) Electron
b) Proton ✅
c) Neutron
d) Positron - Neutron carries:
a) Positive charge
b) Negative charge
c) No charge ✅
d) Variable charge - Molecular mass of CH₄ =
a) 12 u
b) 16 u ✅
c) 14 u
d) 18 u - Which is NOT an example of law of multiple proportions?
a) CO and CO₂ ✅
b) NO and NO₂
c) H₂O
d) CCl₄ and CCl₂ - Number of electrons in neutral Na atom =
a) 10
b) 11 ✅
c) 12
d) 23 - The law explaining fixed mass ratio of elements in a compound is:
a) Law of Conservation of Mass
b) Law of Multiple Proportions
c) Law of Definite Proportions ✅
d) Dalton’s Atomic Theory
✅ Answer Key:
1-b, 2-b, 3-a, 4-b, 5-c, 6-c, 7-b, 8-b, 9-b, 10-a, 11-b, 12-a, 13-b, 14-c, 15-b, 16-b, 17-b, 18-b, 19-b, 20-b, 21-b, 22-a, 23-a, 24-b, 25-a, 26-b, 27-b, 28-b, 29-b, 30-a, 31-a, 32-b, 33-b, 34-a, 35-a, 36-b, 37-a, 38-c, 39-b, 40-a, 41-c, 42-b, 43-b, 44-a, 45-b, 46-c, 47-b, 48-c, 49-b, 50-c
Atoms and Molecules – Class 9 Chemistry
Maximum Marks: 50
Time: 2 Hours
Section A: Very Short Answer Questions (1 mark each) – 5 Questions
(Answer in one line)
- Define an atom.
- Define a molecule.
- State the Law of Conservation of Mass.
- What is an empirical formula?
- Name the subatomic particles of an atom.
Marks: 5 × 1 = 5
Section B: Short Answer Questions (2 marks each) – 10 Questions
(Answer in 1–2 lines)
- Give an example of a compound molecule and an elementary molecule.
- Calculate the mass of 2 moles of H₂O.
- State the Law of Definite Proportions with an example.
- Atomic number of oxygen = 8. Number of neutrons = 8. Find mass number.
- Distinguish between molecular and empirical formula.
- Which law is illustrated by CO and CO₂? Explain briefly.
- Define atomic mass.
- Calculate the number of moles in 44 g of CO₂.
- Define molecular mass.
- Write the structural formula of water.
Marks: 10 × 2 = 20
Section C: Long Answer Questions (5 marks each) – 5 Questions
(Answer in 4–5 lines or with calculations)
- Explain Dalton’s Atomic Theory with examples.
- A compound contains 40% carbon, 6.7% hydrogen, and 53.3% oxygen. Its molecular mass is 180 g/mol. Find:
a) Empirical formula
b) Molecular formula - Explain the Law of Multiple Proportions with an example.
- Calculate the molecular mass of CO₂.
- An atom has 11 protons, 12 neutrons, and 11 electrons. Identify the element and its mass number.
Marks: 5 × 5 = 25
Section D: Multiple Choice Questions (1 mark each) – 5 Questions
- Which is the smallest particle of an element retaining chemical properties?
a) Molecule
b) Atom ✅
c) Compound
d) Electron - Molecular mass of H₂O = ?
a) 16 u
b) 18 u ✅
c) 20 u
d) 10 u - The number of atoms in 1 mole = ?
a) 6.022 × 10²³ ✅
b) 10²³
c) 6.022 × 10²⁴
d) 1 g - Which of the following is an elementary molecule?
a) H₂ ✅
b) H₂O
c) CO₂
d) NaCl - Law of Conservation of Mass was proposed by:
a) Dalton
b) Lavoisier ✅
c) Proust
d) Rutherford
Marks: 5 × 1 = 5
Total Marks = 50
Here’s the complete solution to the 50-mark test paper for the chapter “Atoms and Molecules” (Class 9 Chemistry, NCERT). I’ve included stepwise answers, explanations, and calculations where needed.
Atoms and Molecules – Test Paper Solutions
Section A: Very Short Answer Questions (1 mark each)
- Atom: The smallest particle of an element that retains its chemical properties.
- Molecule: A combination of two or more atoms chemically bonded together.
- Law of Conservation of Mass: Mass is neither created nor destroyed in a chemical reaction; it only gets rearranged.
- Empirical Formula: The simplest ratio of atoms of different elements in a compound.
- Subatomic Particles: Proton (+), Neutron (0), Electron (−)
Section B: Short Answer Questions (2 marks each)
- Examples of molecules:
- Compound molecule: H₂O (water)
- Elementary molecule: O₂ (oxygen)
- Mass of 2 moles of H₂O:
- Molecular mass of H₂O = 2(1) + 16 = 18 g/mol
- Mass of 2 moles = 2 × 18 = 36 g
- Law of Definite Proportions:
- A chemical compound always contains elements in fixed mass proportion.
- Example: Water always has 2 H atoms and 1 O atom → 11.1% H, 88.9% O
- Mass number of oxygen atom:
- Mass number = Protons + Neutrons = 8 + 8 = 16
- Molecular vs Empirical Formula:
- Molecular formula = actual number of atoms in a molecule (C₆H₁₂O₆)
- Empirical formula = simplest whole-number ratio of atoms (CH₂O)
- Law Illustrated by CO and CO₂:
- Law of Multiple Proportions:
- Mass of O combining with 12 g C: CO →16 g O, CO₂ → 32 g O
- Ratio = 16:32 = 1:2
- Atomic Mass: Weighted average mass of an atom of an element compared to 1/12th of carbon-12.
- Number of moles in 44 g CO₂:
- Molecular mass of CO₂ = 12 + 16×2 = 44 g/mol
- Moles = Mass ÷ Molecular mass = 44 ÷ 44 = 1 mole
- Molecular Mass: Sum of atomic masses of all atoms in a molecule.
- Example: H₂O → 2(1) + 16 = 18 u
- Structural formula of water: H–O–H
Section C: Long Answer Questions (5 marks each)
- Dalton’s Atomic Theory:
- Matter is made of indivisible atoms.
- Atoms of same element are identical in mass and properties; atoms of different elements differ.
- Atoms combine in simple whole-number ratios to form compounds.
- In reactions, atoms are rearranged, not created/destroyed.
- Example: H₂ + O → H₂O (2 H + 1 O)
- Empirical & Molecular Formula Problem:
- % composition: C = 40%, H = 6.7%, O = 53.3%
- Assume 100 g → C=40 g, H=6.7 g, O=53.3 g
- Moles: C = 40 ÷ 12 ≈ 3.33, H = 6.7 ÷ 1 ≈ 6.7, O = 53.3 ÷16 ≈ 3.33
- Ratio C:H:O = 3.33:6.7:3.33 ≈ 1:2:1 → Empirical formula = CH₂O
- Molecular mass = 180 g/mol, Empirical mass = 12 + 2 + 16 = 30 g/mol
- Molecular formula = (180 ÷ 30) × CH₂O = 6 × CH₂O = C₆H₁₂O₆
- Law of Multiple Proportions:
- When two elements form more than one compound, the ratio of masses of one element that combines with fixed mass of the other is a small whole number.
- Example: C + O → CO (12 g C + 16 g O), CO₂ (12 g C + 32 g O), ratio 16:32 = 1:2
- Molecular mass of CO₂:
- C = 12, O = 16
- Molecular mass = 12 + 16×2 = 44 u
- Atom with 11 protons, 12 neutrons, 11 electrons:
- Atomic number = 11 → Sodium (Na)
- Mass number = Protons + Neutrons = 11 + 12 = 23
Section D: Multiple Choice Questions (1 mark each)
- Smallest particle of an element retaining chemical properties → Atom ✅
- Molecular mass of H₂O → 18 u ✅
- Number of atoms in 1 mole → 6.022 × 10²³ ✅
- Elementary molecule → H₂ ✅
- Law of Conservation of Mass proposed by → Lavoisier ✅
✅ Marking Scheme
- Section A: 5 × 1 = 5
- Section B: 10 × 2 = 20
- Section C: 5 × 5 = 25
- Section D: 5 × 1 = 5
Total = 50 Marks
Atoms and Molecules – Quick Revision (Class 9 Chemistry)
Atoms and Molecules is a fundamental chapter in chemistry that explains the structure of matter, subatomic particles, chemical formulas, and laws of chemical combination. This guide provides a quick revision for exams.
1. Basic Concepts
- Atom: Smallest particle of an element that retains its chemical properties.
- Molecule: Combination of two or more atoms chemically bonded together.
- Elementary Molecule: Made of the same type of atoms (e.g., H₂, O₂, N₂).
- Compound Molecule: Made of different types of atoms (e.g., H₂O, CO₂).
- Subatomic Particles:
- Proton (p⁺): +1 charge, mass ≈ 1 u, in nucleus
- Neutron (n⁰): Neutral, mass ≈ 1 u, in nucleus
- Electron (e⁻): −1 charge, negligible mass, orbits nucleus
- Atomic Number (Z): Number of protons in the nucleus.
- Mass Number (A): Number of protons + neutrons.
2. Laws of Chemical Combination
- Law of Conservation of Mass (Lavoisier): Mass is neither created nor destroyed in chemical reactions.
- Law of Definite Proportions (Proust): A chemical compound always contains elements in fixed mass ratios.
- Example: Water always contains 11.1% H and 88.9% O by mass.
- Law of Multiple Proportions (Dalton): When two elements form more than one compound, the masses of one element combining with a fixed mass of the other are in small whole-number ratios.
- Example: C + O → CO, CO₂ → ratio of O = 16:32 = 1:2
3. Formulas
- Empirical Formula: Simplest ratio of atoms in a compound (CH₂O for glucose).
- Molecular Formula: Actual number of atoms in a molecule (C₆H₁₂O₆ for glucose).
- Structural Formula: Shows bonding arrangement of atoms (H–O–H for water).
- Formula Unit: Smallest representative unit of ionic compounds (NaCl).
4. Atomic and Molecular Mass
- Atomic Mass: Average mass of an atom compared to 1/12th of carbon-12.
- Molecular Mass: Sum of atomic masses of all atoms in a molecule.
- Example: H₂O = 2 × 1 + 16 = 18 u
- CO₂ = 12 + 16 × 2 = 44 u
5. Mole Concept
- Avogadro’s Number: 6.022 × 10²³ particles/molecules in 1 mole.
- Mole Formula:
- Number of moles = Mass ÷ Molar mass
- Mass of substance = Number of moles × Molar mass
- Example: 2 moles of H₂O → Mass = 2 × 18 = 36 g
6. Important Chemical Reactions
- Atoms are rearranged in chemical reactions; mass remains constant.
- Example: 2H₂ + O₂ → 2H₂O
- Water formation shows Law of Definite Proportions: H:O = 2:1 (atoms), 11.1:88.9 (mass).
- Carbon and oxygen forming CO and CO₂ illustrate Law of Multiple Proportions.
7. Quick Numerical Revision
- Empirical Formula Calculation:
- % composition → grams → moles → simplest ratio → formula.
- Molecular Formula:
- Molecular mass ÷ Empirical mass × Empirical formula.
- Moles & Mass:
- Moles = Mass ÷ Molar mass
- Mass = Moles × Molar mass
8. Key Points for Exams
- Memorize atomic numbers, mass numbers, and subatomic particle properties.
- Remember laws of chemical combination with examples.
- Practice empirical & molecular formula problems.
- Always check mass balance in reactions.
- Revise mole concept for numerical questions.
9. Quick Flowchart
Matter
├─ Atoms
│ ├─ Subatomic particles (p⁺, n⁰, e⁻)
│ └─ Properties (Z, A)
└─ Molecules
├─ Elementary (H₂, O₂)
└─ Compound (H₂O, CO₂)
Laws of Combination
├─ Conservation of Mass
├─ Definite Proportions
└─ Multiple Proportions
Formulas
├─ Empirical (CH₂O)
├─ Molecular (C₆H₁₂O₆)
└─ Structural (H–O–H)
Mole Concept
├─ Avogadro’s Number (6.022×10²³)
└─ Mass ↔ Moles ↔ Particles
This quick revision covers:
- Atoms, molecules, formulas, laws, atomic/molecular mass, mole concept, and reactions.
- Perfect for last-minute exam preparation, numerical revision, and concept recall.



Leave a Reply