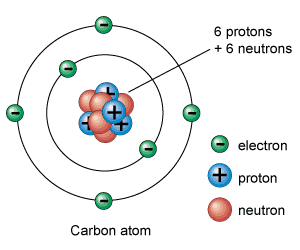
Structure of an Atom Notes, Summary, MCQs & Important Questions
Meta Description:
Complete NCERT Class 9 Chemistry Structure of an Atom notes, summary, MCQs, keywords, and exam questions for quick revision and exam success.
Introduction of the Chapter
The chapter Structure of an Atom Class 9 explains the internal composition of atoms, which are the building blocks of matter. Early scientists believed atoms were indivisible, but later discoveries revealed the presence of subatomic particles such as electrons, protons, and neutrons.
Understanding the Structure of an Atom helps students explain chemical reactions, atomic number, mass number, isotopes, and electronic configuration. This chapter forms the foundation for advanced chemistry topics and is essential for school exams and competitive tests.
Short Notes (Quick Revision Points)
- Atom is the smallest unit of an element that retains its properties.
- Subatomic particles: electrons (–), protons (+), neutrons (neutral).
- Electrons revolve around the nucleus in shells.
- Nucleus contains protons and neutrons.
- Atomic number = number of protons.
- Mass number = protons + neutrons.
- Isotopes: same atomic number, different mass number.
- Isobars: same mass number, different atomic numbers.
- Valency depends on electrons in the outermost shell.
- Bohr’s model explains electron distribution.
Detailed Summary of Structure of an Atom (1200 Words)
The chapter Structure of an Atom Class 9 describes the discovery of subatomic particles and models explaining atomic structure. Early scientists like Dalton proposed that atoms were indivisible particles. However, later experiments proved atoms are made up of smaller particles.
Discovery of Electrons
In 1897, J.J. Thomson discovered electrons through cathode ray experiments. He concluded that atoms contain negatively charged particles called electrons. He proposed the “plum pudding model,” where electrons were embedded in a positively charged sphere.
Discovery of Protons
Eugen Goldstein conducted canal ray experiments and discovered positively charged particles called protons. Protons are located inside the nucleus and determine the atomic number of an element.
Discovery of the Nucleus
Ernest Rutherford conducted the gold foil experiment. He bombarded alpha particles on thin gold foil and observed:
- Most particles passed straight through.
- Some deflected slightly.
- Few bounced back.
He concluded that:
- Most of the atom is empty space.
- Positive charge is concentrated in a tiny central nucleus.
This experiment led to the nuclear model of the atom.
Bohr’s Model of Atom
Niels Bohr improved Rutherford’s model by explaining electron arrangement. According to Bohr:
- Electrons revolve in fixed circular orbits called shells.
- Energy levels are represented as K, L, M, N shells.
- Maximum electrons in a shell = 2n² (where n = shell number).
- Electrons do not lose energy while revolving.
Distribution of Electrons in Shells
- K shell holds 2 electrons.
- L shell holds 8 electrons.
- M shell holds 18 electrons.
Example:
- Oxygen (8 electrons): 2, 6
- Sodium (11 electrons): 2, 8, 1
Valency
Valency is the combining capacity of an atom.
- If outer shell has 1–4 electrons → valency = number of electrons lost.
- If outer shell has 5–7 electrons → valency = electrons gained to complete octet.
Example:
- Sodium (2,8,1) → valency = 1
- Oxygen (2,6) → valency = 2
Atomic Number and Mass Number
- Atomic number (Z) = number of protons = number of electrons
- Mass number (A) = protons + neutrons
Example:
Carbon:
Z = 6, A = 12
Neutrons = 12 – 6 = 6
Isotopes
Atoms of the same element with same atomic number but different mass numbers.
Examples:
- Hydrogen: Protium, Deuterium, Tritium
- Carbon-12, Carbon-14
Uses:
- Uranium isotopes used in nuclear reactors.
- Carbon isotopes used in dating fossils.
Isobars
Atoms with same mass number but different atomic numbers.
Example:
- Calcium (40) and Argon (40)
Importance of Structure of an Atom
Understanding the Structure of an Atom Class 9 helps explain chemical bonding, periodic properties, and formation of compounds. It is a foundational concept in chemistry and physics.
Flowchart / Mind Map (Text-Based)
Structure of an Atom
→ Discovery of subatomic particles
→ Electron (Thomson)
→ Proton (Goldstein)
→ Nucleus (Rutherford)
→ Atomic Models
→ Thomson Model
→ Rutherford Model
→ Bohr Model
→ Subatomic Particles
→ Electron
→ Proton
→ Neutron
→ Atomic Concepts
→ Atomic number
→ Mass number
→ Valency
→ Special Atoms
→ Isotopes
→ Isobars
Important Keywords with Meanings
Atom – smallest unit of matter
Electron – negatively charged particle
Proton – positively charged particle
Neutron – neutral particle
Nucleus – dense central core of atom
Atomic Number – number of protons
Mass Number – protons + neutrons
Valency – combining capacity
Shells – energy levels around nucleus
Isotopes – same atomic number, different mass
Isobars – same mass, different atomic numbers
Electronic Configuration – arrangement of electrons
Important Questions & Answers
Very Short Answer Questions
- Who discovered electrons?
J.J. Thomson - What is atomic number?
Number of protons. - Define mass number.
Sum of protons and neutrons. - What are isotopes?
Atoms with same atomic number but different mass numbers. - Name the shells of an atom.
K, L, M, N. - What is valency?
Combining capacity of an atom. - Who discovered the nucleus?
Ernest Rutherford. - Maximum electrons in K shell?
- Define isobars.
Atoms with same mass number but different atomic numbers. - Charge of electron?
Negative.
Short Answer Questions
- Explain Rutherford’s gold foil experiment.
Answer: Rutherford bombarded gold foil with alpha particles. Most passed through, some deflected, and few bounced back. He concluded atoms have a small dense nucleus and mostly empty space. - Describe Bohr’s model.
Answer: Electrons revolve in fixed shells with definite energy. Maximum electrons = 2n². Shells are K, L, M, N. - Define isotopes with examples and uses.
(Provide explanation and examples) - What is electronic configuration? Explain with examples.
(Explain electron distribution) - Explain atomic number and mass number with examples.
- Write differences between isotopes and isobars.
- Describe subatomic particles.
- Explain valency with examples.
- Why is the atom electrically neutral?
- Draw and explain sodium atom configuration.
MCQs with Answers
- Electron was discovered by:
A. Rutherford
B. Thomson
C. Bohr
D. Dalton
Answer: B - Proton charge is:
A. Positive
B. Negative
C. Neutral
D. Variable
Answer: A - Atomic number equals:
A. Neutrons
B. Protons
C. Mass
D. Valency
Answer: B - Maximum electrons in L shell:
A. 2
B. 8
C. 18
D. 32
Answer: B - Nucleus contains:
A. Electrons
B. Protons & neutrons
C. Only protons
D. Only neutrons
Answer: B - Isotopes differ in:
A. Protons
B. Electrons
C. Mass number
D. Atomic number
Answer: C - Shell formula:
A. 2n
B. n²
C. 2n²
D. n³
Answer: C - Sodium valency:
A. 1
B. 2
C. 3
D. 8
Answer: A - Neutron charge:
A. Positive
B. Neutral
C. Negative
D. Variable
Answer: B - Rutherford used:
A. Copper foil
B. Gold foil
C. Silver foil
D. Iron foil
Answer: B
Exam Tips & Value-Based Questions
Exam Tips
- Remember 2n² rule for shell capacity.
- Atomic number = protons = electrons.
- Practice electronic configuration.
- Learn differences: isotopes vs isobars.
- Revise Rutherford and Bohr models carefully.
Value-Based Questions
- Why should scientists study atomic structure?
Answer: To develop technology and understand matter. - How do isotopes help in medicine?
Answer: Used in cancer treatment and diagnosis. - Why is nuclear energy both useful and dangerous?
Answer: Produces energy but may cause radiation hazards. - What value do scientific discoveries teach us?
Answer: Curiosity and critical thinking. - Why must radioactive materials be handled carefully?
Answer: To prevent harm to living organisms.
Conclusion
The chapter Structure of an Atom Class 9 is one of the most important foundations in chemistry. It explains how matter is built from tiny particles and how scientific discoveries helped shape our modern understanding of atomic structure. From Dalton’s atomic theory to Thomson’s discovery of electrons and Rutherford’s nuclear model, each experiment contributed to uncovering the true nature of atoms.
The discovery of subatomic particles revolutionized science. Electrons, protons, and neutrons together form atoms, and their arrangement determines the chemical behavior of elements. The Structure of an Atom explains why atoms are electrically neutral, how atomic number identifies an element, and how mass number represents the total particles in the nucleus.
Rutherford’s gold foil experiment remains one of the most significant scientific experiments. It showed that most of the atom is empty space and that a dense nucleus exists at the center. Later, Bohr improved this model by explaining electron distribution in fixed energy levels. Bohr’s model successfully described electronic configuration, which is essential for understanding valency and chemical bonding.
Electronic configuration is crucial because it determines how atoms react with each other. Atoms tend to achieve stability by completing their outermost shell, known as the octet rule. This explains why elements form compounds and molecules. For example, sodium loses one electron while chlorine gains one electron, forming a stable compound.
The chapter also introduces isotopes and isobars, which are important in both science and practical applications. Isotopes of hydrogen and carbon have uses in medicine, archaeology, and nuclear energy. Carbon dating helps determine the age of fossils, while radioactive isotopes are used in medical treatments and industrial applications.
Understanding the Structure of an Atom Class 9 also builds a foundation for future topics such as chemical bonding, periodic classification, and nuclear chemistry. Without knowledge of atomic structure, it would be impossible to understand reactions, molecules, and material properties.
This chapter is highly important for school examinations as well as competitive exams. Students should focus on atomic models, electronic configuration, valency, isotopes, and numerical concepts like atomic number and mass number. Practicing diagrams and configurations helps in scoring better marks.
From a practical perspective, atomic structure knowledge has led to innovations in medicine, energy production, electronics, and materials science. Modern technology, including semiconductors, nuclear power plants, and diagnostic imaging tools, relies on atomic science.
In conclusion, the Structure of an Atom provides a deep understanding of matter and its behavior. It highlights the importance of scientific curiosity and experimentation in shaping knowledge. Mastering this chapter strengthens conceptual clarity and prepares students for advanced chemistry learning.
By revising notes, practicing MCQs, and understanding core concepts, students can easily excel in this chapter. The Structure of an Atom Class 9 remains a cornerstone of chemistry and a gateway to understanding the microscopic world that governs everything around us.
10 Long Answer Questions – Structure of an Atom (Class 9)
1. Explain Rutherford’s Gold Foil Experiment and its conclusions.
Answer:
Ernest Rutherford conducted the gold foil experiment in 1911 to study the structure of the atom. In this experiment, fast-moving alpha particles (positively charged) were directed at a very thin sheet of gold foil. A fluorescent screen surrounded the foil to detect the scattered particles.
Observations:
- Most alpha particles passed straight through the foil.
- Some particles deflected at small angles.
- Very few particles bounced back.
Conclusions:
- Most of the atom is empty space.
- Positive charge and mass are concentrated in a tiny central nucleus.
- Electrons revolve around the nucleus.
This experiment disproved Thomson’s plum pudding model and led to the nuclear model of the atom.
2. Describe Bohr’s Model of the Atom and its postulates.
Answer:
Niels Bohr improved Rutherford’s model by explaining electron arrangement.
Postulates of Bohr’s Model:
- Electrons revolve around the nucleus in fixed circular paths called shells or energy levels.
- Each shell has definite energy.
- Electrons do not lose energy while moving in these shells.
- Energy is emitted or absorbed when electrons jump between shells.
- Maximum electrons in a shell = 2n², where n is shell number.
Shell capacity:
- K shell = 2
- L shell = 8
- M shell = 18
Bohr’s model explained atomic stability and electronic configuration.
3. Describe the discovery and properties of subatomic particles.
Answer:
Atoms consist of three main subatomic particles:
Electron
- Discovered by J.J. Thomson.
- Charge: negative (–1)
- Mass: negligible
- Located outside nucleus.
Proton
- Discovered by Eugen Goldstein.
- Charge: positive (+1)
- Located in nucleus.
Neutron
- Discovered by James Chadwick.
- Charge: neutral
- Present in nucleus.
Importance: Protons determine atomic number, electrons control chemical behavior, and neutrons add mass and stability.
4. Explain atomic number and mass number with examples.
Answer:
Atomic Number (Z):
Number of protons present in the nucleus. It also equals the number of electrons in a neutral atom.
Example: Carbon has 6 protons → Atomic number = 6.
Mass Number (A):
Total number of protons and neutrons.
Mass Number = Protons + Neutrons
Example: Carbon-12
Protons = 6
Neutrons = 6
Mass number = 12
Importance:
Atomic number identifies an element, while mass number gives its atomic mass.
5. What is electronic configuration? Explain with examples.
Answer:
Electronic configuration is the arrangement of electrons in different shells around the nucleus.
Rules:
- Maximum electrons in shell = 2n²
- Outer shell cannot have more than 8 electrons.
Examples:
Oxygen (8): 2, 6
Sodium (11): 2, 8, 1
Magnesium (12): 2, 8, 2
Importance:
It determines valency and chemical reactivity.
6. Define valency and explain how it is determined.
Answer:
Valency is the combining capacity of an atom, determined by the number of electrons in the outermost shell.
Rules:
- 1–4 electrons → valency = electrons lost.
- 5–7 electrons → valency = electrons gained to complete octet.
- 8 electrons → valency = 0.
Examples:
Sodium (2,8,1) → valency = 1
Oxygen (2,6) → valency = 2
Neon (2,8) → valency = 0
Valency explains how atoms form compounds.
7. What are isotopes? Explain their properties and uses.
Answer:
Isotopes are atoms of the same element having the same atomic number but different mass numbers.
Examples:
Hydrogen:
- Protium (¹H)
- Deuterium (²H)
- Tritium (³H)
Carbon:
- Carbon-12
- Carbon-14
Properties:
- Same chemical properties.
- Different physical properties due to mass difference.
Uses:
- Carbon-14 used in dating fossils.
- Uranium isotopes used in nuclear reactors.
- Radioisotopes used in cancer treatment.
8. What are isobars? Give examples and explain their characteristics.
Answer:
Isobars are atoms of different elements having the same mass number but different atomic numbers.
Examples:
- Argon (18 protons) and Calcium (20 protons) both have mass number 40.
Characteristics:
- Different chemical properties.
- Same mass number.
- Different atomic numbers.
They help in understanding atomic structure differences.
9. Why is the atom electrically neutral? Explain.
Answer:
An atom is electrically neutral because the number of protons equals the number of electrons.
- Protons carry positive charge.
- Electrons carry negative charge.
- Neutrons have no charge.
Example:
Sodium atom:
- Protons = 11 (+11 charge)
- Electrons = 11 (–11 charge)
Net charge = 0
Thus, the atom remains neutral.
10. Explain the importance of studying the structure of an atom.
Answer:
Studying the structure of an atom is essential because it helps us understand the composition and behavior of matter.
Importance:
- Explains chemical reactions and bonding.
- Helps understand periodic table trends.
- Essential for nuclear energy production.
- Used in medical treatments and imaging.
- Basis of modern electronics and technology.
- Helps in understanding isotopes and radioactivity.
Atomic structure knowledge is the foundation of chemistry and modern scientific advancements.
Assertion Reason 💫
Directions:
For each question, choose the correct option:
A. Both Assertion and Reason are true, and Reason is the correct explanation of Assertion.
B. Both Assertion and Reason are true, but Reason is not the correct explanation of Assertion.
C. Assertion is true, but Reason is false.
D. Assertion is false, but Reason is true.
1.
Assertion (A): Most of the space inside an atom is empty.
Reason (R): Rutherford observed that most alpha particles passed straight through the gold foil.
Answer: A
Explanation: The observation proved that atoms contain large empty spaces.
2.
Assertion (A): The nucleus of an atom is positively charged.
Reason (R): It contains protons which carry positive charge.
Answer: A
Explanation: Protons give the nucleus its positive charge.
3.
Assertion (A): Electrons do not lose energy while revolving in fixed shells.
Reason (R): Bohr proposed that electrons move in stable energy levels.
Answer: A
Explanation: Bohr’s model states electrons remain stable in fixed orbits.
4.
Assertion (A): Atomic number determines the identity of an element.
Reason (R): Atomic number equals the number of protons in an atom.
Answer: A
Explanation: Each element has a unique number of protons.
5.
Assertion (A): Isotopes have identical chemical properties.
Reason (R): Isotopes have the same number of electrons.
Answer: A
Explanation: Chemical properties depend on electron configuration.
6.
Assertion (A): Isobars have different chemical properties.
Reason (R): Isobars have different atomic numbers.
Answer: A
Explanation: Different atomic numbers mean different elements.
7.
Assertion (A): The valency of an atom depends on the outermost shell.
Reason (R): Atoms try to achieve stable octet configuration.
Answer: A
Explanation: Valency is determined by electrons needed to complete octet.
8.
Assertion (A): Neutrons do not affect the chemical properties of an element.
Reason (R): Neutrons have no electric charge.
Answer: B
Explanation: Both statements are true, but chemical properties depend mainly on electrons, not simply neutron charge.
9.
Assertion (A): K shell can hold a maximum of 2 electrons.
Reason (R): Maximum electrons in a shell are given by 2n².
Answer: A
Explanation: For K shell (n=1), 2(1)² = 2.
10.
Assertion (A): Sodium has a valency of 1.
Reason (R): Sodium has one electron in its outermost shell.
Answer: A
Explanation: Sodium loses one electron to achieve stability.
More MCQs – Structure of an Atom
Choose the correct option.
1.
The particle with negligible mass is:
A. Proton
B. Neutron
C. Electron
D. Nucleus
Answer: C
2.
Who discovered the proton?
A. Rutherford
B. Goldstein
C. Thomson
D. Bohr
Answer: B
3.
The central part of the atom is called:
A. Orbit
B. Shell
C. Nucleus
D. Electron cloud
Answer: C
4.
Which particle determines the atomic number?
A. Electron
B. Neutron
C. Proton
D. Orbit
Answer: C
5.
Mass number is equal to:
A. Electrons + neutrons
B. Protons + neutrons
C. Protons + electrons
D. Only protons
Answer: B
6.
Maximum number of electrons in M shell is:
A. 8
B. 18
C. 32
D. 2
Answer: B
7.
Which shell is closest to the nucleus?
A. L
B. M
C. K
D. N
Answer: C
8.
Which scientist proposed the plum pudding model?
A. Bohr
B. Thomson
C. Rutherford
D. Dalton
Answer: B
9.
An atom becomes positively charged when it:
A. Gains electrons
B. Loses electrons
C. Gains neutrons
D. Loses protons
Answer: B
10.
Neutrons were discovered by:
A. Rutherford
B. Chadwick
C. Thomson
D. Bohr
Answer: B
11.
Valency of magnesium (atomic number 12) is:
A. 1
B. 2
C. 3
D. 6
Answer: B
12.
Electronic configuration of aluminium (13) is:
A. 2,8,3
B. 2,6,5
C. 2,8,2
D. 2,8,8
Answer: A
13.
Which subatomic particle is neutral?
A. Electron
B. Proton
C. Neutron
D. Nucleus
Answer: C
14.
An element with atomic number 8 has valency:
A. 1
B. 2
C. 6
D. 8
Answer: B
15.
The number of electrons in a neutral atom equals:
A. Neutrons
B. Protons
C. Mass number
D. Valency
Answer: B
16.
Isotopes differ in number of:
A. Protons
B. Electrons
C. Neutrons
D. Orbits
Answer: C
17.
Which of the following is an isotope pair?
A. Carbon-12 & Carbon-14
B. Argon-40 & Calcium-40
C. Sodium & Magnesium
D. Hydrogen & Helium
Answer: A
18.
Which of the following are isobars?
A. Hydrogen & Deuterium
B. Carbon-12 & Carbon-14
C. Argon-40 & Calcium-40
D. Oxygen-16 & Oxygen-18
Answer: C
19.
Bohr’s model is applicable to explain:
A. Nucleus formation
B. Electron distribution
C. Proton discovery
D. Neutron discovery
Answer: B
20.
Valency of neon is:
A. 0
B. 2
C. 4
D. 8
Answer: A
21.
Which particle is responsible for chemical properties?
A. Proton
B. Neutron
C. Electron
D. Nucleus
Answer: C
22.
Which shell can hold a maximum of 8 electrons?
A. K
B. L
C. M
D. N
Answer: B
23.
Atomic number of sodium is 11. Number of protons present:
A. 10
B. 11
C. 12
D. 23
Answer: B
24.
Mass number of an atom is 23 and atomic number is 11. Number of neutrons:
A. 11
B. 12
C. 13
D. 23
Answer: B
25.
Which model first proposed the nucleus?
A. Dalton
B. Thomson
C. Rutherford
D. Bohr
Answer: C
26.
The charge of an alpha particle is:
A. –1
B. +1
C. +2
D. 0
Answer: C
27.
Electronic configuration of oxygen is:
A. 2,8
B. 2,6
C. 2,8,2
D. 6,2
Answer: B
28.
Which element has valency 1?
A. Oxygen
B. Nitrogen
C. Sodium
D. Neon
Answer: C
29.
The number of shells in an atom depends on:
A. Atomic mass
B. Atomic number
C. Period number
D. Valency
Answer: C
30.
Atoms combine to form compounds to achieve:
A. High energy
B. Stability
C. Larger size
D. More neutrons
Answer: B
Case-Based Questions – Structure of an Atom
Read each case carefully and answer the questions that follow.
Case 1: Rutherford’s Gold Foil Experiment
A group of scientists performed Rutherford’s alpha particle scattering experiment. They observed that most alpha particles passed through the foil, some deflected slightly, and very few bounced back.
Questions:
- What did most particles passing straight indicate?
Answer: Most of the atom is empty space. - Why did a few particles bounce back?
Answer: They struck the dense, positively charged nucleus. - Which model was disproved by this experiment?
Answer: Thomson’s plum pudding model.
Case 2: Electronic Configuration of Sodium
A sodium atom has atomic number 11. Its electrons are arranged as 2, 8, 1.
Questions:
- How many shells are present?
Answer: Three shells (K, L, M). - What is the valency of sodium?
Answer: 1 - Why does sodium lose one electron?
Answer: To achieve a stable octet configuration.
Case 3: Isotopes of Hydrogen
Hydrogen has three isotopes: Protium, Deuterium, and Tritium. They have the same atomic number but different mass numbers.
Questions:
- Why do they have the same chemical properties?
Answer: They have the same number of electrons. - Which isotope contains two neutrons?
Answer: Tritium. - What is the atomic number of hydrogen?
Answer: 1
Case 4: Atomic Number and Mass Number
An atom has atomic number 17 and mass number 35.
Questions:
- Name the element.
Answer: Chlorine. - Number of neutrons present?
Answer: 18 - Number of electrons present?
Answer: 17
Case 5: Bohr’s Model of Atom
According to Bohr, electrons revolve in fixed energy levels around the nucleus.
Questions:
- What are these energy levels called?
Answer: Shells or orbits. - Maximum electrons in the L shell?
Answer: 8 - Write the formula for maximum electrons.
Answer: 2n²
Case 6: Valency of Oxygen
Oxygen has electronic configuration 2,6.
Questions:
- How many valence electrons does oxygen have?
Answer: 6 - What is its valency?
Answer: 2 - Why does oxygen gain electrons?
Answer: To complete its octet.
Case 7: Neutral Atom Concept
An atom contains 12 protons and 12 electrons.
Questions:
- What is the charge of the atom?
Answer: Neutral. - What is its atomic number?
Answer: 12 - Name the element.
Answer: Magnesium.
Case 8: Isobars Example
Argon and calcium both have mass number 40 but different atomic numbers.
Questions:
- What are such atoms called?
Answer: Isobars. - Why do they have different chemical properties?
Answer: They have different atomic numbers. - Do they belong to the same element?
Answer: No.
Case 9: Shell Capacity Rule
A student is filling electrons in shells using the rule 2n².
Questions:
- Maximum electrons in K shell?
Answer: 2 - Maximum electrons in M shell?
Answer: 18 - What does “n” represent?
Answer: Shell number.
Case 10: Formation of Ions
A chlorine atom has electronic configuration 2,8,7. It gains one electron to become stable.
Questions:
- What ion is formed?
Answer: Chloride ion (Cl⁻). - Why does chlorine gain an electron?
Answer: To complete its octet. - What is the valency of chlorine?
Answer: 1
Class 9 Chemistry Test Paper
Chapter: Structure of an Atom
Time: 1 Hour
Maximum Marks: 50
Section A: Multiple Choice Questions (1 × 10 = 10 marks)
Choose the correct answer.
- Who discovered the electron?
a) Rutherford
b) Thomson
c) Bohr
d) Dalton - The charge of a proton is:
a) Negative
b) Positive
c) Neutral
d) Variable - Atomic number represents the number of:
a) Neutrons
b) Protons
c) Orbits
d) Shells - Maximum electrons in K shell are:
a) 8
b) 2
c) 18
d) 32 - Which particle has no charge?
a) Electron
b) Proton
c) Neutron
d) Nucleus - Isotopes differ in number of:
a) Protons
b) Electrons
c) Neutrons
d) Shells - The nucleus was discovered by:
a) Thomson
b) Rutherford
c) Bohr
d) Chadwick - Electronic configuration of sodium is:
a) 2,8
b) 2,8,1
c) 2,6,3
d) 8,2,1 - Valency of oxygen is:
a) 1
b) 2
c) 6
d) 8 - The formula for maximum electrons in a shell is:
a) n²
b) 2n
c) 2n²
d) n³
Section B: Very Short Answer Questions (2 × 5 = 10 marks)
- Define atomic number.
- What is a neutron?
- Write the maximum number of electrons in L shell.
- Define isotopes.
- What is valency?
Section C: Short Answer Questions (3 × 5 = 15 marks)
- State three conclusions of Rutherford’s experiment.
- Write the electronic configuration of magnesium (atomic number 12).
- Differentiate between isotopes and isobars (any three points).
- Define mass number. Calculate neutrons in Carbon-14.
- Explain why atoms are electrically neutral.
Section D: Long Answer Questions (5 × 3 = 15 marks)
- Explain Rutherford’s gold foil experiment with observations and conclusions.
- Describe Bohr’s model of the atom and its postulates.
- Explain electronic configuration and valency with suitable examples.
End of Question Paper
Solution – Test Paper
Chapter: Structure of an Atom
Section A: Multiple Choice Questions
- b) Thomson
- b) Positive
- b) Protons
- b) 2
- c) Neutron
- c) Neutrons
- b) Rutherford
- b) 2,8,1
- b) 2
- c) 2n²
Section B: Very Short Answer Questions
11. Define atomic number.
Atomic number is the number of protons present in the nucleus of an atom.
12. What is a neutron?
A neutron is a neutral subatomic particle present in the nucleus of an atom.
13. Write the maximum number of electrons in L shell.
The L shell can hold a maximum of 8 electrons.
14. Define isotopes.
Isotopes are atoms of the same element having the same atomic number but different mass numbers.
15. What is valency?
Valency is the combining capacity of an atom based on electrons in the outermost shell.
Section C: Short Answer Questions
16. State three conclusions of Rutherford’s experiment.
- Most of the atom is empty space.
- A small dense nucleus exists at the center.
- The nucleus contains positive charge.
17. Write the electronic configuration of magnesium (atomic number 12).
Magnesium = 12 electrons
Electronic configuration: 2, 8, 2
18. Differentiate between isotopes and isobars.
| Isotopes | Isobars |
|---|---|
| Same atomic number | Same mass number |
| Different mass numbers | Different atomic numbers |
| Same chemical properties | Different chemical properties |
| Example: Carbon-12 & Carbon-14 | Example: Argon-40 & Calcium-40 |
19. Define mass number. Calculate neutrons in Carbon-14.
Mass number = number of protons + number of neutrons.
Carbon-14:
Atomic number = 6
Mass number = 14
Neutrons = 14 − 6 = 8
20. Explain why atoms are electrically neutral.
Atoms are neutral because the number of protons (positive charge) equals the number of electrons (negative charge). These charges cancel each other, giving the atom no net charge.
Section D: Long Answer Questions
21. Explain Rutherford’s gold foil experiment with observations and conclusions.
Rutherford directed fast-moving alpha particles at a thin gold foil and observed their scattering using a fluorescent screen.
Observations:
- Most particles passed straight through.
- Some deflected slightly.
- Very few bounced back.
Conclusions:
- Most of the atom is empty space.
- Positive charge and mass are concentrated in a small nucleus.
- Electrons revolve around the nucleus.
This led to the nuclear model of the atom.
22. Describe Bohr’s model of the atom and its postulates.
Bohr proposed that electrons revolve around the nucleus in fixed circular paths called shells.
Postulates:
- Electrons move in fixed energy levels.
- Each shell has definite energy.
- Electrons do not lose energy while revolving.
- Energy is emitted or absorbed when electrons jump between shells.
- Maximum electrons in shell = 2n².
Shell capacity:
- K = 2
- L = 8
- M = 18
Bohr’s model explained atomic stability and electronic configuration.
23. Explain electronic configuration and valency with examples.
Electronic configuration is the arrangement of electrons in shells.
Rules:
- Maximum electrons = 2n²
- Outer shell cannot exceed 8 electrons.
Examples:
Sodium (11): 2,8,1
Magnesium (12): 2,8,2
Oxygen (8): 2,6
Valency is the combining capacity of an atom.
- Sodium → loses 1 electron → valency 1
- Magnesium → loses 2 electrons → valency 2
- Oxygen → gains 2 electrons → valency 2
Valency helps atoms achieve a stable octet configuration.
Structure of an Atom – Quick Revision Sheet (Class 9)
The chapter Structure of an Atom explains the internal composition of atoms, which are the basic building blocks of matter. Earlier, Dalton proposed that atoms were indivisible, but later discoveries showed that atoms are made of smaller particles called subatomic particles.
Subatomic Particles
Atoms contain three fundamental particles:
- Electron – discovered by J.J. Thomson; negatively charged and found outside the nucleus.
- Proton – discovered by Eugen Goldstein; positively charged and present in the nucleus.
- Neutron – discovered by James Chadwick; neutral particle located in the nucleus.
Protons and neutrons together form the nucleus, while electrons revolve around it in fixed energy levels.
Rutherford’s Nuclear Model
Rutherford conducted the gold foil experiment by bombarding alpha particles on a thin gold sheet.
Observations:
- Most particles passed straight.
- Some deflected slightly.
- Very few bounced back.
Conclusions:
- Most of the atom is empty space.
- A small, dense, positively charged nucleus exists at the center.
Bohr’s Model of Atom
Bohr improved Rutherford’s model by explaining electron distribution.
Postulates:
- Electrons revolve in fixed shells (K, L, M, N).
- Each shell has definite energy.
- Maximum electrons in a shell = 2n².
- Electons do not lose energy while revolving.
Shell Capacity:
- K = 2
- L = 8
- M = 18
Electronic Configuration
Arrangement of electrons in shells is called electronic configuration.
Examples:
- Oxygen (8) → 2,6
- Sodium (11) → 2,8,1
- Magnesium (12) → 2,8,2
Outer shell electrons are called valence electrons.
Valency
Valency is the combining capacity of an atom.
- If outer shell has 1–4 electrons → valency = electrons lost.
- If outer shell has 5–7 electrons → valency = electrons gained.
- Noble gases have valency 0 (stable octet).
Examples:
- Sodium → 1
- Oxygen → 2
- Neon → 0
Atomic Number and Mass Number
- Atomic number (Z) = number of protons (also equals electrons).
- Mass number (A) = protons + neutrons.
Neutrons = Mass number − Atomic number
Example:
Carbon (A=12, Z=6) → Neutrons = 6
Isotopes
Atoms of the same element with same atomic number but different mass numbers.
Examples: Hydrogen isotopes (Protium, Deuterium, Tritium)
Uses:
- Carbon dating
- Nuclear reactors
- Medical treatment
Isobars
Atoms with same mass number but different atomic numbers.
Example: Argon-40 and Calcium-40
Key Points to Remember
- Atom is electrically neutral (protons = electrons).
- Nucleus contains most of the mass.
- Electrons determine chemical properties.
- Valency depends on outermost shell electrons.
- 2n² rule is essential for electron distribution.
- Isotopes have same chemical properties but different masses.
Exam Focus
- Rutherford experiment conclusions
- Bohr model postulates
- Electronic configuration practice
- Differences: isotopes vs isobars
- Atomic number & mass number numericals
This chapter forms the foundation for understanding chemical bonding, periodic classification, and advanced chemistry concepts. Mastering the Structure of an Atom ensures strong conceptual clarity and better exam performance.




Leave a Reply