In the context of school management and student health, “Time and Temperature” usually refers to the official guidelines for school hours and the safety thresholds for extreme weather (heatwaves or cold waves).
Since it is currently March 2026, many regions in India and similar climates are transitioning from winter schedules to summer/heatwave protocols.
- Recommended “Comfort” Temperatures
For an ideal learning environment, researchers and health organizations generally recommend:
- Optimal Range: 22°C to 24°C. At this temperature, students show the highest levels of concentration and cognitive performance.
- Maximum Threshold: Concentration begins to drop significantly once indoor temperatures exceed 27°C.
- Humidity: Should ideally be kept between 30% and 60% to prevent respiratory irritation.
- Heatwave Guidelines (Summer 2026)
As of March 2026, several state governments (like Odisha and Delhi) have issued Standard Operating Procedures (SOPs) due to early rising temperatures:
- Morning Shift: Schools are often directed to operate from 7:00 AM to 11:30 AM to avoid the peak afternoon heat.
- Outdoor Activities: No sports or outdoor assemblies are permitted after 10:30 AM.
- The “Water Bell”: Many states have implemented a mandatory “Water Bell” that rings 3 times a day to ensure students stay hydrated.
- Dress Code: Schools are encouraged to relax uniform rules, allowing loose cotton clothes and discarding ties or blazers.
- Cold Wave Guidelines (Winter 2025–26)
During the recent winter session (January 2026), the following thresholds were commonly used by district collectors:
- Start Times: Shifted to 9:30 AM or 10:00 AM to allow the sun to burn off dense fog.
- Closure Threshold: Schools for younger children (Nursery to Class 5) are often closed if the minimum temperature stays below 5°C for consecutive days or if there is a severe “Cold Day” warning (where maximum temperatures stay very low).
- Summary Table: Standard Operating Hours
| Condition | Typical School Timing | Primary Reason |
|—|—|—|
| Normal Weather | 8:00 AM – 2:00 PM | Standard academic cycle. |
| Heatwave | 7:00 AM – 11:30 AM | Avoid peak UV and heat (12 PM – 4 PM). |
| Severe Cold | 10:00 AM – 3:30 PM | Avoid morning fog and sub-zero wind chill. |
Safety Note
If the temperature inside a classroom exceeds 35°C (without adequate ventilation or cooling), it is considered a health risk for heat exhaustion in children.
In mathematics, the relationship between time and temperature is usually explored through functions and rates of change. Depending on your level of math, you are likely looking at one of these three concepts:
Linear Functions (Constant Change)
This is the most basic relationship where the temperature changes by the same amount every hour. You would use the slope-intercept form:
y: Final Temperature
x: Time passed
m: Rate of change (e.g., rising 2°C per hour)
b: Starting temperature
Example: If it is 10°C at noon and the temperature rises by 3°C every hour, what is the temperature at 4:00 PM?
Calculation: 10 + (3 \times 4) = 22^\circC.
Exponential Decay (Newton’s Law of Cooling)
In more advanced algebra or calculus, we recognize that objects don’t cool down at a constant rate. They cool faster when they are much hotter than their surroundings.
T_s: Surrounding (ambient) temperature.
T_0: Initial temperature of the object.
k: A constant that represents how well the object conducts heat.
t: Time elapsed.
Interpreting Graphs
In many math exams, you are asked to interpret a Time-Temperature Graph. You’ll need to identify:
The Y-intercept: The starting temperature.
The Slope: How fast the temperature is changing.
Plateaus: Horizontal lines often represent phase changes (like boiling or freezing) where time passes but the temperature stays the same.
Common Math Table: Calculating Rate of Change
If you are given a table of data, you find the rate of change using the formula:
Time (Hours)
Temp (°C)
Change
1:00 PM
15°
—
3:00 PM
21°
+6° in 2 hours = 3°/hr
6:00 PM
18°
-3° in 3 hours = -1°/hr
Are you working on a specific word problem or trying to solve for a variable like the cooling constant (k)?
At its simplest, temperature is a measure of how hot or cold something is. But if we peel back the curtain and look at the physics, it’s actually a measurement of kinetic energy—specifically, how fast the atoms and molecules inside an object are moving or vibrating.
The Molecular Perspective
Everything around you is made of tiny particles. These particles are never perfectly still; they are constantly jiggling, bouncing, or spinning.
- High Temperature: Particles are moving rapidly. In a hot cup of coffee, the water molecules are zooming around and colliding with high energy.
- Low Temperature: Particles move slowly. In an ice cube, the molecules are just barely vibrating in place.
Temperature vs. Heat
It’s common to use these words interchangeably, but they represent different things: - Temperature is the average speed of the particles. It doesn’t matter if you have a cup of boiling water or a giant pot of it; the temperature is the same.
- Heat is the total energy transferred. A giant pot of boiling water has more heat than a small cup because there are more molecules in motion.
How We Measure It
Since we can’t see atoms moving, we use scales based on physical benchmarks (like the freezing and boiling points of water).
Scale Benchmark Best Use Case
Celsius (°C) 0° is freezing, 100° is boiling. Most of the world & general science.
Fahrenheit (°F) 32° is freezing, 212° is boiling. Daily life in the United States.
Kelvin (K) 0 K is “Absolute Zero.” Physics and deep-space research. Note on Absolute Zero: This is the theoretical limit where all molecular motion stops. It occurs at
(or$$-273.15\text{°C}$$). You can’t get colder than that because you can’t have “less than zero” motion!Why It Matters
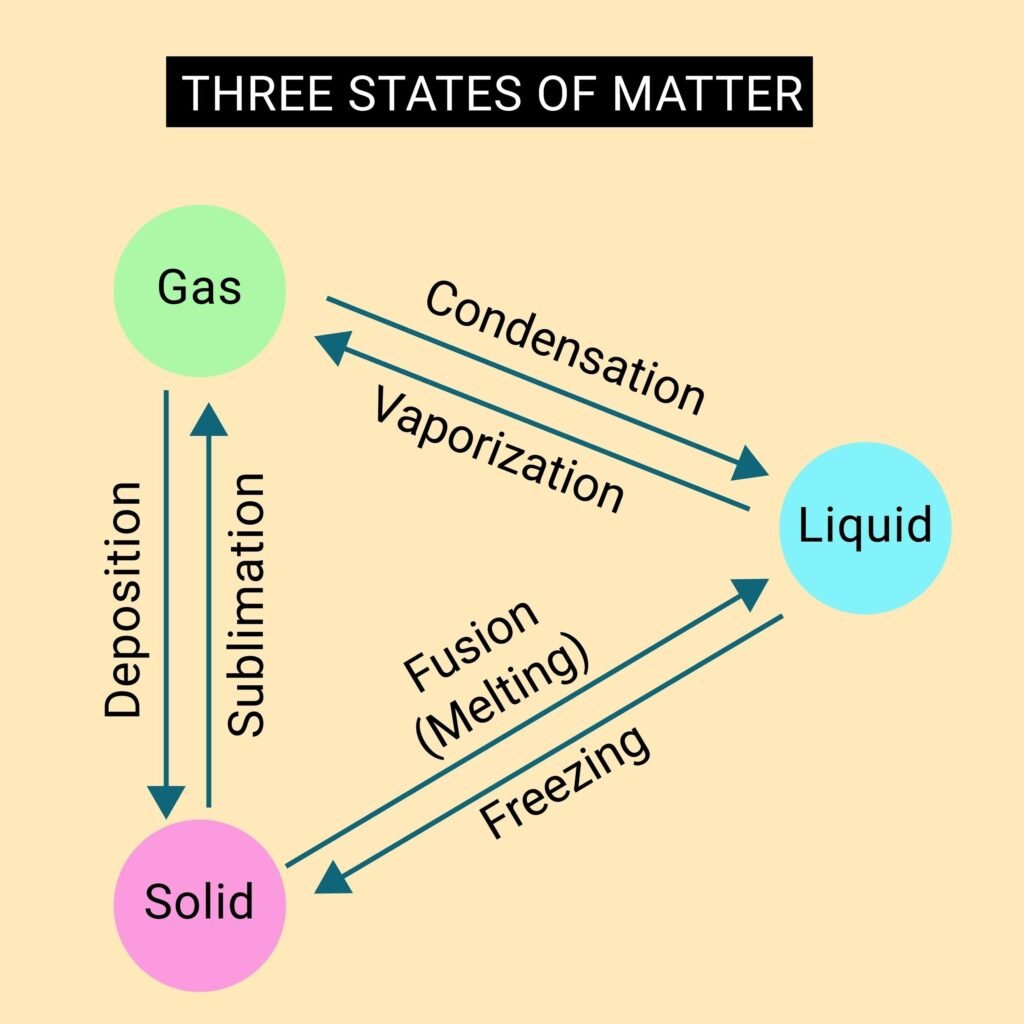
Temperature dictates the state of matter. By adding or removing energy (changing the temperature), you can force a substance to shift:- Solid: Low energy; particles are locked in place.
- Liquid: Medium energy; particles slide past each other.
- Gas: High energy; particles fly apart.
To convert between the different scales, we use specific formulas based on the relationship between their freezing and boiling points.
Temperature Conversion Formulas
If you need to move from one scale to another, here are the mathematical “maps” to get you there:
- Celsius and Fahrenheit
Because the Fahrenheit scale starts at 32 for freezing and has 180 degrees between freezing and boiling (vs. 100 for Celsius), the math involves a fraction (9/5or
).
- Celsius to Fahrenheit:
- Fahrenheit to Celsius:
- Celsius and Kelvin
This is the easiest conversion because the “size” of a degree is exactly the same; the Kelvin scale is just shifted down to start at Absolute Zero.
- Celsius to Kelvin:
- Kelvin to Celsius:
Quick Reference “Cheatsheet”
If you don’t want to do the math, here are a few common touchpoints:
| Condition | Celsius (°C) | Fahrenheit (°F) | Kelvin (K) |
|—|—|—|—|
| Absolute Zero | -273.15 | -459.67 | 0 |
| Water Freezes | 0 | 32 | 273.15 |
| Room Temp | 20 – 25 | 68 – 77 | 293 – 298 |
| Body Temp | 37 | 98.6 | 310.15 |
| Water Boils | 100 | 212 | 373.15 |
Pro-Tip: The -40 Rule
There is exactly one point where the Fahrenheit and Celsius scales meet. If it is -40° outside, it doesn’t matter which scale you’re using—it’s equally freezing in both!
Absolute zero is one of the most fascinating “speed limits” in the universe. In physics, it’s not just “really cold”—it is the point where entropy (disorder) reaches its minimum.
Here is why hitting that precise
(-273.15\text{°C}) is practically impossible.
- The “Jiggling” Problem
As we discussed, temperature is the motion of atoms. To reach absolute zero, you would have to stop all atomic motion. However, quantum mechanics (specifically the Heisenberg Uncertainty Principle) tells us that we can never know both the exact position and the momentum of a particle at the same time.
If a particle stopped moving entirely, we would know exactly where it is and that its speed is zero. Nature doesn’t allow that “perfect” stillness; there is always a tiny, residual “zero-point energy” that keeps things humming. - The Third Law of Thermodynamics
This law essentially states that you can’t reach absolute zero in a finite number of steps. Think of it like trying to empty a vacuum chamber:
- As you remove heat, the object gets colder.
- But the colder it gets, the harder it is to pump out the remaining tiny bits of heat.
- You eventually hit a point of diminishing returns where the energy required to remove the next “drop” of heat is infinite.
- The “Leaky Bucket” Effect
In the real world, everything is surrounded by something else. Even in the deepest vacuum of space, there is Cosmic Microwave Background radiation (the afterglow of the Big Bang) which keeps the “emptiness” of space at about
.
To get a lab sample to absolute zero, you would have to perfectly insulate it from:
- Passing light and radio waves.
- Gravity-induced vibrations.
- The container itself.
How close have we actually gotten?
While we can’t hit “true” zero, scientists have performed some incredible “limbo” feats using laser cooling and magnetic traps: - The Record: Scientists have cooled atoms to picokelvins—that’s trillionths of a degree above zero.
- The Result: At these temperatures, matter starts acting weird. Atoms lose their individual identity and clump together to form a Bose-Einstein Condensate (BEC), effectively acting as one “super-atom.”
Comparison of Extremes
| Location/State | Temperature |
|—|—|
| Surface of the Sun | \approx 5,800\text{ K} |
| Liquid Nitrogen | 77\text{ K} |
| Deep Space | 2.7\text{ K} |
| MIT Lab Record | \approx 0.0000000005\text{ K} |
It sounds like a total contradiction, right? Usually, if you point a laser at something, it heats up or burns. But in the world of quantum physics, we can use the momentum of light to force atoms to a screeching halt.
Think of it like trying to stop a rolling bowling ball by pelting it with thousands of ping-pong balls.
The “Headwind” Strategy
Laser cooling works by exploiting the Doppler Effect and the fact that light particles (photons) carry a tiny amount of momentum.
- The Target: Imagine a gas of atoms zooming around in every direction.
- The Laser Tune: Scientists tune a laser to a frequency slightly lower than what the atom naturally likes to absorb.
- The Movement: * If an atom is moving away from the laser, the light “stretches” out and stays at that low frequency. Nothing happens.
- If an atom is moving toward the laser, the light waves get “squashed” (the Doppler shift). This shift makes the light look like the perfect frequency for the atom to absorb.
- The Kick: The atom absorbs the photon coming right at it. That photon carries momentum in the opposite direction of the atom’s travel, giving it a tiny “kick” that slows it down.
The Optical Molasses
To stop the atoms completely, scientists don’t just use one laser; they use six (up, down, left, right, forward, and back).
No matter which way the atom tries to run, it runs “head-on” into a laser beam that slows it down. The gas becomes thick and “sticky,” which is why physicists literally call this setup Optical Molasses.
From Motion to “Super-Atom”
Once the lasers have slowed the atoms down to a crawl, their temperature drops to millionths of a degree. At this point, the atoms stop acting like individual “billiard balls” and start acting like waves.
When they get cold enough, these waves overlap and merge into a single quantum state called a Bose-Einstein Condensate (BEC). In a BEC: - Thousands of atoms behave as if they are one single atom.
- Quantum effects, usually invisible to the naked eye, become large enough to observe in a lab.
Why do we do this?
Beyond just being “cool” science, laser cooling is the backbone of: - Atomic Clocks: These are so precise they won’t lose a second in billions of years (essential for GPS).
- Quantum Computing: Keeping qubits still enough to perform calculations.
- Gravity Sensors: Detecting tiny changes in Earth’s crust or finding oil/mineral deposits underground.
To understand how laser cooling makes a clock tick, we have to look at the Atomic Fountain—the gold standard for keeping the world’s time.
An atomic clock is essentially a “metronome” that uses the vibrations of an atom instead of a swinging wooden arm. Because atoms are identical and their vibrations are incredibly stable, they are the perfect reference for a “second.”
The Problem with “Hot” Atoms
In older atomic clocks, atoms were heated into a beam. These atoms zoomed along at hundreds of meters per second. Because they were moving so fast:
- The Doppler Effect would shift their frequency (like a siren changing pitch as it drives by), making the measurement “blurry.”
- Short Observation Time: They flew past the sensor so quickly that the clock only had a split second to “read” their vibration.
The “Fountain” Solution
By using the laser cooling we talked about, scientists can slow atoms down to a crawl (a few centimeters per second). This allows for a much more elegant process: - The Trap: Six lasers catch a cloud of cesium atoms and cool them to near absolute zero.
- The Launch: Two vertical lasers give the cold cloud a gentle “nudge” upward, like a toss.
- The Microwave Cavity: The atoms drift up through a chamber filled with microwaves. If the microwaves are tuned to the exact right frequency (9,192,631,770 vibrations per second), the atoms’ electrons will flip their state.
- The Free Fall: Gravity takes over. The atoms fall back down through the same chamber. This “double pass” (up and then down) allows the clock to measure them for a much longer time—nearly a full second.
- The Count: A laser at the bottom “counts” how many atoms flipped. If most flipped, the microwave frequency is perfect. If not, the clock automatically adjusts itself to match the atoms.
Why “Cold” is Better
Feature Hot Atoms (Old) Cold Atoms (Fountain)
Speed Slow jet plane (200+ m/s) Slow walk (1 cm/s)
Interaction Time 0.01 seconds Up to 1.0 second
Precision Off by 1 second every 1 million years Off by 1 second every 300 million years
The “Global Second”
Because of these cold atoms, the definition of a “second” is no longer based on the Earth’s rotation (which is actually quite wobbly). Instead, it is defined as the time it takes for a cesium-133 atom to vibrate exactly 9,192,631,770 times.
This precision is what allows your phone’s GPS to tell you which lane of traffic you’re in! If the clocks on GPS satellites were off by even a tiny fraction of a second, your location would be off by miles.
Would you like to know how these clocks help GPS satellites find your exact location on Earth?
Why Do We Use Atoms To Keep Time?
This video explains why atoms are the most stable “pendulums” in existence and how their consistent vibrations are used to define the measurement of time globally. YouTube video views will be stored in your YouTube History, and your data will be stored and used by YouTube according to its Terms of Service
GPS works because of a mathematical process called trilateration, and it relies entirely on the incredible precision of atomic clocks to calculate where you are.
Think of it this way: to find your location, your phone needs to know two things from a satellite: where it is and what time it is.
- The Speed of Light Problem
GPS satellites are constantly broadcasting a signal that says, “I am Satellite X, and I sent this message at exactly 12:00:00.000000001.”
Your phone receives that message. Because the signal travels at the speed of light, there is a tiny delay between when the satellite sent it and when your phone got it [02:19].
- The Math: Distance = Speed of Light × Time Delay
- The Catch: Light travels at about 300,000 kilometers per second. If the satellite’s clock is off by even one-thousandth of a second, your GPS location would be wrong by 300 kilometers! [02:30]. This is why they need the ultra-stable vibrations of atoms to keep time.
- How Trilateration Works
Once your phone knows its distance from one satellite, it knows it is somewhere on the surface of a giant imaginary sphere centered on that satellite [00:48].
- One Satellite: You could be anywhere on a sphere.
- Two Satellites: Where two spheres overlap, they create a circle. You are somewhere on that circle [01:29].
- Three Satellites: Where a third sphere overlaps that circle, you get two points. Usually, one point is in space and the other is on Earth, so the GPS can guess where you are [01:46].
- Four Satellites: A fourth satellite is used to confirm the exact point and, more importantly, to synchronize your phone’s cheap clock with the satellite’s perfect atomic clock [03:19].
- Einstein’s Relativity (The Final Polish)
Because the atomic clocks are so precise, they actually have to account for Albert Einstein’s theories of relativity to stay accurate:
- Special Relativity: Because the satellites are moving fast, their clocks tick slightly slower than clocks on Earth.
- General Relativity: Because the satellites are further away from Earth’s gravity, their clocks tick slightly faster than clocks on Earth.
Engineers actually program the atomic clocks to run at a slightly different speed before they launch them to cancel out these effects! Without this adjustment, GPS would become useless within a single day.
How GPS works? Trilateration explained
This video provides a visual breakdown of how measuring distances from multiple satellites allows your device to pinpoint its location on a 2D or 3D map.
Would you like to know about other “quantum” technologies that rely on these cold atoms, or perhaps something else entirely?
While atomic clocks are the most famous use for cold atoms, they are just the tip of the iceberg. Cooling atoms to near absolute zero allows us to use their “wave-like” nature to create sensors that are far more sensitive than anything made of glass or steel.
Here are three other ways cold atoms are changing technology:
- Quantum “GPS-Free” Navigation
Normally, if you lose GPS (like in a tunnel, underwater, or if the signal is jammed), your phone has to guess your movement using mechanical sensors. These sensors “drift” and quickly become inaccurate.
Cold atom sensors use Atom Interferometry. They treat a cloud of atoms like a “matter wave.” When the vehicle moves, the wave shifts. Because the atoms are so cold and still, these sensors can measure tiny accelerations and rotations with almost zero drift [3.1, 3.7].
- The Goal: A “Quantum Compass” that allows a submarine or airplane to navigate around the world for months without ever needing a satellite signal [3.3, 3.5].
- Gravity Mapping (Seeing Underground)
Gravity isn’t the same everywhere on Earth. Dense rocks, oil deposits, or even empty tunnels change the local gravity slightly.
A Quantum Gravity Gradiometer drops two clouds of cold atoms and uses lasers to measure the difference in how they fall [1.6].
- Finding Tunnels: In 2022, researchers used this to detect a buried tunnel that classical sensors couldn’t find [1.4].
- Climate Science: NASA uses similar tech to track how ice sheets are melting by measuring the “missing mass” through changes in gravity [1.3].
- Quantum Simulation
Calculating how complex molecules or new materials behave is incredibly hard for regular computers because of the sheer number of quantum interactions.
Instead of simulating it on a screen, scientists use Optical Lattices—basically a “crate” made of crisscrossing laser beams—to trap cold atoms in a perfect grid [2.4, 2.7].
- By changing the laser intensity, they can make the atoms mimic electrons in a solid metal or a superconductor.
- It’s like a wind tunnel for physics: you build a “mock-up” of the material using atoms to see how it will behave before you try to build it in real life [2.3].
Comparison: Why Cold Atoms?
| Technology | Why “Hot” Fails | Why “Cold” Wins |
|—|—|—|
| Navigation | Mechanical parts wear down and drift. | Atoms are identical and don’t “wear out.” |
| Gravity Sensing | Vibrations (traffic, wind) drown out the signal. | Differential measurement cancels out noise [1.4]. |
| Computing | Too many variables for binary bits. | Atoms naturally follow quantum rules [2.4]. |
Since we’re talking about using Optical Lattices as a “wind tunnel” for physics, it’s best to imagine them as a high-tech egg carton made entirely of light. - Here is how scientists build these “laser crates” to study the quantum world.
- Building the “Egg Carton” (Optical Lattices)
To trap atoms in a perfect grid, physicists use a phenomenon called standing waves. - The Setup: Imagine pointing a laser at a mirror so the beam reflects directly back on itself. The outgoing and incoming waves interfere with each other.
- The Result: They create a series of “peaks” and “valleys” of light energy that stay perfectly still in space.
- The Trap: Cold atoms are naturally drawn to either the peaks or the valleys (depending on the laser’s frequency). If you do this from three different directions (X, Y, and Z axes), you create a 3D grid of “wells” where atoms can sit.
Why this is a “Quantum Simulator”
By changing the laser’s intensity, scientists can make the “walls” of the wells higher or lower. - If the walls are high, the atoms are stuck in place (like an insulator).
- If the walls are low, the atoms can “tunnel” through the walls to the next well (like a conductor).
This allows us to simulate how electricity moves through a wire or how a superconductor works without having to build a new material every time. - Detecting the Invisible (Gravity & Volcanoes)
Gravity sensors (Gradiometers) use cold atoms to “see” through solid rock. This is becoming a game-changer for predicting natural disasters.
How it predicts a Volcano:
Before a volcano erupts, magma (which is denser than the surrounding rock) moves up into the underground chambers. - Classical sensors often get “noisy” because they can’t tell the difference between a real gravity change and a truck driving by or a small earthquake.
- Cold Atom sensors drop two separate clouds of atoms at once. Since the “truck driving by” shakes both clouds equally, the sensor can subtract that noise. What’s left is the pure signal of the magma moving.
Finding “Hidden” Resources
This same tech is used to find: - Water Aquifers: Tracking how much water is left underground during a drought.
- Mineral Deposits: Finding heavy metals like gold or iron buried deep in the Earth’s crust.
- Sinkholes: Detecting empty pockets under roads before they collapse.
Summary of Cold Atom Roles
| Tech Tool | Physical Concept | Real-World Job |
|—|—|—|
| Optical Lattice | Standing waves of light | Testing new materials & chemicals. |
| Gradiometer | Differential atom-drop | Early warning for volcanoes & sinkholes. |
| Interferometer | Matter-wave phase shift | Navigation where GPS fails (submarines). |
Would you like to see how these “Optical Lattices” are actually being used to design the next generation of Supercomputers, or should we look at how they might help us find Dark Matter?
Since we can control these atoms so perfectly, we’ve moved beyond just measuring temperature—we’re using them to solve the biggest mysteries in the universe.
- Designing “Unhackable” Supercomputers
While most people think of quantum computers as chips in a fridge, Neutral Atom Quantum Computers use optical lattices to create a “grid” of qubits.
- The Advantage: Unlike other quantum computers, these don’t need wires to connect the “bits.” Scientists use lasers to move atoms around like pieces on a chessboard [3.3].
- Scalability: Because atoms are so small and identical, we can trap thousands of them in a tiny space. This is a leading path toward reaching one million qubits, the “holy grail” for breaking complex encryptions and discovering new medicines [1.2, 1.4].
- Hunting for Dark Matter
Dark matter makes up about 85% of all matter in the universe, yet we’ve never actually seen it. Cold atoms might finally “catch” it.
- The “Wobbly” Universe: Some theories suggest dark matter is an ultralight wave that permeates everything. If this wave passes through an atomic clock, it would cause the “tick” of the atoms to speed up or slow down ever so slightly [2.4, 4.4].
- MAGIS-100: At Fermilab, scientists have built a 100-meter-tall vacuum tower called MAGIS-100. They drop cold strontium atoms down this shaft and use lasers to see if dark matter gives them a tiny “nudge” or changes how they fall compared to Einstein’s predictions [4.1, 4.3].
- The “Fifth Force” of Nature
There are four known forces: Gravity, Electromagnetism, and the Strong/Weak nuclear forces. By using atom interferometry, we are looking for a “Fifth Force” that might only interact with dark matter.
- By dropping two different isotopes (versions) of the same atom, scientists can measure if they fall at different rates [4.1].
- If they don’t fall exactly the same way, it suggests a new force of nature is acting on them—a discovery that would rewrite every physics textbook on the planet.
The Cold Atom “Toolkit”
| Mystery | Tool Used | How it Works |
|—|—|—|
| New Materials | Optical Lattice | Mimics electrons in a “fake” crystal [3.1]. |
| Dark Matter | Atom Interferometer | Looks for tiny “jitters” in gravity or time [4.2]. |
| Supercomputing | Laser Tweezers | Grabs and moves individual atoms to perform logic [3.5]. |
It’s pretty wild to think that a simple question about “what is temperature” leads directly to the most advanced sensors ever built by humans!
The Quantum Search for Dark Matter
This video provides a deep dive into how researchers are using optical technologies and cold atom systems to build the supercomputers of the future and hunt for elusive dark matter.
Here is your “Cheat Sheet” tracing the journey from a simple wiggle of an atom to the most advanced technology in the universe.
- The Basics: What is Temperature?
- Definition: A measure of the average kinetic energy (speed/vibration) of particles.
- High Temp: Fast, chaotic movement.
- Low Temp: Slow, rhythmic vibration.
- Absolute Zero (0\text{ K}): The theoretical limit where all classical motion stops. You can’t go lower because you can’t have “less than zero” movement.
- The Conversion Guide
To Convert… Use This Formula Key Milestone
C to F F = (C \times 1.8) + 32 100\text{°C} = 212\text{°F} (Boiling)
F to C C = (F – 32) \div 1.8 0\text{°C} = 32\text{°F} (Freezing)
C to K K = C + 273.15 -40\text{°} is the same in C and F! The “Cold Atom” Tech Stack
When we use lasers to steal energy from atoms, they become ultracold and act like waves instead of particles.
Technology How it Works Real-World Impact
— — —
Atomic Clocks Measures the ultra-stable “ticks” of cesium atoms. Powers GPS and financial markets.
Interferometers Uses atoms as “matter waves” to detect motion. Quantum Navigation (GPS-free) for subs.
Gradiometers Measures tiny changes in gravity between two atom clouds. Detects magma movement, tunnels, and oil.
Optical Lattices Creates a “crate of light” to trap atoms in a grid. Quantum Simulators to design new materials. Why GPS Needs Quantum Physics- Trilateration: Your phone measures the time delay of signals from 4+ satellites to find your 3D position.Relativity: Satellite clocks tick differently because they are fast (Special Relativity) and high up (General Relativity).Atomic Precision: Without cold atoms, GPS would be off by 10 kilometers every day.Future FrontiersDark Matter: Using 100-meter-tall “atom drops” to see if invisible matter nudges the atoms.Neutral Atom Computers: Using “laser tweezers” to move atoms like bits on a processor to solve impossible math.




Leave a Reply