Classification of Elements and Periodicity in Properties – Class 11 Chemistry Notes
Meta Description:
Comprehensive Class 11 Chemistry notes on Classification of Elements and Periodicity in Properties with summary, MCQs, keywords, and exam tips.
Introduction to Classification of Elements and Periodicity in Properties
The study of Classification of Elements and Periodicity in Properties is fundamental to understanding Chemistry. The chapter focuses on how elements are organized based on their atomic structure, properties, and behavior. From Dmitri Mendeleev’s Periodic Table to the modern long-form periodic table, this chapter explains the periodic trends such as atomic size, ionization energy, electron affinity, and electronegativity. Learning this chapter helps students predict the chemical behavior of elements and is crucial for both board exams and competitive exams like NEET and JEE.
Short Notes (Bullet Points)
- Periodic Law: Properties of elements are a periodic function of their atomic numbers.
- Mendeleev’s Periodic Table: Based on increasing atomic mass; predicted properties of undiscovered elements.
- Modern Periodic Table: Based on increasing atomic number; consists of 18 groups and 7 periods.
- Groups: Vertical columns with similar chemical properties.
- Periods: Horizontal rows showing gradual change in properties.
- s, p, d, f-block Elements: Classification based on valence electron configuration.
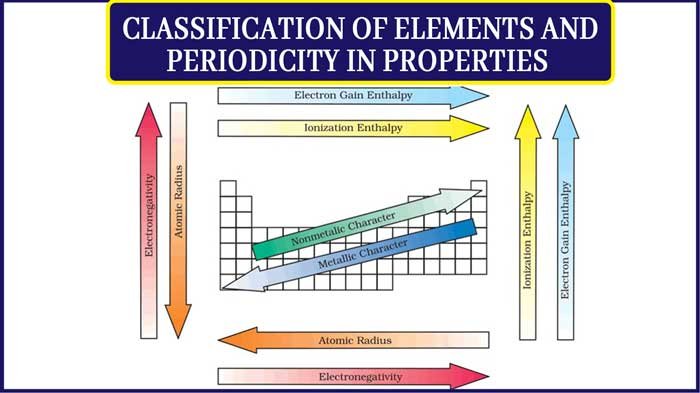
- Atomic Radius: Decreases across a period, increases down a group.
- Ionization Energy: Energy required to remove an electron; increases across a period, decreases down a group.
- Electron Affinity: Energy change on gaining an electron; generally increases across a period.
- Electronegativity: Tendency of an atom to attract electrons; increases across a period, decreases down a group.
- Metallic Character: Decreases across a period, increases down a group.
- Non-Metals: High ionization energy, high electronegativity, gain electrons to form anions.
Detailed Summary (1000–1200 words)
1. Introduction to Periodic Classification
The diversity of elements requires a systematic arrangement. Early attempts included Dobereiner’s Triads and Newlands’ Law of Octaves, but these could not accommodate all known elements. Mendeleev arranged elements by increasing atomic mass, leaving gaps for undiscovered elements. Modern Periodic Law, proposed by Henry Moseley, states that the properties of elements are periodic functions of their atomic number (Z).
2. Mendeleev’s Periodic Table
Mendeleev’s table classified elements into vertical groups and horizontal periods. Advantages: predicted properties of undiscovered elements like gallium (Ga) and germanium (Ge). Limitations: placement of isotopes and hydrogen anomalies.
3. Modern Periodic Table
- Arrangement: By increasing atomic number.
- Periods: 7 horizontal rows; properties gradually change across a period.
- Groups: 18 vertical columns; elements in the same group have similar valence electron configurations.
- Blocks:
- s-block: Groups 1–2
- p-block: Groups 13–18
- d-block: Transition metals, Groups 3–12
- f-block: Lanthanides and Actinides
4. Periodicity in Properties
A. Atomic Radius
- Distance from nucleus to outermost electron.
- Trend across a period: Decreases due to increased nuclear charge.
- Trend down a group: Increases due to addition of electron shells.
B. Ionization Energy (IE)
- Energy required to remove an electron from a gaseous atom.
- Trend across a period: Increases due to higher nuclear charge.
- Trend down a group: Decreases due to increased distance from nucleus.
C. Electron Affinity (EA)
- Energy change when an atom gains an electron.
- Trend across a period: Generally increases (halogens have highest EA).
- Trend down a group: Decreases as electrons are added farther from nucleus.
D. Electronegativity
- Ability of an atom to attract electrons in a covalent bond.
- Trend across a period: Increases (fluorine highest).
- Trend down a group: Decreases due to distance from nucleus.
E. Metallic and Non-Metallic Character
- Metals lose electrons easily; non-metals gain electrons.
- Metallic character: Decreases across a period, increases down a group.
- Non-metallic character: Opposite trend.
5. Significance of Periodicity
- Explains chemical reactivity.
- Helps predict physical properties like melting point, boiling point, density.
- Useful in chemical bonding and molecular structure studies.
6. Special Trends
- Transition Elements: Show variable oxidation states and form colored compounds.
- Lanthanides & Actinides: Exhibit f-orbital contraction, high atomic and ionic sizes.
This chapter forms the basis of inorganic chemistry and helps in predicting reactions and compound formation. Understanding these trends is essential for students aiming for competitive exams as questions are often application-based.
Flowchart / Mind Map (Text-based)
Classification of Elements and Periodicity in Properties
|
|-- Early Attempts
| |-- Dobereiner's Triads
| |-- Newlands’ Law of Octaves
|
|-- Mendeleev’s Periodic Table
| |-- Arranged by atomic mass
| |-- Predicted undiscovered elements
|
|-- Modern Periodic Table
| |-- Based on atomic number
| |-- 7 Periods, 18 Groups
| |-- s, p, d, f-block
|
|-- Periodicity in Properties
|-- Atomic Radius (↓ across period, ↑ down group)
|-- Ionization Energy (↑ across period, ↓ down group)
|-- Electron Affinity (↑ across period, ↓ down group)
|-- Electronegativity (↑ across period, ↓ down group)
|-- Metallic Character (↓ across period, ↑ down group)
|-- Non-Metallic Character (↑ across period, ↓ down group)
Important Keywords with Meanings
- Periodic Law: Properties of elements are periodic functions of atomic number.
- Atomic Radius: Distance from nucleus to outermost electron.
- Ionization Energy: Energy to remove an electron.
- Electron Affinity: Energy released when an atom gains an electron.
- Electronegativity: Ability to attract electrons.
- s-block, p-block, d-block, f-block: Classification based on valence orbitals.
- Metallic Character: Tendency to lose electrons.
- Non-Metallic Character: Tendency to gain electrons.
- Transition Elements: d-block metals with variable oxidation states.
- Lanthanides/Actinides: f-block elements with unique properties.
Important Questions & Answers
💫Very Short Answer Questions (10)
- Define Periodic Law.
- Answer: Properties of elements are a periodic function of their atomic numbers.
- Which element has the smallest atomic radius in Period 2?
- Answer: Neon (Ne).
- State one limitation of Mendeleev’s Periodic Table.
- Answer: Cannot correctly place isotopes.
- Define electronegativity.
- Answer: Ability of an atom to attract electrons in a covalent bond.
- Why does ionization energy decrease down a group?
- Answer: Outer electrons are farther from nucleus; attraction is weaker.
- Give an example of a transition element.
- Answer: Iron (Fe).
- What is electron affinity?
- Answer: Energy change when an atom gains an electron.
- Which group contains alkali metals?
- Answer: Group 1.
- Define metallic character.
- Answer: Tendency to lose electrons.
- What is the trend of atomic radius across a period?
- Answer: Decreases from left to right.
💫Short Answer Questions (10)
- Explain the limitations of Dobereiner’s Triads.
- Answer: Could only classify a few elements; no clear pattern for all elements.
- Discuss the modern periodic table and its significance.
- Answer: Arranged by atomic number; explains periodic trends and predicts properties.
- Compare Mendeleev’s table with Modern Periodic Table.
- Answer: Modern table based on atomic number, resolves anomalies of Mendeleev’s table.
- Describe s-block and p-block elements with examples.
- Answer: s-block: Groups 1–2 (Li, Be); p-block: Groups 13–18 (B, C, N).
- Explain periodic trends in atomic radius.
- Answer: Decreases across period, increases down group due to nuclear charge and shielding.
- Discuss ionization energy trends.
- Answer: Increases across a period; decreases down a group.
- Explain electronegativity trends with examples.
- Answer: F highest in period 2; decreases down a group.
- Describe metallic and non-metallic character trends.
- Answer: Metals: left side; non-metals: right side of periodic table.
- Explain electron affinity trend across periods.
- Answer: Increases left to right; halogens highest EA.
- Discuss the significance of periodicity in chemical properties.
- Answer: Helps predict reactivity, bonding, and physical properties.
30 MCQs with Answers
- Who proposed the Modern Periodic Law?
- Answer: Henry Moseley
- The atomic radius decreases across a period because:
- Answer: Nuclear charge increases
- Which element has the highest electronegativity?
- Answer: Fluorine
- s-block includes which groups?
- Answer: 1 and 2
- Transition elements are:
- Answer: d-block elements
- Group 17 elements are called:
- Answer: Halogens
- Which element has the lowest ionization energy in Period 2?
- Answer: Lithium
- Which block contains f-orbitals?
- Answer: f-block
- Electron affinity is highest for:
- Answer: Chlorine
- Metals are located on which side of periodic table?
- Answer: Left
Exam Tips / 5 Value-Based Questions with Answers
- Tip: Remember trends using mnemonic: “FINE RN” – Fluorine highest electronegativity, Radius decreases, Ionization Energy increases, Nuclear charge matters.
- Value-Based Q1: How does knowledge of periodic properties help in daily life?
- Answer: Helps in choosing materials for batteries, medicines, and alloys.
- Value-Based Q2: Predict reactivity of alkali metals with water.
- Answer: Highly reactive; increases down the group.
- Value-Based Q3: Why is fluorine used in toothpaste?
- Answer: High electronegativity prevents dental cavities.
- Value-Based Q4: How do periodic trends assist chemists in synthesis?
- Answer: Predict behavior of elements and reactions.
- Value-Based Q5: Why is hydrogen placed separately in periodic table?
- Answer: Exhibits properties of both alkali metals and halogens.
Conclusion
The chapter Classification of Elements and Periodicity in Properties forms the foundation of inorganic chemistry. It enables students to understand and predict the behavior of elements based on their position in the periodic table. The study of atomic radius, ionization energy, electron affinity, and electronegativity helps explain reactivity, bonding, and compound formation.
From Mendeleev’s early periodic table to the modern long-form table, this chapter demonstrates the systematic nature of chemistry. Knowledge of periodic trends is essential for solving board exam questions and performing well in competitive exams like NEET and JEE.
Students should focus on:
- Understanding group and period trends.
- Memorizing s, p, d, f-block classifications.
- Practicing MCQs, short and long answer questions regularly.
- Drawing flowcharts/mind maps for quick revision.
By mastering this chapter, students develop a strong foundation for chemical bonding, reactivity, and advanced chemistry concepts. The ability to predict the chemical and physical properties of elements gives an edge in exams and practical chemistry applications.
With short notes, detailed summaries, MCQs, keywords, and exam tips, students can revise effectively and score high marks. Remember, practice, visualization, and understanding trends are key to mastering Classification of Elements and Periodicity in Properties.
This chapter not only improves academic performance but also enhances analytical and problem-solving skills, which are crucial for a career in science, research, and competitive exams. A consistent approach combining notes, questions, and periodic trend analysis will make students confident and exam-ready.
Long question Answer 💫
1. Explain the limitations of Dobereiner’s Triads.
Answer:
Dobereiner grouped elements in triads, where the atomic mass of the middle element was approximately the average of the other two.
Limitations:
- Could classify only a few elements (like Li, Na, K).
- Could not accommodate all known elements.
- Did not explain trends in properties for heavier elements.
- Failed to predict new elements.
2. Describe Newlands’ Law of Octaves and its limitations.
Answer:
- John Newlands arranged elements by increasing atomic mass and noticed that every eighth element had similar properties, called Law of Octaves.
Limitations: - Worked only for lighter elements.
- Did not leave gaps for undiscovered elements.
- Grouping was inconsistent for heavier elements.
3. Explain Mendeleev’s Periodic Table and its significance.
Answer:
- Mendeleev arranged elements by increasing atomic mass.
- Placed elements with similar properties in vertical columns (groups).
- Left gaps for undiscovered elements.
Significance: - Predicted properties of elements like Gallium (Ga) and Germanium (Ge).
- Helped in systematizing chemistry knowledge.
Limitations: - Could not correctly place isotopes.
- Hydrogen placement was uncertain.
4. Compare Mendeleev’s Periodic Table with Modern Periodic Table.
Answer:
| Feature | Mendeleev’s Table | Modern Periodic Table |
|---|---|---|
| Basis | Atomic Mass | Atomic Number |
| Periodic Law | Properties repeat after atomic mass | Properties repeat after atomic number |
| Gaps | Left for undiscovered elements | No gaps, isotopes considered |
| Hydrogen | Ambiguous placement | Placed based on electronic configuration |
| Groups | 8 vertical groups | 18 vertical groups |
| Significance | Predicted new elements | Explains periodic trends precisely |
5. Describe s-block and p-block elements with examples.
Answer:
- s-block: Groups 1–2; valence electrons in s-orbital.
- Examples: Lithium (Li), Beryllium (Be)
- Properties: Highly reactive metals, form basic oxides.
- p-block: Groups 13–18; valence electrons in p-orbital.
- Examples: Boron (B), Carbon (C), Nitrogen (N)
- Properties: Includes metals, metalloids, and non-metals; forms acidic oxides.
6. Explain the trends in Atomic Radius across a period and down a group.
Answer:
- Across a period: Atomic radius decreases because nuclear charge increases, pulling electrons closer to the nucleus.
- Down a group: Atomic radius increases because new electron shells are added, increasing distance from the nucleus.
Example: Li > Be > B > C > N > O > F (across Period 2).
7. Discuss the trends in Ionization Energy (IE).
Answer:
- Ionization Energy: Energy required to remove an electron from a gaseous atom.
- Across a period: IE increases due to higher nuclear charge.
- Down a group: IE decreases due to shielding effect and increased distance from nucleus.
Example: Na (495 kJ/mol) < Mg (737 kJ/mol) < Al (577 kJ/mol).
8. Explain Electronegativity and its periodic trends.
Answer:
- Electronegativity: Ability of an atom to attract shared electrons in a covalent bond.
- Across a period: Electronegativity increases because atoms are smaller and nuclear charge is higher.
- Down a group: Electronegativity decreases due to increased atomic size.
Example: F > O > N > C (Period 2), decreases from top to bottom in Group 17.
9. Describe Metallic and Non-Metallic Character trends.
Answer:
- Metallic Character: Tendency to lose electrons and form cations.
- Trend: Decreases across a period, increases down a group.
- Non-Metallic Character: Tendency to gain electrons and form anions.
- Trend: Increases across a period, decreases down a group.
Example: Sodium (metallic) vs. Chlorine (non-metallic).
- Trend: Increases across a period, decreases down a group.
10. Explain the significance of Periodicity in Chemical Properties.
Answer:
- Predicts chemical reactivity: Helps determine how elements form compounds.
- Explains physical properties: Such as melting point, boiling point, density.
- Assists in chemical bonding: Knowledge of electronegativity and atomic size predicts bond type.
- Foundation for advanced studies: Essential for understanding inorganic chemistry, transition elements, and f-block elements.
- Useful in exams and competitive exams: Helps solve application-based questions efficiently.
Assertion-Reason Questions (20)
Instructions for Students:
- Assertion (A): A statement that may be true or false.
- Reason (R): The explanation for the assertion.
- Choose:
- Both A and R are true, and R is the correct explanation of A
- Both A and R are true, but R is not the correct explanation of A
- A is true, R is false
- A is false, R is true
1.
A: Atomic radius decreases across a period.
R: Nuclear charge increases across a period, pulling electrons closer.
Answer: 1 – Both true, R correctly explains A.
2.
A: Ionization energy decreases down a group.
R: Atomic size increases down a group.
Answer: 1 – Both true, R correctly explains A.
3.
A: Fluorine has the highest electronegativity in Period 2.
R: Fluorine has the smallest atomic size in the period.
Answer: 1 – Both true, R correctly explains A.
4.
A: Mendeleev’s table is arranged by increasing atomic mass.
R: Modern periodic table is arranged by increasing atomic number.
Answer: 1 – Both true, but R does not explain A → Option 2.
5.
A: Electron affinity increases across a period.
R: Atomic radius decreases across a period.
Answer: 1 – Both true, R explains the trend → Option 1.
6.
A: Alkali metals are highly reactive.
R: They have low ionization energy.
Answer: 1 – Both true, R correctly explains A.
7.
A: Chlorine is more reactive than fluorine.
R: Chlorine is larger in size compared to fluorine.
Answer: 3 – A is false, R is true (Actually, fluorine is more reactive).
8.
A: Ionization energy of noble gases is very high.
R: Noble gases have stable electronic configuration.
Answer: 1 – Both true, R correctly explains A.
9.
A: Metallic character increases down a group.
R: Atomic size increases down a group.
Answer: 1 – Both true, R correctly explains A.
10.
A: Neon has a larger atomic radius than sodium.
R: Neon has 10 electrons while sodium has 11 electrons.
Answer: 3 – A is false, R is true (Neon radius is smaller than Na).
11.
A: Transition metals exhibit variable oxidation states.
R: They have incompletely filled d-orbitals.
Answer: 1 – Both true, R correctly explains A.
12.
A: Electron affinity of halogens is high.
R: Halogens need only one electron to complete their octet.
Answer: 1 – Both true, R correctly explains A.
13.
A: Ionization energy increases across a period.
R: Electrons are added to the same shell while nuclear charge increases.
Answer: 1 – Both true, R correctly explains A.
14.
A: Hydrogen is placed separately in the periodic table.
R: Hydrogen exhibits properties of both alkali metals and halogens.
Answer: 1 – Both true, R correctly explains A.
15.
A: Atomic radius of lithium is larger than that of fluorine.
R: Lithium has fewer protons than fluorine.
Answer: 1 – Both true, R correctly explains A.
16.
A: Lanthanides are called inner transition elements.
R: Their valence electrons are in 4f-orbitals.
Answer: 1 – Both true, R correctly explains A.
17.
A: Alkali metals form basic oxides.
R: They have low electronegativity.
Answer: 1 – Both true, R correctly explains A.
18.
A: Chlorine has a higher electron affinity than oxygen.
R: Chlorine is in Group 17 while oxygen is in Group 16.
Answer: 1 – Both true, R correctly explains A.
19.
A: Atomic size decreases from K to Cl in Period 4.
R: Nuclear charge increases and shields outer electrons effectively.
Answer: 3 – A is true, but R is partially incorrect (Shielding is almost constant; nuclear attraction dominates).
20.
A: Non-metals are located on the right side of the periodic table.
R: Non-metals have high ionization energy and electronegativity.
Answer: 1 – Both true, R correctly explains A.
Cased Based question 💫
Case 1:
A student observes the atomic radii of elements in Period 3: Na (186 pm), Mg (160 pm), Al (143 pm), Si (118 pm), P (110 pm), S (104 pm), Cl (99 pm), Ar (71 pm).
Q1: Which element has the smallest atomic radius?
Answer: Argon (Ar) – 71 pm
Q2: Explain why atomic radius decreases across the period.
Answer: Nuclear charge increases across the period, pulling electrons closer to the nucleus, reducing atomic size.
Q3: Predict the metallic character trend across the period.
Answer: Metallic character decreases from Na → Ar.
Case 2:
The ionization energies (kJ/mol) of elements in Period 2 are: Li (520), Be (900), B (800), C (1086), N (1402), O (1314), F (1681), Ne (2080).
Q4: Why is there a decrease in ionization energy from Be → B?
Answer: B has its outer electron in the p-orbital, which is farther from the nucleus and slightly shielded, making it easier to remove.
Q5: Which element has the highest ionization energy?
Answer: Neon (Ne) – 2080 kJ/mol
Case 3:
The electronegativity of halogens (Pauling scale) is: F (3.98), Cl (3.16), Br (2.96), I (2.66).
Q6: Which halogen is the most electronegative?
Answer: Fluorine (F)
Q7: Explain the trend of electronegativity down the group.
Answer: Electronegativity decreases down the group because atomic size increases, reducing the attraction for bonding electrons.
Case 4:
Elements X, Y, Z are in the same group. Their atomic radii are: X (52 pm), Y (110 pm), Z (152 pm).
Q8: Arrange X, Y, Z in order of increasing metallic character.
Answer: X < Y < Z
Q9: Which element is likely at the top of the group?
Answer: X – smallest atomic radius, least metallic.
Case 5:
Consider elements with electron configurations:
- A: 1s² 2s² 2p⁶ 3s¹
- B: 1s² 2s² 2p⁶ 3s²
- C: 1s² 2s² 2p⁶ 3s² 3p¹
Q10: Identify which element is an alkali metal.
Answer: A (3s¹ → Na)
Q11: Identify the p-block element.
Answer: C (3s²3p¹ → Al)
Case 6:
A chemistry teacher gives the following data: Electron affinity (kJ/mol) – Cl (−349), S (−200), P (−72).
Q12: Which element has the highest tendency to gain an electron?
Answer: Chlorine (Cl)
Q13: Explain why phosphorus has the lowest electron affinity.
Answer: Phosphorus has a half-filled p-orbital, which is more stable and less likely to gain an electron.
Case 7:
Elements Q, R, S show the following ionization energy trend: Q < R < S.
Q14: Which element is likely to be a metal?
Answer: Q – lower ionization energy, easier to lose electrons.
Q15: Which element is likely to be a noble gas?
Answer: S – very high ionization energy.
Case 8:
A student observes that lithium reacts vigorously with water while cesium reacts explosively.
Q16: Explain the trend in reactivity of alkali metals down the group.
Answer: Reactivity increases down the group because ionization energy decreases; outer electrons are easily lost.
Q17: Which alkali metal is least reactive?
Answer: Lithium (Li)
Case 9:
Consider these elements: Be, Mg, Ca. Their metallic oxides react with water as follows:
- BeO: No reaction
- MgO: Slight reaction
- CaO: Vigorous reaction
Q18: Explain the trend in basicity of oxides down the group.
Answer: Basicity increases down the group as metallic character increases.
Q19: Which oxide is amphoteric?
Answer: BeO – reacts with both acids and bases.
Case 10:
The halogens F₂, Cl₂, Br₂, I₂ displace each other from compounds as follows: Cl₂ displaces Br⁻, but Br₂ cannot displace Cl⁻.
Q20: Explain this observation.
Answer: Reactivity of halogens decreases down the group; a more reactive halogen can displace a less reactive halogen from its compounds.
Sample Paper 📜
Total Marks: 70
Time: 3 Hours
Section A – Very Short Answer Questions (1 mark each)
Q1. Define periodic law.
Q2. Which element has the highest electronegativity in Period 2?
Q3. Give the group number of halogens.
Q4. State the trend in metallic character across a period.
Q5. What is the electron configuration of Aluminum (Al)?
Q6. Name the block to which calcium belongs.
Q7. Which element has the smallest atomic radius in Period 3?
Q8. Define electron affinity.
Q9. Which element is an s-block element with one valence electron?
Q10. State the trend of ionization energy down a group.
Section B – Short Answer Questions (2–3 marks each)
Q11. Compare Mendeleev’s Periodic Table with Modern Periodic Table.
Q12. Explain why ionization energy increases across a period.
Q13. Arrange Li, Na, K, and Rb in order of increasing metallic character.
Q14. Explain why fluorine is the most electronegative element.
Q15. Give the general trends of atomic radius down a group.
Q16. Identify the following elements from their electronic configurations:
- (i) 1s² 2s² 2p⁶ 3s² 3p⁵
- (ii) 1s² 2s² 2p⁶ 3s¹
Q17. Why are noble gases chemically inert?
Q18. Explain why oxygen has higher electron affinity than nitrogen.
Q19. Differentiate between metallic and non-metallic character trends.
Q20. Which element in Period 3 is likely to form an amphoteric oxide?
Section C – Long Answer Questions (5 marks each)
Q21. Explain s-block and p-block elements with examples and properties.
Q22. Describe the periodic trends in atomic radius, ionization energy, and electronegativity across a period.
Q23. Discuss the significance of periodicity in chemical properties.
Q24. Explain the variation in metallic character and non-metallic character in the periodic table.
Q25. A student is given three elements A, B, and C with increasing atomic number. A is a metal, B is a metalloid, and C is a non-metal. Explain the trends in their properties.
Section D – Case-Based Questions (4 marks each)
Q26. Period 3 elements have atomic radii: Na (186 pm), Mg (160 pm), Al (143 pm), Si (118 pm), P (110 pm), S (104 pm), Cl (99 pm), Ar (71 pm).
- (i) Identify the element with smallest atomic radius.
- (ii) Explain the trend in atomic radius across the period.
- (iii) Predict which element is most metallic.
Q27. The ionization energies of elements in Period 2 are: Li (520), Be (900), B (800), C (1086), N (1402), O (1314), F (1681), Ne (2080) kJ/mol.
- (i) Identify the element with the highest ionization energy.
- (ii) Explain the decrease in ionization energy from Be → B.
- (iii) Identify which elements are most likely metals.
Q28. Elements X, Y, Z are in the same group. Atomic radii: X (52 pm), Y (110 pm), Z (152 pm).
- (i) Arrange X, Y, Z in order of increasing metallic character.
- (ii) Which element is at the top of the group?
- (iii) Explain why metallic character increases down the group.
Q29. Elements A, B, C have the following electron configurations:
- A: 1s² 2s² 2p⁶ 3s¹
- B: 1s² 2s² 2p⁶ 3s² 3p¹
- C: 1s² 2s² 2p⁶ 3s² 3p⁶
- (i) Identify the s-block, p-block, and noble gas elements.
- (ii) Predict which element has the highest electronegativity.
- (iii) Explain trends in electron affinity for these elements.
Q30. Halogens F, Cl, Br, I react with halide salts. Cl₂ displaces Br⁻, but Br₂ cannot displace Cl⁻.
- (i) Explain the reactivity trend among halogens.
- (ii) Which halogen is most reactive?
- (iii) Why does iodine not displace chlorine?
Solutions – Sample Paper: Classification of Elements and Periodicity in Properties
Section A – Very Short Answer Questions (1 mark each)
Q1. Define periodic law.
Answer: Properties of elements are a periodic function of their atomic number.
Q2. Which element has the highest electronegativity in Period 2?
Answer: Fluorine (F) – 3.98 (Pauling scale)
Q3. Give the group number of halogens.
Answer: Group 17
Q4. State the trend in metallic character across a period.
Answer: Metallic character decreases from left to right across a period.
Q5. What is the electron configuration of Aluminum (Al)?
Answer: 1s² 2s² 2p⁶ 3s² 3p¹
Q6. Name the block to which calcium belongs.
Answer: s-block
Q7. Which element has the smallest atomic radius in Period 3?
Answer: Argon (Ar) – 71 pm
Q8. Define electron affinity.
Answer: Energy released when an electron is added to a neutral atom in gaseous state.
Q9. Which element is an s-block element with one valence electron?
Answer: Sodium (Na)
Q10. State the trend of ionization energy down a group.
Answer: Ionization energy decreases down a group due to increased atomic size and shielding effect.
Section B – Short Answer Questions (2–3 marks each)
Q11. Compare Mendeleev’s Periodic Table with Modern Periodic Table.
| Feature | Mendeleev’s Table | Modern Periodic Table |
|---|---|---|
| Basis | Atomic Mass | Atomic Number |
| Periodic Law | Properties repeat after atomic mass | Properties repeat after atomic number |
| Groups | 8 vertical groups | 18 vertical groups |
| Gaps | Left for undiscovered elements | No gaps |
| Hydrogen | Ambiguous | Placed based on electronic configuration |
Q12. Explain why ionization energy increases across a period.
Answer: Across a period, nuclear charge increases, pulling electrons closer. The outer electrons are held tightly, requiring more energy to remove → ionization energy increases.
Q13. Arrange Li, Na, K, Rb in increasing metallic character.
Answer: Li < Na < K < Rb
(Metallic character increases down the group as ionization energy decreases.)
Q14. Explain why fluorine is the most electronegative element.
Answer: Fluorine has small size and high nuclear charge, which strongly attracts bonding electrons.
Q15. Give the general trends of atomic radius down a group.
Answer: Atomic radius increases down a group due to addition of electron shells.
Q16. Identify elements from their electronic configurations:
- (i) 1s² 2s² 2p⁶ 3s² 3p⁵ → Chlorine (Cl), p-block
- (ii) 1s² 2s² 2p⁶ 3s¹ → Sodium (Na), s-block
Q17. Why are noble gases chemically inert?
Answer: Noble gases have complete valence shells, making them stable and non-reactive.
Q18. Explain why oxygen has higher electron affinity than nitrogen.
Answer: Nitrogen has a half-filled p-orbital, which is stable; oxygen gains an electron more easily → higher electron affinity.
Q19. Differentiate between metallic and non-metallic character trends.
| Property | Trend Across Period | Trend Down Group |
|---|---|---|
| Metallic character | Decreases | Increases |
| Non-metallic character | Increases | Decreases |
Q20. Which element in Period 3 is likely to form an amphoteric oxide?
Answer: Aluminum (Al) – reacts with both acids and bases.
Section C – Long Answer Questions (5 marks each)
Q21. Explain s-block and p-block elements with examples and properties.
- s-block: Groups 1–2; valence electrons in s-orbital
- Examples: Li, Na, Be, Mg
- Properties: Highly reactive, form basic oxides, metallic
- p-block: Groups 13–18; valence electrons in p-orbital
- Examples: B, C, N, O
- Properties: Includes metals, metalloids, non-metals; form acidic oxides
Q22. Periodic trends in atomic radius, ionization energy, electronegativity across a period:
- Atomic radius: Decreases → due to increased nuclear charge
- Ionization energy: Increases → electrons held more tightly
- Electronegativity: Increases → stronger pull on bonding electrons
Q23. Significance of periodicity in chemical properties:
- Predicts chemical reactivity
- Helps in determining bonding and compound formation
- Explains physical properties (melting/boiling point, density)
- Forms foundation for inorganic chemistry
Q24. Variation in metallic and non-metallic character:
- Metallic: Decreases across a period, increases down a group
- Non-metallic: Increases across a period, decreases down a group
- Reason: Related to ionization energy and electron affinity trends
Q25. Elements A (metal), B (metalloid), C (non-metal) – trends:
| Property | A (Metal) | B (Metalloid) | C (Non-metal) |
|---|---|---|---|
| Reactivity | Forms cations | Amphoteric | Forms anions |
| Electronegativity | Low | Moderate | High |
| Metallic character | High | Moderate | Low |
Section D – Case-Based Questions (4 marks each)
Q26. Period 3 atomic radii:
- Smallest radius → Ar
- Trend: Decreases Na → Ar due to increasing nuclear charge
- Most metallic → Na
Q27. Period 2 ionization energy:
- Highest IE → Ne
- Be → B decrease → B’s p-electron is easier to remove (shielded)
- Metals → Li, Be
Q28. Elements X, Y, Z (atomic radii 52, 110, 152 pm):
- Increasing metallic character → X < Y < Z
- Top of group → X (smallest radius, least metallic)
- Metallic character increases down group due to easier loss of outer electrons
Q29. Electron configurations:
- s-block → A (3s¹)
- p-block → B (3s²3p¹)
- Noble gas → C (3s²3p⁶)
- Highest electronegativity → B (p-block element)
- Electron affinity trend: increases across period (A < B < C)
Q30. Halogens reactivity:
- Reactivity decreases down the group (F > Cl > Br > I)
- Most reactive → Fluorine (F)
- Iodine cannot displace chlorine → less reactive
Additional Sample paper 📜
Total Marks: 70
Time: 3 Hours
Section A – Very Short Answer Questions (1 mark each)
Q1. What is the main basis of the Modern Periodic Table?
Q2. Which element in Period 3 has the highest first ionization energy?
Q3. Name the group that contains alkaline earth metals.
Q4. Define electronegativity in simple terms.
Q5. Give the electronic configuration of sulfur (S).
Q6. State the trend of atomic radius down a group.
Q7. Which noble gas has the smallest atomic radius?
Q8. What is metallic character?
Q9. Identify an element with one electron in its outermost shell in Period 2.
Q10. What is the general trend of electron affinity across a period?
Section B – Short Answer Questions (2–3 marks each)
Q11. Explain why ionization energy of Group 1 elements is low.
Q12. Arrange Be, Mg, Ca, Sr in order of increasing atomic radius.
Q13. Define s-block, p-block, d-block, and f-block elements with examples.
Q14. Explain why noble gases are chemically inert.
Q15. Compare metallic and non-metallic character trends across a period.
Q16. Why does oxygen have a higher electronegativity than sulfur?
Q17. Identify the following elements:
- (i) 1s² 2s² 2p⁶ 3s² 3p²
- (ii) 1s² 2s² 2p⁶ 3s² 3p⁵
Q18. Explain the trend of ionization energy down a group.
Q19. Which oxide among Na₂O, Al₂O₃, and SiO₂ is amphoteric?
Q20. Why does lithium have a smaller size than sodium?
Section C – Long Answer Questions (5 marks each)
Q21. Discuss the significance of periodicity in chemical properties.
Q22. Explain the trends in atomic radius, ionization energy, and electronegativity across a period and down a group.
Q23. Describe s-block and p-block elements with their properties and examples.
Q24. Explain how metallic and non-metallic character changes across a period and down a group.
Q25. The elements Li, Be, B, C, N show a variation in properties. Explain the trends in atomic radius, ionization energy, and electronegativity in this series.
Section D – Case-Based Questions (4 marks each)
Q26. Period 3 elements have ionization energies (kJ/mol) as follows: Na (495), Mg (737), Al (577), Si (786), P (1012), S (999), Cl (1251), Ar (1521).
- (i) Which element has the highest ionization energy?
- (ii) Explain why there is a decrease from Mg → Al.
- (iii) Identify the metals in this period.
Q27. The halogens F, Cl, Br, I show decreasing reactivity down the group.
- (i) Which halogen is most reactive?
- (ii) Explain the reactivity trend.
- (iii) Predict which halogen can displace bromide from KBr solution.
Q28. Elements X, Y, Z are in the same period. Their atomic radii are X (186 pm), Y (160 pm), Z (143 pm).
- (i) Arrange X, Y, Z in order of decreasing metallic character.
- (ii) Which element is likely to be a non-metal?
- (iii) Explain why metallic character decreases across the period.
Q29. Elements A, B, C have electron configurations:
- A: 1s² 2s² 2p⁶ 3s¹
- B: 1s² 2s² 2p⁶ 3s² 3p³
- C: 1s² 2s² 2p⁶ 3s² 3p⁶
- (i) Identify s-block, p-block, and noble gas elements.
- (ii) Predict the element most likely to form cations.
- (iii) Explain which element is chemically inert.
Q30. Consider the elements K, Ca, and Cl.
- (i) Identify the metal, metalloid, and non-metal.
- (ii) Explain the trend in electronegativity among these elements.
- (iii) Which element will have the largest atomic radius?
Solution 💫
Section A – Very Short Answer Questions (1 mark each)
Q1. Main basis of Modern Periodic Table: Atomic number.
Q2. Element in Period 3 with highest first ionization energy: Argon (Ar) – 1521 kJ/mol.
Q3. Group that contains alkaline earth metals: Group 2.
Q4. Electronegativity: Ability of an atom to attract shared electrons in a bond.
Q5. Electronic configuration of sulfur (S): 1s² 2s² 2p⁶ 3s² 3p⁴
Q6. Trend of atomic radius down a group: Increases down the group due to additional electron shells.
Q7. Noble gas with smallest atomic radius: Helium (He)
Q8. Metallic character: Tendency of an element to lose electrons and form cations.
Q9. Element with one valence electron in Period 2: Lithium (Li)
Q10. General trend of electron affinity across a period: Increases across a period due to smaller atomic size and higher nuclear charge.
Section B – Short Answer Questions (2–3 marks each)
Q11. Ionization energy of Group 1 elements is low because they have a single valence electron which is loosely held, making it easy to remove.
Q12. Arrange Be, Mg, Ca, Sr in increasing atomic radius: Be < Mg < Ca < Sr
Q13. Blocks of elements:
| Block | Valence Electrons | Example |
|---|---|---|
| s-block | s¹ or s² | Li, Na, Mg |
| p-block | p¹–p⁶ | B, C, N |
| d-block | d¹–d¹⁰ | Fe, Cu |
| f-block | f¹–f¹⁴ | La, Ce |
Q14. Noble gases are inert because they have complete valence shells → stable electronic configuration.
Q15. Metallic and non-metallic character trends:
| Trend | Across Period | Down Group |
|---|---|---|
| Metallic | Decreases | Increases |
| Non-metallic | Increases | Decreases |
Q16. Oxygen has higher electronegativity than sulfur because O has smaller size, so the nucleus attracts bonding electrons more strongly.
Q17. Identify elements:
- (i) 1s² 2s² 2p⁶ 3s² 3p² → Carbon (C), p-block
- (ii) 1s² 2s² 2p⁶ 3s² 3p⁵ → Chlorine (Cl), p-block
Q18. Ionization energy trend down a group: Decreases because atomic size increases → valence electrons are farther from nucleus and more shielded.
Q19. Amphoteric oxide among Na₂O, Al₂O₃, SiO₂: Al₂O₃
Q20. Lithium has smaller size than sodium because Li has fewer electron shells and higher nuclear attraction on its electrons.
Section C – Long Answer Questions (5 marks each)
Q21. Significance of periodicity in chemical properties:
- Helps predict reactivity of elements
- Explains bonding and compound formation
- Helps understand physical properties like melting/boiling points
- Foundation for inorganic chemistry and chemical trends
Q22. Trends in periodic table:
- Atomic radius: Decreases across a period, increases down a group
- Ionization energy: Increases across a period, decreases down a group
- Electronegativity: Increases across a period, decreases down a group
Q23. s-block and p-block elements:
- s-block: Groups 1–2, metallic, form basic oxides, valence in s-orbital
- Examples: Li, Na, Mg
- p-block: Groups 13–18, includes metals/non-metals/metalloids, form acidic oxides
- Examples: B, C, N, O
Q24. Metallic and non-metallic character trends:
- Metallic: Decreases across a period, increases down a group
- Non-metallic: Increases across a period, decreases down a group
- Reason: Related to ionization energy, electron affinity, and atomic size
Q25. Elements Li, Be, B, C, N – trends:
| Property | Li | Be | B | C | N |
|---|---|---|---|---|---|
| Atomic radius | Largest | ↓ | ↓ | ↓ | Smallest |
| Ionization energy | Low | ↑ | ↑ | ↑ | Highest |
| Electronegativity | Low | ↑ | ↑ | ↑ | High |
Section D – Case-Based Questions (4 marks each)
Q26. Period 3 ionization energies:
- Highest IE → Ar
- Decrease Mg → Al → outer electron in Al’s p-orbital is easier to remove
- Metals in period → Na, Mg, Al
Q27. Halogens F, Cl, Br, I:
- Most reactive → Fluorine (F)
- Reactivity decreases down group due to increasing atomic size
- Chlorine can displace bromide from KBr solution
Q28. Elements X (186 pm), Y (160 pm), Z (143 pm):
- Decreasing metallic character → X > Y > Z
- Non-metal → Z (smallest radius)
- Metallic character decreases across period due to increasing nuclear charge
Q29. Electron configurations:
- s-block → A (3s¹)
- p-block → B (3s²3p³)
- Noble gas → C (3s²3p⁶)
- Forms cations → A
- Chemically inert → C
Q30. Elements K, Ca, Cl:
- Metal → K, Metalloid → none, Non-metal → Cl
- Electronegativity trend → Cl > Ca > K
- Largest atomic radius → K
20 Multiple Choice Questions – Classification of Elements and Periodicity in Properties
Q1. The periodic table is arranged on the basis of:
A) Atomic mass
B) Atomic number
C) Valence electrons
D) Electronegativity
Answer: B) Atomic number
Q2. Which element has the smallest atomic radius in Period 2?
A) Li
B) Be
C) F
D) Ne
Answer: D) Ne
Q3. The element with configuration 1s² 2s² 2p⁶ 3s² 3p³ belongs to:
A) s-block
B) p-block
C) d-block
D) f-block
Answer: B) p-block
Q4. The ionization energy trend across a period:
A) Decreases
B) Increases
C) Remains constant
D) Random
Answer: B) Increases
Q5. Which element is an alkali metal?
A) Be
B) Mg
C) Na
D) Al
Answer: C) Na
Q6. Which of the following elements is most electronegative?
A) Li
B) F
C) O
D) Cl
Answer: B) F
Q7. The oxide that is amphoteric:
A) Na₂O
B) Al₂O₃
C) SiO₂
D) MgO
Answer: B) Al₂O₃
Q8. Metallic character in a period:
A) Increases left to right
B) Decreases left to right
C) Remains constant
D) Increases down the period
Answer: B) Decreases left to right
Q9. Which group contains alkaline earth metals?
A) Group 1
B) Group 2
C) Group 17
D) Group 18
Answer: B) Group 2
Q10. The element with the configuration 1s² 2s² 2p⁶ 3s² 3p⁶ is:
A) Na
B) Mg
C) Ar
D) Cl
Answer: C) Ar
Q11. The trend in electron affinity across a period:
A) Decreases
B) Increases
C) Remains constant
D) Random
Answer: B) Increases
Q12. Which of the following elements has the lowest ionization energy?
A) Li
B) Be
C) Na
D) F
Answer: C) Na
Q13. The halogen that is least reactive:
A) F
B) Cl
C) Br
D) I
Answer: D) I
Q14. Atomic radius down a group:
A) Decreases
B) Increases
C) Remains constant
D) Random
Answer: B) Increases
Q15. Which element is most likely to form cations?
A) Na
B) Cl
C) O
D) Ne
Answer: A) Na
Q16. Which element is in the d-block?
A) Mg
B) Fe
C) Cl
D) Ne
Answer: B) Fe
Q17. Which of the following is a noble gas?
A) Ne
B) O
C) Cl
D) Na
Answer: A) Ne
Q18. The element in Period 3 that is a metalloid:
A) Na
B) Al
C) Si
D) Cl
Answer: C) Si
Q19. Reactivity of alkali metals down the group:
A) Increases
B) Decreases
C) Remains same
D) Random
Answer: A) Increases
Q20. Electron affinity of halogens:
A) F < Cl < Br < I
B) F > Cl > Br > I
C) Cl > F > Br > I
D) I > Br > Cl > F
Answer: B) F > Cl > Br > I






Leave a Reply