1. Introduction to Carbon
Carbon is one of the most important elements in chemistry. It is the backbone of all living organisms and forms a vast number of compounds. The branch of chemistry that deals with carbon compounds is called Organic Chemistry.
Carbon has atomic number 6 and electronic configuration 2,4. Since it has four electrons in its outermost shell, it needs four more electrons to complete its octet. This property makes carbon form covalent bonds by sharing electrons.
Carbon is unique because of:
- Tetravalency
- Catenation
- Ability to form multiple bonds
- Formation of long chains and rings
Due to these properties, carbon forms millions of compounds.
2. Bonding in Carbon – Covalent Bonding
Carbon forms covalent bonds by sharing electrons instead of gaining or losing them.
Example:
- Hydrogen (1 electron) shares one electron with carbon.
- In methane (CH₄), carbon shares four electrons with four hydrogen atoms.
Covalent Compounds – Properties
- Low melting and boiling points
- Poor conductors of electricity
- Insoluble in water but soluble in organic solvents
- Exist as gases or liquids
Examples: Methane (CH₄), Ethane (C₂H₆), Carbon dioxide (CO₂)
3. Versatile Nature of Carbon
(a) Tetravalency
Carbon has valency 4, meaning it can form four covalent bonds.
Example: CH₄ (Methane)
(b) Catenation
Carbon can form bonds with other carbon atoms, leading to long chains, branched chains, and rings.
Example:
- Ethane (C₂H₆)
- Propane (C₃H₈)
- Butane (C₄H₁₀)
This property leads to a large number of compounds.
4. Allotropes of Carbon
Allotropes are different forms of the same element having different physical properties.
1. Diamond



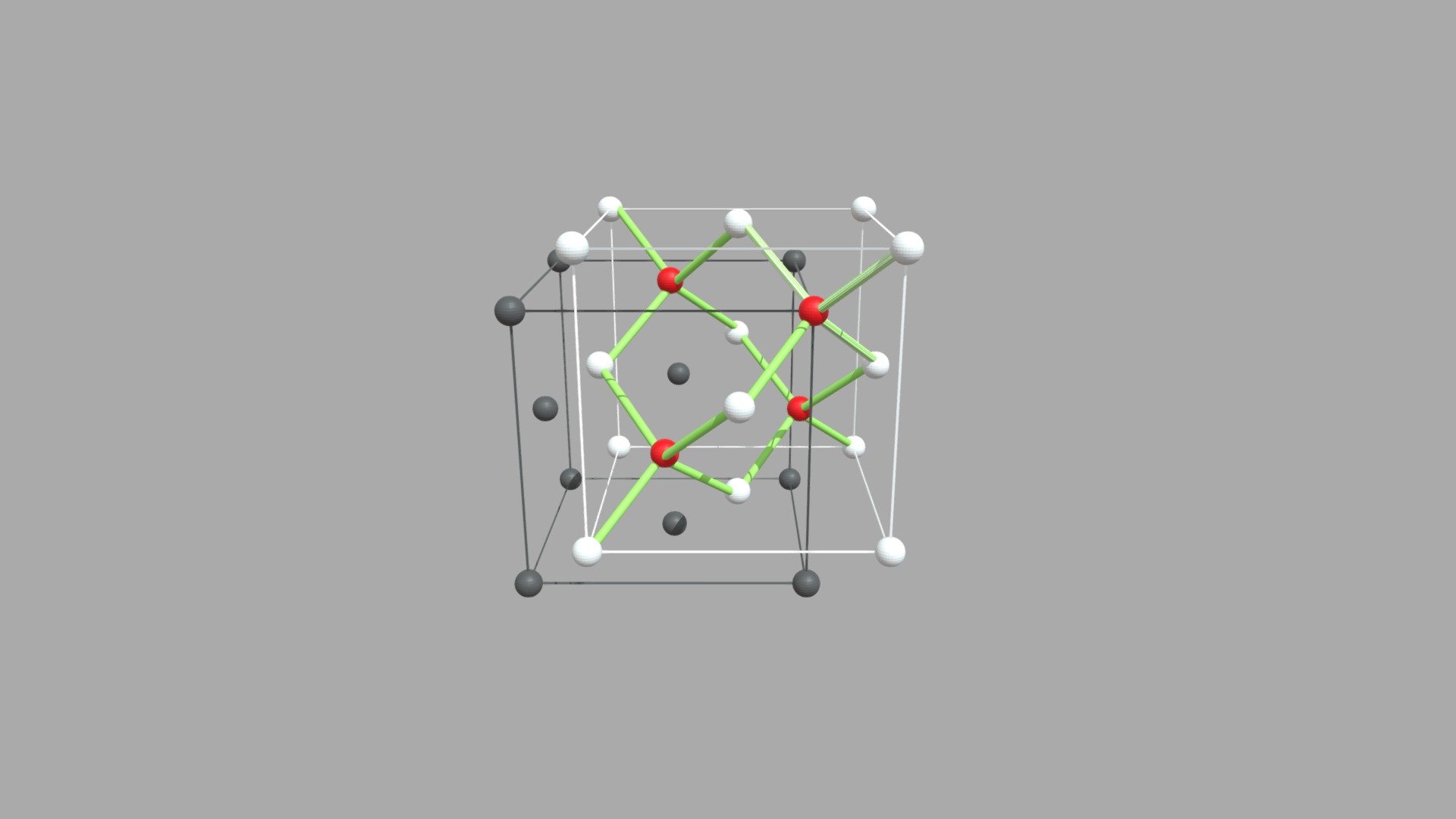
- Hardest natural substance
- Each carbon atom bonded to four others
- 3D tetrahedral structure
- High melting point
- Does not conduct electricity
Uses: Cutting tools, jewellery.
2. Graphite



- Soft and slippery
- Each carbon bonded to three others
- Layered hexagonal structure
- Conducts electricity
Uses: Pencil lead, lubricants, electrodes.
3. Fullerenes
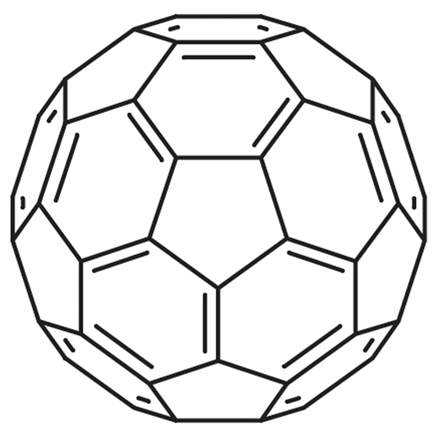


- Discovered in 1985
- C₆₀ structure resembles football
- Used in nanotechnology
5. Hydrocarbons
Hydrocarbons are compounds containing only carbon and hydrogen.
(A) Saturated Hydrocarbons (Alkanes)
- Single bonds only
- General formula: CₙH₂ₙ₊₂
- Example: Methane, Ethane
(B) Unsaturated Hydrocarbons
- Alkenes – Contain double bond
General formula: CₙH₂ₙ - Alkynes – Contain triple bond
General formula: CₙH₂ₙ₋₂
Example:
- Ethene (C₂H₄)
- Ethyne (C₂H₂)
6. Homologous Series
A homologous series is a group of organic compounds having:
- Same functional group
- Same general formula
- Similar chemical properties
- Successive members differ by –CH₂
Example:
Methane → Ethane → Propane → Butane
Each differs by CH₂ unit.
7. Functional Groups
Functional group is an atom or group of atoms that determines the chemical properties of a compound.
| Functional Group | Formula | Example |
|---|---|---|
| Alcohol | –OH | Ethanol |
| Carboxylic acid | –COOH | Ethanoic acid |
| Aldehyde | –CHO | Methanal |
| Ketone | –CO– | Propanone |
| Halogen | –Cl, –Br | Chloroethane |
Functional groups make compounds reactive.
8. Nomenclature of Carbon Compounds
Rules:
- Identify longest carbon chain.
- Number the chain.
- Identify functional group.
- Name according to IUPAC system.
Example:
CH₃–CH₂–OH → Ethanol
CH₃–COOH → Ethanoic acid
9. Chemical Properties of Carbon Compounds
(1) Combustion
Carbon compounds burn in air producing carbon dioxide, water, heat, and light.
CH₄ + 2O₂ → CO₂ + 2H₂O
Saturated hydrocarbons give clean flame. Unsaturated give sooty flame.
(2) Oxidation
Oxidizing agents like alkaline KMnO₄ oxidize alcohols to acids.
Ethanol → Ethanoic acid
(3) Addition Reaction
Unsaturated hydrocarbons add hydrogen in presence of nickel catalyst.
C₂H₄ + H₂ → C₂H₆
Used in hydrogenation of vegetable oils.
(4) Substitution Reaction
In saturated hydrocarbons, one hydrogen is replaced by another atom.
CH₄ + Cl₂ → CH₃Cl + HCl
Occurs in sunlight.
10. Ethanol (C₂H₅OH)



Properties:
- Colorless liquid
- Soluble in water
- Boiling point: 78°C
Uses:
- Solvent
- Fuel
- Alcoholic beverages
- Antiseptic
Chemical Reactions:
- With sodium → Hydrogen gas
- With acids → Ester formation
11. Ethanoic Acid (Acetic Acid)




Properties:
- Sour taste
- Vinegar contains 5–8% ethanoic acid
- Turns blue litmus red
Reactions:
- With alcohol → Ester
- With base → Salt + water
- With carbonate → CO₂ gas
12. Soaps and Detergents
Soaps
Made by reaction of fats/oils with NaOH (Saponification).
Soap molecule has:
- Hydrophilic head
- Hydrophobic tail
Forms micelles in water.




Detergents
- Work in hard water
- Do not form scum
- Synthetic compounds
Difference:
Soap → Biodegradable
Detergent → Some are non-biodegradable
13. Carbon Cycle
Carbon moves between atmosphere, plants, animals, and soil.
Processes:
- Photosynthesis
- Respiration
- Combustion
- Decomposition
Maintains balance of CO₂ in atmosphere.
14. Importance of Carbon Compounds
- Fuels (Petrol, Diesel, LPG)
- Plastics
- Medicines
- Fibres (Nylon, Polyester)
- Food substances (Carbohydrates, Proteins, Fats)
Without carbon, life would not exist.
15. Environmental Impact
Burning fossil fuels releases CO₂ causing:
- Global warming
- Air pollution
- Acid rain
Incomplete combustion produces carbon monoxide (CO), a poisonous gas.
16. Summary
Carbon is a versatile element due to tetravalency and catenation. It forms covalent bonds and millions of compounds. Its allotropes include diamond, graphite, and fullerenes. Hydrocarbons may be saturated or unsaturated. Functional groups determine chemical behavior. Important compounds include ethanol and ethanoic acid. Soaps and detergents are cleaning agents derived from carbon compounds. Carbon plays a vital role in life and environment.
1–20
- Carbon has atomic number:
A) 4
B) 6
C) 8
D) 12
Answer: B - Carbon has how many valence electrons?
A) 2
B) 3
C) 4
D) 6
Answer: C - The valency of carbon is:
A) 2
B) 3
C) 4
D) 1
Answer: C - Carbon mainly forms:
A) Ionic bonds
B) Covalent bonds
C) Metallic bonds
D) Hydrogen bonds
Answer: B - Property of carbon to form long chains is called:
A) Tetravalency
B) Catenation
C) Polymerization
D) Combustion
Answer: B - The hardest natural substance is:
A) Graphite
B) Diamond
C) Coal
D) Coke
Answer: B - Graphite conducts electricity because:
A) It is metallic
B) It has free electrons
C) It is hard
D) It is ionic
Answer: B - C₆₀ is known as:
A) Methane
B) Fullerene
C) Ethane
D) Propane
Answer: B - Hydrocarbons contain:
A) Carbon & Oxygen
B) Carbon & Hydrogen
C) Hydrogen & Oxygen
D) Carbon only
Answer: B - General formula of alkanes:
A) CₙH₂ₙ
B) CₙH₂ₙ₊₂
C) CₙH₂ₙ₋₂
D) CₙHₙ
Answer: B - Ethene belongs to:
A) Alkane
B) Alkene
C) Alkyne
D) Alcohol
Answer: B - Ethyne contains:
A) Single bond
B) Double bond
C) Triple bond
D) Ionic bond
Answer: C - Compounds in homologous series differ by:
A) CH₃
B) CH₂
C) H₂
D) CO₂
Answer: B - –OH is functional group of:
A) Acid
B) Alcohol
C) Ketone
D) Ester
Answer: B - –COOH represents:
A) Alcohol
B) Ester
C) Carboxylic acid
D) Aldehyde
Answer: C - CH₄ is:
A) Ethane
B) Methane
C) Propane
D) Butane
Answer: B - Combustion of carbon compounds produces:
A) CO₂ & H₂O
B) CO & H₂
C) O₂
D) N₂
Answer: A - Addition reaction occurs in:
A) Alkanes
B) Alkenes
C) Alcohols
D) Acids
Answer: B - Hydrogenation uses catalyst:
A) Iron
B) Copper
C) Nickel
D) Zinc
Answer: C - Ethanol formula is:
A) CH₃OH
B) C₂H₅OH
C) CH₄
D) C₂H₆
Answer: B
21–40
- Vinegar contains:
A) Methanoic acid
B) Ethanoic acid
C) HCl
D) H₂SO₄
Answer: B - Saponification is reaction of:
A) Acid & base
B) Fat & NaOH
C) Alcohol & acid
D) Salt & water
Answer: B - Soap forms:
A) Foam
B) Micelles
C) Ions
D) Gas
Answer: B - Non-biodegradable cleaning agents are:
A) Soaps
B) Detergents
C) Water
D) Alcohol
Answer: B - Ethanol boils at:
A) 50°C
B) 78°C
C) 100°C
D) 120°C
Answer: B - Ethanoic acid turns blue litmus:
A) Blue
B) Red
C) Green
D) Colorless
Answer: B - Carbon monoxide is:
A) Safe gas
B) Poisonous
C) Sweet smelling
D) Basic
Answer: B - Saturated hydrocarbons burn with:
A) Yellow flame
B) Sooty flame
C) Blue flame
D) No flame
Answer: C - Unsaturated hydrocarbons burn with:
A) Clean flame
B) Sooty flame
C) Green flame
D) No flame
Answer: B - IUPAC naming system is used for:
A) Metals
B) Organic compounds
C) Salts
D) Bases
Answer: B - CH₃–CH₃ is:
A) Methane
B) Ethane
C) Propane
D) Butane
Answer: B - CH₃–CH₂–OH is:
A) Methanol
B) Ethanol
C) Propanol
D) Butanol
Answer: B - –CHO group represents:
A) Ketone
B) Aldehyde
C) Alcohol
D) Ester
Answer: B - Graphite structure is:
A) 3D
B) Layered
C) Linear
D) Branched
Answer: B - Diamond is:
A) Soft
B) Conductor
C) Hard
D) Liquid
Answer: C - Carbon forms how many bonds?
A) 1
B) 2
C) 3
D) 4
Answer: D - Ethyne formula:
A) C₂H₂
B) C₂H₄
C) C₂H₆
D) CH₄
Answer: A - Oxidation of alcohol forms:
A) Alkane
B) Acid
C) Alkene
D) Salt
Answer: B - Ester formation is between:
A) Acid & alcohol
B) Acid & base
C) Salt & water
D) Base & metal
Answer: A - Carbon cycle maintains:
A) Oxygen
B) Nitrogen
C) CO₂ balance
D) Water
Answer: C
41–100 (Compact Format)
- Methanol formula → CH₃OH
- Propane formula → C₃H₈
- Butane formula → C₄H₁₀
- Alkene general formula → CₙH₂ₙ
- Alkyne general formula → CₙH₂ₙ₋₂
- CH₃COOH is → Ethanoic acid
- Functional group –CO– → Ketone
- Soap head is → Hydrophilic
- Soap tail is → Hydrophobic
- Catenation means → Self-linking property
- Diamond conducts electricity → No
- Graphite conducts electricity → Yes
- Ethanol is soluble in → Water
- Hydrogenation converts → Unsaturated to saturated
- CO₂ is → Greenhouse gas
- Incomplete combustion forms → CO
- Sooty flame indicates → Unsaturation
- Organic chemistry studies → Carbon compounds
- CH₃Cl is formed by → Substitution
- Nickel acts as → Catalyst
- Longest chain rule used in → IUPAC naming
- Methane has bonds → Single
- Ethene has bonds → Double
- Ethyne has bonds → Triple
- Vinegar contains → 5–8% acetic acid
- Ester has smell → Fruity
- Coal is form of → Carbon
- Coke is → Almost pure carbon
- Hydrocarbons are → Fuel
- Plastics are → Polymers
- Functional group determines → Chemical properties
- Diamond structure → Tetrahedral
- Graphite layers held by → Weak forces
- C₆₀ resembles → Football
- Photosynthesis uses → CO₂
- Respiration releases → CO₂
- Carbon forms million compounds due to → Catenation
- Soap does not work well in → Hard water
- Detergents work in → Hard water
- Ethanol reacts with sodium forming → H₂ gas
- Boiling point of ethanol → 78°C
- Ethanoic acid freezes at → 16.6°C
- Pure ethanoic acid is called → Glacial acetic acid
- Carbon valency → 4
- Methane burns with → Blue flame
- IUPAC full form → International Union of Pure and Applied Chemistry
- Addition reaction needs → Catalyst
- Organic compounds have → Low melting points
- Most covalent compounds are → Poor conductors
- Carbon dioxide is → Non-supporter of combustion
- Ethanol is used as → Fuel
- Carbon monoxide binds with → Hemoglobin
- Soaps are salts of → Fatty acids
- Unsaturated compounds decolorize → Bromine water
- Saturated compounds → Do not decolorize bromine
- Carbon cycle prevents → CO₂ imbalance
- Long carbon chains form → Polymers
- LPG contains → Propane & Butane
- CNG contains → Methane
- Carbon is basis of → Life
Here are 100 Very Short Answer Questions with Answers
1–20
- What is atomic number of carbon?
Answer: 6 - What is valency of carbon?
Answer: 4 - What type of bond does carbon mainly form?
Answer: Covalent bond - What is catenation?
Answer: Self-linking property of carbon atoms - Name hardest allotrope of carbon.
Answer: Diamond - Which allotrope conducts electricity?
Answer: Graphite - What is formula of methane?
Answer: CH₄ - General formula of alkanes?
Answer: CₙH₂ₙ₊₂ - What is an alkene?
Answer: Hydrocarbon with double bond - What is an alkyne?
Answer: Hydrocarbon with triple bond - Functional group of alcohol?
Answer: –OH - Functional group of carboxylic acid?
Answer: –COOH - What is ethanol formula?
Answer: C₂H₅OH - What is ethanoic acid formula?
Answer: CH₃COOH - What is homologous series?
Answer: Series differing by –CH₂ group - What is combustion?
Answer: Burning in oxygen - What gas forms in complete combustion?
Answer: Carbon dioxide - What is hydrogenation?
Answer: Addition of hydrogen - Catalyst in hydrogenation?
Answer: Nickel - What is fullerene formula?
Answer: C₆₀
21–40
- Which compound is vinegar?
Answer: Ethanoic acid solution - Boiling point of ethanol?
Answer: 78°C - Soap is salt of?
Answer: Fatty acids - Saponification is reaction between?
Answer: Fat and NaOH - What is micelle?
Answer: Soap molecule cluster - Unsaturated hydrocarbons give which flame?
Answer: Sooty flame - Saturated hydrocarbons give which flame?
Answer: Blue flame - Carbon monoxide is?
Answer: Poisonous gas - What is IUPAC?
Answer: International naming system - Graphite structure type?
Answer: Layered hexagonal - Diamond structure type?
Answer: Tetrahedral - CNG mainly contains?
Answer: Methane - LPG contains?
Answer: Propane and butane - Ester has smell like?
Answer: Fruity smell - Alcohol + acid gives?
Answer: Ester - Ethene formula?
Answer: C₂H₄ - Ethyne formula?
Answer: C₂H₂ - Carbon forms how many bonds?
Answer: Four - Carbon compounds have high/low melting point?
Answer: Low - Bromine water test detects?
Answer: Unsaturation
41–60
- Pure ethanoic acid is called?
Answer: Glacial acetic acid - What is substitution reaction?
Answer: Replacement of hydrogen atom - What is addition reaction?
Answer: Adding atoms to double/triple bond - Organic chemistry studies?
Answer: Carbon compounds - Coal is form of?
Answer: Carbon - Coke is almost?
Answer: Pure carbon - Photosynthesis uses which gas?
Answer: CO₂ - Respiration releases?
Answer: CO₂ - Carbon dioxide is supporter of combustion?
Answer: No - Carbon monoxide combines with?
Answer: Hemoglobin - Alcohol turns blue litmus?
Answer: No change - Ethanoic acid turns blue litmus?
Answer: Red - Detergents work in?
Answer: Hard water - Soaps work poorly in?
Answer: Hard water - Methane contains which bonds?
Answer: Single bonds - Ethene contains which bond?
Answer: Double bond - Ethyne contains which bond?
Answer: Triple bond - Functional group determines?
Answer: Chemical properties - Carbon cycle maintains?
Answer: CO₂ balance - Carbon backbone forms?
Answer: Chains and rings
61–80
- Methanol formula?
Answer: CH₃OH - Propane formula?
Answer: C₃H₈ - Butane formula?
Answer: C₄H₁₀ - Oxidation of alcohol gives?
Answer: Acid - Hydrogenation converts oil into?
Answer: Vanaspati ghee - Unsaturated compounds decolorize?
Answer: Bromine water - Long carbon chains form?
Answer: Polymers - Plastics are example of?
Answer: Polymers - Ethanol reacts with sodium to release?
Answer: Hydrogen gas - Carbon dioxide causes?
Answer: Global warming - Carbon compounds mainly are?
Answer: Covalent - Covalent compounds conduct electricity?
Answer: No - Graphite has free?
Answer: Electrons - Diamond is used for?
Answer: Cutting tools - Ethanoic acid tastes?
Answer: Sour - Methane is main component of?
Answer: Natural gas - Carbon atoms share electrons to complete?
Answer: Octet - Saturated hydrocarbons are also called?
Answer: Alkanes - Double bond hydrocarbons called?
Answer: Alkenes - Triple bond hydrocarbons called?
Answer: Alkynes
81–100
- –CHO group is?
Answer: Aldehyde - –CO– group is?
Answer: Ketone - Soaps are biodegradable?
Answer: Yes - Some detergents are biodegradable?
Answer: No - Incomplete combustion produces?
Answer: CO - Ethanol is used as?
Answer: Fuel and solvent - Homologous series members show?
Answer: Similar chemical properties - Each successive member differs by?
Answer: CH₂ - Carbon forms stable bonds due to?
Answer: Small size - Ethene reacts with hydrogen to form?
Answer: Ethane - Methane burns with?
Answer: Blue flame - Carbon compounds are basis of?
Answer: Life - Diamond is conductor?
Answer: No - Graphite is conductor?
Answer: Yes - Vinegar contains how much acetic acid?
Answer: 5–8% - Carbon cycle includes?
Answer: Photosynthesis and respiration - Longest carbon chain rule used in?
Answer: IUPAC naming - C₆₀ resembles?
Answer: Football - Carbon dioxide is acidic/basic?
Answer: Acidic oxide - Carbon belongs to which group?
Answer: Group 14
Here are Short Answer Questions with Answers
1. Explain tetravalency of carbon.
Carbon has four electrons in its outermost shell. To complete its octet, it shares four electrons with other atoms. Therefore, it forms four covalent bonds. This property is called tetravalency and allows carbon to form stable compounds.
2. What is catenation?
Catenation is the property of carbon to form bonds with other carbon atoms. This leads to long chains, branched chains, and ring structures. Because of strong carbon–carbon bonds, carbon forms millions of compounds.
3. Why does carbon form covalent compounds?
Carbon has four valence electrons and needs four more to complete its octet. Losing or gaining four electrons requires too much energy. Hence, carbon shares electrons and forms covalent bonds instead of ionic bonds.
4. Write differences between diamond and graphite.
Diamond is hard and does not conduct electricity. Each carbon atom is bonded to four atoms in a 3D structure. Graphite is soft and conducts electricity due to free electrons. It has a layered hexagonal structure.
5. What are hydrocarbons?
Hydrocarbons are compounds containing only carbon and hydrogen atoms. They are classified into saturated (alkanes) and unsaturated (alkenes and alkynes). Hydrocarbons are important fuels such as methane, propane, and butane.
6. What are saturated hydrocarbons?
Saturated hydrocarbons contain only single bonds between carbon atoms. They are called alkanes and have the general formula CₙH₂ₙ₊₂. They burn with a clean blue flame and are less reactive.
7. What are unsaturated hydrocarbons?
Unsaturated hydrocarbons contain at least one double or triple bond. Alkenes have double bonds, while alkynes have triple bonds. They are more reactive and burn with a sooty flame.
8. Define homologous series.
A homologous series is a group of organic compounds having the same functional group and general formula. Successive members differ by –CH₂ unit. They show similar chemical properties and gradual change in physical properties.
9. What is a functional group?
A functional group is an atom or group of atoms responsible for characteristic chemical properties of a compound. Examples include –OH (alcohol), –COOH (carboxylic acid), and –CHO (aldehyde).
10. Explain combustion of carbon compounds.
Combustion is the reaction of carbon compounds with oxygen producing carbon dioxide, water, heat, and light. Complete combustion gives blue flame, while incomplete combustion produces carbon monoxide and soot.
11. What is oxidation reaction in carbon compounds?
Oxidation involves addition of oxygen or removal of hydrogen. For example, ethanol oxidizes to ethanoic acid using oxidizing agents like alkaline potassium permanganate.
12. What is addition reaction?
Addition reaction occurs in unsaturated hydrocarbons. Hydrogen adds across double or triple bonds in presence of nickel catalyst. For example, ethene converts to ethane during hydrogenation.
13. What is substitution reaction?
In substitution reaction, one atom replaces another atom in a compound. For example, methane reacts with chlorine in sunlight where hydrogen is replaced by chlorine.
14. Describe ethanol properties.
Ethanol is a colorless liquid with pleasant smell. It is soluble in water and has boiling point 78°C. It is used as fuel, solvent, antiseptic, and in alcoholic beverages.
15. Describe ethanoic acid properties.
Ethanoic acid is a colorless liquid with sour taste. It turns blue litmus red and is present in vinegar (5–8%). Pure form is called glacial acetic acid.
16. What is esterification?
Esterification is reaction between alcohol and carboxylic acid in presence of concentrated sulfuric acid to form ester and water. Esters have pleasant fruity smell.
17. What is saponification?
Saponification is reaction of fats or oils with sodium hydroxide to form soap and glycerol. It is used in soap manufacturing process.
18. Explain cleansing action of soap.
Soap molecules have hydrophilic head and hydrophobic tail. In water, they form micelles trapping oil and dirt inside. These micelles are washed away, cleaning the surface.
19. Why do soaps not work well in hard water?
Hard water contains calcium and magnesium salts. These react with soap to form insoluble scum, reducing cleaning efficiency.
20. Why are detergents better than soaps?
Detergents do not form scum with hard water. They clean effectively in both soft and hard water. Hence, they are more efficient cleaning agents.
21. What is carbon cycle?
Carbon cycle is movement of carbon between atmosphere, plants, animals, and soil. It includes photosynthesis, respiration, combustion, and decomposition processes.
22. Why is carbon monoxide dangerous?
Carbon monoxide is poisonous because it binds with hemoglobin in blood, reducing oxygen transport. This may cause suffocation and death.
23. What is IUPAC naming?
IUPAC naming is systematic method of naming organic compounds based on longest carbon chain and functional groups.
24. Why do carbon compounds have low melting points?
Carbon compounds are covalent. They have weak intermolecular forces, requiring less energy to break, resulting in low melting and boiling points.
25. Why is methane important?
Methane is main component of natural gas. It is used as fuel and produces less pollution on burning.
26. What is fullerene?
Fullerene is allotrope of carbon with closed cage structure like C₆₀. It resembles a football and is used in nanotechnology.
27. Why does diamond not conduct electricity?
In diamond, all four valence electrons are used in bonding. There are no free electrons available for conduction.
28. Why does graphite conduct electricity?
In graphite, each carbon bonds with three atoms, leaving one free electron. These free electrons move between layers, allowing conduction.
29. What is hydrogenation of oils?
Hydrogenation is addition of hydrogen to vegetable oils using nickel catalyst to convert them into saturated fats like vanaspati ghee.
30. What is glacial acetic acid?
Pure ethanoic acid without water is called glacial acetic acid. It solidifies at 16.6°C and looks like ice.
31. What are alkanes?
Alkanes are saturated hydrocarbons containing only single covalent bonds between carbon atoms. They have the general formula CₙH₂ₙ₊₂. Alkanes are relatively less reactive and burn with a clean blue flame.
32. What are alkenes?
Alkenes are unsaturated hydrocarbons containing at least one carbon–carbon double bond. Their general formula is CₙH₂ₙ. They are more reactive than alkanes and undergo addition reactions.
33. What are alkynes?
Alkynes are unsaturated hydrocarbons containing at least one carbon–carbon triple bond. Their general formula is CₙH₂ₙ₋₂. They are highly reactive and participate in addition reactions.
34. What is the importance of functional groups?
Functional groups determine the chemical properties and reactivity of organic compounds. Compounds with the same functional group show similar chemical behavior despite differences in carbon chain length.
35. Explain the structure of methane.
Methane (CH₄) consists of one carbon atom bonded to four hydrogen atoms by single covalent bonds. The molecule has a tetrahedral shape with bond angle of 109.5°.
36. Why are carbon–carbon bonds strong?
Carbon atoms are small in size, allowing effective overlap of orbitals. This results in strong covalent bonds, enabling formation of stable long chains and complex structures.
37. What is decolorization of bromine water?
Unsaturated hydrocarbons decolorize reddish-brown bromine water due to addition reaction across double or triple bonds. It is used as a test for unsaturation.
38. What is polymerization?
Polymerization is the process in which small molecules called monomers join together to form large molecules called polymers. Plastics are common examples of polymers.
39. Why are esters used in perfumes?
Esters have pleasant fruity smells. Due to their sweet odor, they are widely used in perfumes, flavoring agents, and food industries.
40. What happens during incomplete combustion?
Incomplete combustion occurs when oxygen supply is limited. It produces carbon monoxide and soot along with less heat energy, causing pollution.
41. What are aldehydes?
Aldehydes are organic compounds containing –CHO functional group. They are formed by oxidation of primary alcohols and are important in perfumes and preservatives.
42. What are ketones?
Ketones are organic compounds containing carbonyl group (–CO–) within the carbon chain. They are formed by oxidation of secondary alcohols.
43. What is the role of sulfuric acid in esterification?
Concentrated sulfuric acid acts as a catalyst and dehydrating agent. It removes water formed during esterification, increasing the yield of ester.
44. Why is ethanol used as fuel?
Ethanol burns cleanly, producing carbon dioxide and water. It produces less smoke and is renewable, making it a good alternative fuel.
45. Why are carbon compounds poor conductors?
Most carbon compounds are covalent and do not have free ions or electrons. Therefore, they cannot conduct electricity effectively.
46. What are biodegradable substances?
Biodegradable substances are materials that can be decomposed by microorganisms into simpler substances. Soaps are biodegradable cleaning agents.
47. Why do unsaturated hydrocarbons burn with sooty flame?
Unsaturated hydrocarbons have higher carbon content and incomplete combustion produces unburnt carbon particles, giving a yellow sooty flame.
48. What is the importance of methane?
Methane is widely used as domestic and industrial fuel. It is a major component of CNG and natural gas.
49. What is the role of carbon in living organisms?
Carbon forms complex molecules like carbohydrates, proteins, fats, and nucleic acids, which are essential for life processes.
50. Explain the term isomerism.
Isomerism is the phenomenon where compounds have same molecular formula but different structural arrangements and properties.
51. What are structural isomers?
Structural isomers have the same molecular formula but different structural formulas. Example: Butane and isobutane.
52. Why does diamond have high melting point?
Diamond has strong covalent bonds throughout its 3D structure. A large amount of energy is required to break these bonds.
53. Why is graphite soft?
Graphite has layered structure with weak forces between layers. Layers slide over each other easily, making it soft and slippery.
54. What is natural gas?
Natural gas is a fossil fuel mainly composed of methane. It is used for cooking, heating, and power generation.
55. What is LPG?
Liquefied Petroleum Gas consists mainly of propane and butane. It is used as domestic cooking fuel.
56. What is CNG?
Compressed Natural Gas mainly contains methane. It is used as eco-friendly fuel in vehicles.
57. What is global warming?
Global warming is rise in Earth’s temperature due to greenhouse gases like carbon dioxide produced from burning fossil fuels.
58. What is an acid test for unsaturation?
Bromine water test is used to detect unsaturation. Unsaturated compounds decolorize bromine water.
59. Why is carbon versatile?
Carbon is versatile due to tetravalency, catenation, and ability to form single, double, and triple bonds.
60. What is a covalent bond?
A covalent bond is formed by sharing of electrons between atoms to achieve stable electronic configuration.
61. What are carboxylic acids?
Carboxylic acids contain –COOH functional group. They are weak acids and have sour taste.
62. What is fermentation?
Fermentation is process in which sugars are converted into ethanol and carbon dioxide by yeast in absence of oxygen.
63. What is ethene used for?
Ethene is used in manufacture of plastics like polythene and as ripening agent for fruits.
64. What is ethyne used for?
Ethyne (acetylene) is used in oxy-acetylene welding due to its high flame temperature.
65. What are fossil fuels?
Fossil fuels are carbon-containing fuels formed from remains of plants and animals, such as coal, petroleum, and natural gas.
66. Why is carbon dioxide non-supporter of combustion?
Carbon dioxide does not burn and does not support burning. It is used in fire extinguishers.
67. What is vanaspati ghee?
Vanaspati ghee is hydrogenated vegetable oil formed by hydrogenation of unsaturated oils.
68. What is octet rule?
Octet rule states that atoms tend to gain, lose, or share electrons to achieve eight electrons in outer shell.
69. Why does carbon not form C⁴⁺ or C⁴⁻ ions?
Forming C⁴⁺ or C⁴⁻ requires very high energy. Hence, carbon shares electrons instead of forming ions.
70. What is acetone?
Acetone is a ketone with formula CH₃COCH₃. It is used as solvent and nail polish remover.
LONG QUESTION ANSWER
1. Explain the versatile nature of carbon.
Carbon is considered a versatile element because it forms a vast number of compounds. This versatility is mainly due to tetravalency and catenation. Carbon has four valence electrons and forms four covalent bonds, allowing it to combine with different elements such as hydrogen, oxygen, nitrogen, and halogens. It also forms strong carbon–carbon bonds, enabling the formation of long chains, branched chains, and ring structures. Additionally, carbon can form single, double, and triple bonds, increasing structural diversity. These properties allow carbon to form millions of organic compounds, including fuels, plastics, medicines, and biomolecules. The strength and stability of carbon bonds further contribute to the existence of complex life forms on Earth.
2. Describe the allotropes of carbon and their properties.
Carbon exists in different forms called allotropes. The three main allotropes are diamond, graphite, and fullerenes. Diamond is the hardest natural substance, where each carbon atom is bonded to four others in a three-dimensional tetrahedral structure. It does not conduct electricity and has a very high melting point. Graphite has a layered hexagonal structure in which each carbon atom is bonded to three others, leaving one free electron that allows it to conduct electricity. It is soft and slippery. Fullerenes, such as C₆₀, have spherical cage-like structures and are used in nanotechnology. The difference in properties arises due to different bonding arrangements of carbon atoms.
3. Explain covalent bonding in carbon compounds.
Carbon forms covalent bonds by sharing electrons with other atoms to complete its octet. Since carbon has four electrons in its outermost shell, losing or gaining four electrons is energetically unfavorable. Therefore, it shares electrons with atoms like hydrogen, oxygen, or other carbon atoms. For example, in methane (CH₄), carbon shares one electron with each hydrogen atom, forming four single covalent bonds. Covalent compounds generally have low melting and boiling points because the forces between molecules are weak. They are poor conductors of electricity due to absence of free ions or electrons. Covalent bonding is responsible for the stability and variety of carbon compounds.
4. What are hydrocarbons? Classify them with examples.
Hydrocarbons are organic compounds that contain only carbon and hydrogen atoms. They are classified into saturated and unsaturated hydrocarbons. Saturated hydrocarbons, called alkanes, contain only single bonds and have the general formula CₙH₂ₙ₊₂. Examples include methane and ethane. Unsaturated hydrocarbons contain double or triple bonds. Alkenes have double bonds (CₙH₂ₙ), such as ethene. Alkynes have triple bonds (CₙH₂ₙ₋₂), such as ethyne. Saturated hydrocarbons burn with a clean blue flame, while unsaturated hydrocarbons burn with a sooty flame. Hydrocarbons are widely used as fuels and in the manufacture of plastics and chemicals.
5. Explain homologous series with characteristics.
A homologous series is a group of organic compounds having the same functional group and similar chemical properties. Members of a homologous series follow a general formula and differ from each other by a –CH₂ unit. For example, methane, ethane, propane, and butane belong to the alkane homologous series. As molecular mass increases, physical properties such as boiling point and melting point gradually change. However, chemical properties remain similar due to the same functional group. Homologous series helps in systematic study and classification of organic compounds.
6. Describe the chemical properties of carbon compounds.
Carbon compounds show various chemical reactions such as combustion, oxidation, addition, and substitution. In combustion, carbon compounds react with oxygen to produce carbon dioxide, water, heat, and light. Oxidation involves addition of oxygen or removal of hydrogen, such as conversion of ethanol into ethanoic acid. Addition reactions occur in unsaturated hydrocarbons where atoms add across double or triple bonds, for example hydrogenation of ethene. Substitution reactions mainly occur in saturated hydrocarbons where one atom replaces another, such as chlorination of methane. These reactions highlight the reactivity of carbon compounds.
7. Explain properties and uses of ethanol.
Ethanol (C₂H₅OH) is a colorless liquid with a pleasant smell and boiling point of 78°C. It is soluble in water and neutral in nature. Ethanol is produced by fermentation of sugars using yeast. Chemically, it reacts with sodium to produce hydrogen gas and forms esters with acids. Ethanol is widely used as a solvent in medicines and perfumes. It is also used as fuel and blended with petrol to reduce pollution. Additionally, it is used in alcoholic beverages and as an antiseptic.
8. Explain properties and reactions of ethanoic acid.
Ethanoic acid (CH₃COOH), also called acetic acid, is a weak organic acid with sour taste. It turns blue litmus red and is present in vinegar (5–8%). Pure ethanoic acid is called glacial acetic acid. It reacts with alcohols to form esters, with metals to produce hydrogen gas, and with bases to form salts and water. It also reacts with carbonates to produce carbon dioxide. Ethanoic acid is used in food preservation, vinegar preparation, and chemical industries.
9. Describe the cleansing action of soaps and detergents.
Soaps are sodium or potassium salts of long-chain fatty acids. Each soap molecule has a hydrophilic (water-attracting) head and hydrophobic (water-repelling) tail. In water, soap molecules form micelles around oil or grease particles. The hydrophobic tails dissolve in grease, while hydrophilic heads remain in water. When rinsed, micelles are washed away, removing dirt. However, soaps do not work well in hard water because they form scum. Detergents overcome this problem as they do not form insoluble salts with hard water ions.
10. Explain the carbon cycle and its importance.
The carbon cycle is the continuous circulation of carbon between atmosphere, plants, animals, and the Earth’s crust. Plants absorb carbon dioxide during photosynthesis to prepare food. Animals obtain carbon by eating plants. Respiration and decomposition release carbon dioxide back into the atmosphere. Combustion of fossil fuels also adds carbon dioxide to air. This cycle maintains the balance of carbon dioxide in nature. If the balance is disturbed, it may lead to environmental problems such as global warming. Thus, the carbon cycle is essential for sustaining life on Earth.
Here are 50 Assertion–Reason Questions
Directions:
A. Both Assertion (A) and Reason (R) are true and R is correct explanation of A.
B. Both A and R are true but R is not correct explanation of A.
C. A is true but R is false.
D. A is false but R is true.
1–10
- A: Carbon forms a large number of compounds.
R: Carbon shows catenation and tetravalency.
Answer: A - A: Diamond does not conduct electricity.
R: All four valence electrons of carbon are used in bonding.
Answer: A - A: Graphite conducts electricity.
R: It has free electrons between layers.
Answer: A - A: Carbon forms ionic compounds easily.
R: Carbon has four valence electrons.
Answer: D - A: Unsaturated hydrocarbons decolorize bromine water.
R: They contain double or triple bonds.
Answer: A - A: Saturated hydrocarbons are less reactive.
R: They contain only single bonds.
Answer: A - A: Ethanol reacts with sodium.
R: Ethanol contains –OH group.
Answer: A - A: Soaps do not work well in hard water.
R: Hard water contains calcium and magnesium salts.
Answer: A - A: Detergents work in hard water.
R: They do not form insoluble scum.
Answer: A - A: Carbon monoxide is poisonous.
R: It combines with hemoglobin.
Answer: A
11–20
- A: Methane burns with blue flame.
R: It is a saturated hydrocarbon.
Answer: A - A: Ethene undergoes addition reaction.
R: It contains a double bond.
Answer: A - A: Homologous series members show similar chemical properties.
R: They have same functional group.
Answer: A - A: Carbon compounds have low melting points.
R: They are covalent compounds.
Answer: A - A: Ethanoic acid turns blue litmus red.
R: It is an acid.
Answer: A - A: Diamond is hard.
R: It has strong 3D network of covalent bonds.
Answer: A - A: Graphite is soft.
R: Layers are held by weak forces.
Answer: A - A: Hydrogenation converts oils into fats.
R: Hydrogen adds across double bonds.
Answer: A - A: Carbon forms four bonds.
R: Carbon has valency four.
Answer: A - A: CNG is cleaner fuel.
R: It mainly contains methane.
Answer: B
21–30
- A: Ethyne is more reactive than ethane.
R: Ethyne has triple bond.
Answer: A - A: Methane undergoes substitution reaction.
R: Methane is saturated hydrocarbon.
Answer: A - A: Esters have fruity smell.
R: They are formed from acids and alcohols.
Answer: B - A: Carbon dioxide supports combustion.
R: It is an oxide of carbon.
Answer: D - A: Carbon does not form C⁴⁺ ions easily.
R: It requires high ionization energy.
Answer: A - A: Ethanol is used as solvent.
R: It dissolves many organic compounds.
Answer: A - A: Fullerene has spherical structure.
R: It contains hexagonal and pentagonal rings.
Answer: A - A: Soap solution is basic.
R: It contains sodium salts of fatty acids.
Answer: A - A: Carbon compounds conduct electricity.
R: They are covalent.
Answer: D - A: LPG contains propane and butane.
R: These are hydrocarbons.
Answer: B
31–40
- A: Ethanoic acid reacts with metals.
R: It is an acid.
Answer: A - A: Polythene is a polymer.
R: It is formed by polymerization of ethene.
Answer: A - A: Methanol is poisonous.
R: It affects nervous system.
Answer: A - A: Carbon cycle is important.
R: It maintains CO₂ balance.
Answer: A - A: Unsaturated hydrocarbons burn with sooty flame.
R: They have higher carbon content.
Answer: A - A: Carbon monoxide is colorless.
R: It is formed during complete combustion.
Answer: C - A: Ethanol boils at 78°C.
R: It is a volatile liquid.
Answer: B - A: IUPAC naming is systematic.
R: It is internationally accepted.
Answer: B - A: Butane has isomers.
R: It has four carbon atoms.
Answer: A - A: Graphite is used in electrodes.
R: It conducts electricity.
Answer: A
41–50
- A: Ethene decolorizes bromine water.
R: It contains double bond.
Answer: A - A: Methane is greenhouse gas.
R: It traps heat in atmosphere.
Answer: A - A: Diamond is used in cutting tools.
R: It is very hard.
Answer: A - A: Soaps are biodegradable.
R: They are decomposed by microorganisms.
Answer: A - A: Carbon compounds form isomers.
R: Carbon forms chains and branches.
Answer: A - A: Acetone is a ketone.
R: It contains –CO– group.
Answer: A - A: Ethanoic acid reacts with carbonates.
R: It produces CO₂ gas.
Answer: A - A: Carbon dioxide causes global warming.
R: It is a greenhouse gas.
Answer: A - A: Saturated hydrocarbons do not decolorize bromine water.
R: They lack double bonds.
Answer: A - A: Carbon is basis of life.
R: It forms complex biomolecules.
Answer: A



Leave a Reply