Structure of an Atom Class 11 Chemistry Notes, Summary, MCQs & Important Questions
Meta Description
Complete notes on Structure of an Atom Class 11 Chemistry including summary, formulas, MCQs, important questions, and exam tips for quick revision.
Introduction of the Chapter
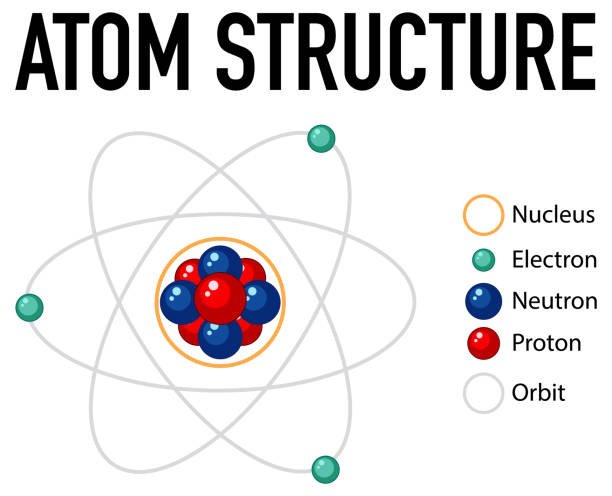
The chapter Structure of an Atom in Class 11 Chemistry explains how scientists discovered the internal structure of atoms and how subatomic particles are arranged. Earlier, atoms were believed to be indivisible. However, experiments by scientists revealed that atoms consist of smaller particles such as electrons, protons, and neutrons.
Understanding the Structure of an Atom is essential because it forms the foundation of chemistry. Concepts like electronic configuration, quantum numbers, and atomic models help explain chemical bonding, periodic trends, and reactivity.
This chapter introduces major atomic models proposed by J. J. Thomson, Ernest Rutherford, and Niels Bohr, and explains the modern quantum mechanical model of the atom.
Short Notes (Quick Revision)
• Atom consists of electrons, protons, and neutrons.
• Electron was discovered by J. J. Thomson using cathode ray tube experiment.
• Proton was discovered by Goldstein through canal rays.
• Neutron was discovered by Chadwick.
• Rutherford proposed nuclear model of atom.
• Bohr gave quantized orbits for electrons.
• Atomic number (Z) = number of protons.
• Mass number (A) = protons + neutrons.
• Isotopes have same atomic number but different mass numbers.
• Orbitals represent probability of finding electrons.
• Quantum numbers describe position and energy of electrons.
• Aufbau principle explains filling of electrons.
• Pauli Exclusion Principle: no two electrons have same quantum numbers.
• Hund’s Rule: electrons fill orbitals singly first.
Detailed Summary of Structure of an Atom (Class 11 Chemistry)
The Structure of an Atom chapter explains how the concept of atom evolved through scientific experiments and theoretical models.
Discovery of Subatomic Particles
The first breakthrough came from J. J. Thomson, who discovered electrons using a cathode ray discharge tube. He observed that cathode rays consist of negatively charged particles present in all atoms.
Eugen Goldstein discovered canal rays, which led to the discovery of positively charged particles called protons.
Later, James Chadwick discovered neutrons in 1932. Neutrons are neutral particles located in the nucleus.
Properties of Subatomic Particles
| Particle | Charge | Mass |
|---|---|---|
| Electron | −1 | negligible |
| Proton | +1 | 1 amu |
| Neutron | 0 | 1 amu |
Atomic Models
Thomson’s Plum Pudding Model
Thomson proposed that electrons are embedded in a positively charged sphere like raisins in pudding. This model could not explain experimental observations.
Rutherford’s Nuclear Model
Rutherford performed the gold foil experiment. He observed:
• Most alpha particles passed straight.
• Some deflected.
• Very few bounced back.
Conclusions: • Atom has a dense, positively charged nucleus.
• Most of the atom is empty space.
• Electrons revolve around nucleus.
Limitations: Could not explain stability of atom.
Bohr’s Atomic Model
Niels Bohr proposed that:
• Electrons revolve in fixed circular orbits.
• Each orbit has fixed energy (quantized).
• Energy is emitted or absorbed when electrons jump between orbits.
Energy of electron:
Eₙ = −2.18 × 10⁻¹⁸ (Z²/n²) J
Bohr model successfully explained hydrogen spectrum but failed for multi-electron atoms.
Dual Nature of Matter
Louis de Broglie proposed that electrons behave both as particles and waves.
λ = h / mv
This concept led to the development of quantum mechanics.
Heisenberg’s Uncertainty Principle
It states that the exact position and momentum of an electron cannot be determined simultaneously.
Δx × Δp ≥ h/4π
This means electrons do not move in fixed paths.
Quantum Mechanical Model of Atom
This modern model describes electrons in terms of probability.
• Orbit → replaced by orbital
• Orbital → region of maximum probability
Types of Orbitals
| Orbital | Shape | Max Electrons |
|---|---|---|
| s | spherical | 2 |
| p | dumbbell | 6 |
| d | cloverleaf | 10 |
| f | complex | 14 |
Quantum Numbers
Quantum numbers describe electron location and energy.
- Principal quantum number (n) → shell
- Azimuthal quantum number (l) → subshell
- Magnetic quantum number (m) → orbital orientation
- Spin quantum number (s) → electron spin
Electronic Configuration
Rules for filling electrons:
Aufbau Principle → lowest energy filled first
Pauli Exclusion Principle → max 2 electrons per orbital
Hund’s Rule → half-filled orbitals preferred
Example:
Oxygen (8) → 1s² 2s² 2p⁴
Important Concepts
Atomic Number (Z)
Number of protons.
Mass Number (A)
Protons + neutrons.
Isotopes
Same Z, different A (e.g., Hydrogen isotopes).
Isobars
Same mass number but different elements.
Flowchart / Mind Map (Text Format)
Atom
→ Subatomic Particles
→ Electron
→ Proton
→ Neutron
→ Atomic Models
→ Thomson Model
→ Rutherford Model
→ Bohr Model
→ Quantum Mechanical Model
→ Quantum Concepts
→ Dual Nature
→ Uncertainty Principle
→ Orbitals
→ Quantum Numbers
→ Electronic Configuration
→ Isotopes & Isobars
Important Keywords with Meanings
Atom – smallest unit of matter
Electron – negatively charged particle
Proton – positively charged particle
Neutron – neutral particle
Atomic number – number of protons
Mass number – protons + neutrons
Orbital – region of electron probability
Quantum numbers – describe electron position
Isotopes – same element, different masses
Aufbau principle – filling order rule
Valence shell – outermost shell
Important Questions & Answers
Short Answer Questions
- Who discovered the electron?
Ans: J. J. Thomson. - What is atomic number?
Ans: Number of protons. - Define isotope.
Ans: Atoms with same atomic number but different mass numbers. - What is an orbital?
Ans: Region where probability of finding electron is high. - State Pauli Exclusion Principle.
Ans: No two electrons have same four quantum numbers. - What is mass number?
Ans: Sum of protons and neutrons. - Who discovered neutron?
Ans: James Chadwick. - What is principal quantum number?
Ans: It denotes shell number. - What is dual nature of matter?
Ans: Matter shows wave and particle properties. - Maximum electrons in p-orbital?
Ans: 6
Long Answer Questions
- Explain Rutherford’s gold foil experiment and conclusions.
Answer: Rutherford bombarded thin gold foil with alpha particles. Most passed straight, some deflected, few rebounded. This proved the presence of a small dense nucleus and empty space. - Describe Bohr’s atomic model and its limitations.
Answer: Bohr proposed fixed energy orbits and quantization. It explained hydrogen spectra but failed for multi-electron atoms. - Explain quantum numbers in detail.
Answer: Quantum numbers describe energy, shape, orientation, and spin of electrons. - Define orbitals and explain types.
Answer: Orbitals are probability regions. Types include s, p, d, f. - State Heisenberg Uncertainty Principle.
Answer: Exact position and momentum cannot be determined simultaneously. - Write differences between orbit and orbital.
Answer: Orbit is fixed path; orbital is probability region. - Explain Aufbau principle with examples.
Answer: Electrons fill lowest energy orbitals first. - What are isotopes? Give examples.
Answer: Hydrogen isotopes: protium, deuterium, tritium. - Explain dual nature of electrons.
Answer: Electrons exhibit wave and particle nature. - Describe electronic configuration rules.
Answer: Aufbau, Pauli exclusion, and Hund’s rule govern electron filling.
MCQs with Answers
- Electron was discovered by
A. Rutherford
B. Thomson
C. Bohr
D. Chadwick
Ans: B - Proton charge is
A. −1
B. 0
C. +1
D. +2
Ans: C - Neutron was discovered by
A. Goldstein
B. Chadwick
C. Bohr
D. Thomson
Ans: B - Maximum electrons in s orbital
A. 1
B. 2
C. 6
D. 10
Ans: B - Atomic number equals
A. electrons
B. neutrons
C. protons
D. nucleons
Ans: C - Shape of p orbital
A. spherical
B. dumbbell
C. circular
D. square
Ans: B - Bohr model explains
A. all atoms
B. hydrogen atom
C. molecules
D. solids
Ans: B - Orbital represents
A. path
B. energy
C. probability
D. motion
Ans: C - Quantum number for shell
A. n
B. l
C. m
D. s
Ans: A - Aufbau principle deals with
A. shape
B. filling
C. spin
D. charge
Ans: B - Maximum electrons in d subshell = 10
- Isotopes have same atomic number
- Mass number = protons + neutrons
- Hund’s rule → maximum multiplicity
- 2p subshell has 3 orbitals
- Electron charge = −1
- Proton mass ≈ 1 amu
- Orbital shape of s is spherical
- n=1 shell is K shell
- Total orbitals in p subshell = 3
Exam Tips & Value-Based Questions
Exam Tips
✔ Learn quantum numbers carefully.
✔ Practice electronic configurations daily.
✔ Understand differences between models.
✔ Memorize formulas and principles.
✔ Revise orbital shapes and capacities.
Value-Based Questions
- Why is scientific experimentation important?
Answer: It helps discover truths and correct wrong beliefs. - What does Rutherford’s experiment teach about curiosity?
Answer: Curiosity leads to major discoveries. - Why should we accept scientific changes?
Answer: Science evolves with evidence. - What value do atomic models reflect?
Answer: Continuous improvement and innovation. - How does studying atoms help society?
Answer: Leads to advancements in medicine, energy, and technology.
Conclusion
The chapter Structure of an Atom is one of the most fundamental topics in chemistry and forms the backbone for understanding advanced chemical concepts. It explains how scientific discoveries transformed the idea of atoms from indivisible particles to complex systems composed of subatomic particles.
Through experiments conducted by scientists, the internal structure of atoms became clearer. The discovery of electrons, protons, and neutrons revealed that atoms are divisible and contain charged particles arranged in a specific manner. The development of atomic models—from Thomson’s model to Rutherford’s nuclear model and Bohr’s quantized orbits—demonstrates how scientific understanding evolves with new evidence.
The Structure of an Atom also introduces the revolutionary quantum mechanical model, which explains electron behavior using probability rather than fixed paths. Concepts such as orbitals, quantum numbers, and electronic configuration help describe how electrons are arranged around the nucleus. These ideas are essential for understanding chemical bonding, periodic trends, and the properties of elements.
Learning the Structure of an Atom helps students understand why elements behave differently and how chemical reactions occur. Knowledge of atomic number, mass number, isotopes, and electronic configurations is crucial not only for school exams but also for competitive examinations.
Moreover, this chapter highlights the importance of scientific thinking, experimentation, and acceptance of new ideas. Each atomic model improved upon the previous one, showing that science is a continuous process of discovery and refinement.
A strong grasp of the Structure of an Atom makes it easier to study future topics such as chemical bonding, periodic classification, thermodynamics, and organic chemistry. Students should focus on understanding concepts rather than memorizing facts, practice electronic configurations, and revise quantum number rules regularly.
In summary, the Structure of an Atom provides the foundation for modern chemistry and helps explain the behavior of matter at the microscopic level. Mastering this chapter ensures conceptual clarity, better exam performance, and a deeper appreciation of the scientific world.
Long Question Answer ✨
1. Explain the cathode ray discharge tube experiment and the discovery of the electron.
Answer:
J. J. Thomson performed the cathode ray discharge tube experiment using a sealed glass tube fitted with electrodes and containing gas at low pressure. When high voltage was applied, rays emerged from the cathode and moved toward the anode.
Observations: • Rays traveled in straight lines.
• They were deflected toward the positive plate, showing negative charge.
• Their properties were independent of the gas used.
Conclusion:
Cathode rays consist of negatively charged particles called electrons, present in all atoms.
2. Describe Rutherford’s gold foil experiment and its conclusions.
Answer:
Ernest Rutherford bombarded a thin gold foil with alpha particles.
Observations: • Most particles passed straight through.
• Some were deflected.
• A few bounced back.
Conclusions: • Atom contains a tiny dense nucleus.
• Nucleus is positively charged.
• Most of the atom is empty space.
• Electrons revolve around the nucleus.
3. Explain Rutherford’s atomic model and its limitations.
Answer:
Rutherford proposed that electrons revolve around a central positively charged nucleus.
Limitations: • Could not explain stability of atom.
• According to classical physics, revolving electrons should lose energy and collapse into nucleus.
• Failed to explain line spectra of atoms.
4. State Bohr’s atomic model and its postulates.
Answer:
Niels Bohr proposed the following:
• Electrons revolve in fixed circular orbits.
• Each orbit has fixed energy.
• Energy is absorbed/emitted when electrons jump between orbits.
• Angular momentum is quantized.
Energy formula:
Eₙ = −2.18 × 10⁻¹⁸ (Z²/n²) J
5. Discuss the successes and limitations of Bohr’s model.
Successes: • Explained hydrogen spectrum.
• Explained atomic stability.
• Introduced energy levels.
Limitations: • Failed for multi-electron atoms.
• Could not explain Zeeman effect and Stark effect.
• Could not explain electron wave nature.
6. Explain the dual nature of matter proposed by de Broglie.
Answer:
Louis de Broglie suggested that particles like electrons exhibit wave properties.
Equation:
λ = h / mv
Where
λ = wavelength
h = Planck’s constant
m = mass
v = velocity
This explains electron diffraction and wave behavior.
7. State and explain Heisenberg’s Uncertainty Principle.
Answer:
Werner Heisenberg stated that it is impossible to determine both position and momentum of an electron simultaneously.
Δx × Δp ≥ h/4π
This means electrons do not travel in fixed paths, leading to the concept of orbitals.
8. Describe the quantum mechanical model of the atom.
Answer:
This modern model describes electrons using probability.
• Developed by Schrödinger.
• Electrons exist in orbitals.
• Orbitals represent probability regions.
• Exact path of electron cannot be predicted.
This model best explains atomic structure.
9. What are orbitals? Describe different types of orbitals.
Answer:
Orbitals are regions where the probability of finding an electron is maximum.
Types:
s orbital: spherical, holds 2 electrons
p orbital: dumbbell shaped, holds 6 electrons
d orbital: cloverleaf, holds 10 electrons
f orbital: complex, holds 14 electrons
10. Explain quantum numbers and their significance.
Answer:
Quantum numbers describe electron location and energy.
- Principal quantum number (n): shell number & energy level
- Azimuthal quantum number (l): subshell & shape
- Magnetic quantum number (m): orbital orientation
- Spin quantum number (s): electron spin (+½ or −½)
They uniquely identify an electron.
11. Explain the Aufbau principle, Pauli Exclusion Principle, and Hund’s rule.
Answer:
Aufbau Principle:
Electrons fill lowest energy orbitals first.
Pauli Exclusion Principle:
No two electrons have same four quantum numbers; one orbital holds max 2 electrons with opposite spins.
Hund’s Rule:
Electrons fill degenerate orbitals singly before pairing.
12. Write a note on electronic configuration with examples.
Answer:
Electronic configuration describes distribution of electrons in orbitals.
Example:
Carbon (6) → 1s² 2s² 2p²
Oxygen (8) → 1s² 2s² 2p⁴
Sodium (11) → 1s² 2s² 2p⁶ 3s¹
It helps predict chemical behavior.
13. Define atomic number and mass number. Explain isotopes with examples.
Answer:
Atomic number (Z): number of protons
Mass number (A): protons + neutrons
Isotopes: atoms with same atomic number but different mass numbers.
Example: Hydrogen isotopes
¹H, ²H, ³H
They have similar chemical properties but different physical properties.
14. Differentiate between orbit and orbital.
Answer:
| Orbit | Orbital |
|---|---|
| Fixed path | Probability region |
| 2D circular path | 3D region |
| Proposed by Bohr | Quantum mechanical model |
| Exact position known | Position uncertain |
15. Explain the shapes and capacities of different subshells.
Answer:
| Subshell | Shape | Orbitals | Max Electrons |
|---|---|---|---|
| s | spherical | 1 | 2 |
| p | dumbbell | 3 | 6 |
| d | cloverleaf | 5 | 10 |
| f | complex | 7 | 14 |
The shape influences bonding and molecular geometry.
Assertion Reason 💫
Directions:
Choose the correct option:
A. Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A.
B. Both A and R are true, but R is not the correct explanation of A.
C. Assertion is true, but Reason is false.
D. Assertion is false, but Reason is true.
1.
Assertion (A): Electrons revolve around the nucleus in fixed energy levels.
Reason (R): Energy of electrons is quantized.
Answer: A
Energy levels are fixed because electron energy is quantized.
2.
Assertion (A): Rutherford’s model could not explain atomic stability.
Reason (R): Revolving electrons should lose energy and fall into the nucleus.
Answer: A
3.
Assertion (A): Isotopes have identical chemical properties.
Reason (R): They have the same atomic number.
Answer: A
4.
Assertion (A): Maximum number of electrons in p subshell is six.
Reason (R): A p subshell has three orbitals.
Answer: A
5.
Assertion (A): No two electrons in an atom can have the same set of four quantum numbers.
Reason (R): Electrons in the same orbital must have opposite spins.
Answer: A
6.
Assertion (A): The exact position of an electron cannot be determined.
Reason (R): Electrons exhibit wave nature.
Answer: B
Both are true, but uncertainty principle explains position limitation.
7.
Assertion (A): s-orbital is spherical in shape.
Reason (R): Its angular momentum quantum number (l) is zero.
Answer: A
8.
Assertion (A): Bohr’s model explains the spectrum of hydrogen atom.
Reason (R): Hydrogen has only one electron.
Answer: A
9.
Assertion (A): The mass of an atom is concentrated in the nucleus.
Reason (R): Protons and neutrons are present in the nucleus.
Answer: A
10.
Assertion (A): According to Hund’s rule, pairing of electrons occurs only after all orbitals are singly filled.
Reason (R): Half-filled orbitals provide extra stability.
Answer: A
Cased Based Questions 💫
Below are 15 Case-Based Questions from Class 11 Chemistry – Structure of an Atom. These questions test understanding, reasoning, and application of concepts, as required in board and competitive exams.
Case 1: Cathode Ray Experiment
A student performs a cathode ray discharge tube experiment. The rays travel in straight lines and are deflected toward the positive plate.
Questions:
- What are cathode rays made of?
- What does deflection toward the positive plate indicate?
- Name the scientist who discovered these particles.
Answers:
- Electrons
- They carry negative charge
- J. J. Thomson
Case 2: Gold Foil Experiment
In an experiment, most alpha particles pass through gold foil, some deflect, and a few bounce back.
Questions:
- What does this suggest about atomic structure?
- Which part of atom causes deflection?
- Name the scientist who conducted the experiment.
Answers:
- Atom is mostly empty space
- Positively charged nucleus
- Ernest Rutherford
Case 3: Hydrogen Spectrum
A hydrogen atom emits light of specific wavelengths when an electron jumps from higher to lower energy levels.
Questions:
- What causes emission of light?
- Which model explains this phenomenon?
- What happens to energy during electron transition?
Answers:
- Electron transition between energy levels
- Niels Bohr model
- Energy is released as radiation
Case 4: Atomic Number & Mass Number
An atom has atomic number 17 and mass number 35.
Questions:
- Number of protons?
- Number of neutrons?
- Number of electrons in neutral atom?
Answers:
- 17
- 18
- 17
Case 5: Isotopes
Two atoms have atomic number 6 but mass numbers 12 and 14.
Questions:
- What are such atoms called?
- Do they have same chemical properties?
- Which property differs significantly?
Answers:
- Isotopes
- Yes
- Physical properties (mass, density)
Case 6: Quantum Numbers
An electron has quantum numbers n=2, l=1.
Questions:
- Which subshell does it belong to?
- How many orbitals are present in this subshell?
- Maximum electrons in this subshell?
Answers:
- p subshell
- 3
- 6
Case 7: Orbital Shape
A student observes an orbital shaped like a dumbbell.
Questions:
- Identify the orbital.
- Maximum electrons it can hold?
- How many such orbitals exist in one subshell?
Answers:
- p orbital
- 2
- 3
Case 8: Electronic Configuration
An element has electronic configuration 1s² 2s² 2p⁶ 3s¹.
Questions:
- Identify the element.
- Atomic number of the element?
- Valence electrons?
Answers:
- Sodium
- 11
- 1
Case 9: Hund’s Rule
Electrons occupy three p orbitals singly before pairing.
Questions:
- Name the rule.
- Why does this occur?
- What stability is achieved?
Answers:
- Hund’s rule
- To minimize repulsion
- Half-filled stability
Case 10: Heisenberg Principle
A scientist states that the position and momentum of an electron cannot be known simultaneously.
Questions:
- Name the principle.
- What does it imply about electron motion?
- Does electron follow a fixed path?
Answers:
- Heisenberg Uncertainty Principle
- Electron position is probabilistic
- No
Case 11: Dual Nature of Electron
Electrons show diffraction patterns in experiments.
Questions:
- What property does this demonstrate?
- Who proposed this concept?
- Write the relation between wavelength and momentum.
Answers:
- Wave nature
- Louis de Broglie
- λ = h/mv
Case 12: Subshell Capacity
A subshell has five orbitals.
Questions:
- Identify the subshell.
- Maximum electrons it can hold.
- Name its general shape.
Answers:
- d subshell
- 10
- Cloverleaf
Case 13: Atomic Stability
An electron jumps from n=3 to n=2.
Questions:
- What happens to energy?
- What type of spectrum is observed?
- Is energy absorbed or emitted?
Answers:
- Energy decreases
- Emission spectrum
- Emitted
Case 14: Spin Quantum Number
Two electrons occupy the same orbital.
Questions:
- What must be their spins?
- Which principle governs this rule?
- Maximum electrons per orbital?
Answers:
- Opposite spins
- Pauli Exclusion Principle
- 2
Case 15: Empty Space in Atom
An atom’s nucleus occupies very little volume compared to the whole atom.
Questions:
- Which experiment proved this?
- What occupies most of the atom’s volume?
- Where is most of the mass concentrated?
Answers:
- Rutherford gold foil experiment
- Empty space with electron movement
- Nucleus
Below is a Sample Question Paper for Class 11 Chemistry – Structure of an Atom, designed according to school and competitive exam patterns.
Sample Question Paper
Class 11 Chemistry
Chapter: Structure of an Atom
Time: 3 Hours
Maximum Marks: 70
Section A – Multiple Choice Questions (1 × 10 = 10 marks)
Choose the correct option:
- Electron was discovered by:
a) Rutherford
b) Chadwick
c) Thomson
d) Bohr - Charge on proton is:
a) −1
b) 0
c) +1
d) +2 - The nucleus of an atom was discovered by:
a) J. J. Thomson
b) Ernest Rutherford
c) Niels Bohr
d) Chadwick - Maximum electrons in p subshell:
a) 2
b) 6
c) 8
d) 10 - Shape of s orbital is:
a) Dumbbell
b) Spherical
c) Cloverleaf
d) Linear - Atomic number represents number of:
a) Neutrons
b) Protons
c) Electrons + neutrons
d) Nucleons - According to Bohr, energy levels are:
a) Continuous
b) Negative
c) Quantized
d) Random - Which particle has no charge?
a) Electron
b) Proton
c) Neutron
d) Positron - Total orbitals in p subshell:
a) 1
b) 2
c) 3
d) 5 - Pauli Exclusion Principle states that:
a) Orbitals fill singly first
b) Electrons move in circles
c) No two electrons have same quantum numbers
d) Energy levels are fixed
Section B – Very Short Answer (2 × 10 = 20 marks)
- Define atomic number.
- What is an orbital?
- Who discovered neutron?
- Write the formula for mass number.
- Define isotope.
- What is principal quantum number?
- Maximum electrons in d subshell?
- State Hund’s rule.
- What is the charge on electron?
- Define electronic configuration.
Section C – Short Answer Questions (3 × 8 = 24 marks)
- Explain Rutherford’s model of atom.
- Write differences between orbit and orbital.
- Explain Bohr’s postulates.
- Define quantum numbers and list their types.
- What are isotopes? Give two examples.
- Explain Pauli Excretion Principle.
- Write electronic configuration of oxygen.
- State Heisenberg Uncertainty Principle.
Section D – Long Answer Questions (4 × 4 = 16 marks)
- Explain the cathode ray experiment and its conclusions.
- Describe Bohr’s atomic model and its limitations.
- Explain the quantum mechanical model of atom.
- Write notes on subshells and shapes of orbitals.
Internal Choice (Optional)
(Attempt any ONE)
A. Explain Aufbau principle with examples.
OR
B. Define isotopes and isobars with differences.
End of Question Paper
Solution ✨
Section A – Multiple Choice Questions
- Electron was discovered by:
Answer: c) Thomson - Charge on proton is:
Answer: c) +1 - The nucleus of an atom was discovered by:
Answer: b) Ernest Rutherford - Maximum electrons in p subshell:
Answer: b) 6 - Shape of s orbital is:
Answer: b) Spherical - Atomic number represents number of:
Answer: b) Protons - According to Bohr, energy levels are:
Answer: c) Quantized - Which particle has no charge?
Answer: c) Neutron - Total orbitals in p subshell:
Answer: c) 3 - Pauli Exclusion Principle states that:
Answer: c) No two electrons have same quantum numbers
Section B – Very Short Answers
- Atomic number:
Number of protons present in the nucleus. - Orbital:
A region around the nucleus where the probability of finding an electron is maximum. - Who discovered neutron?
James Chadwick. - Mass number formula:
Mass number = Protons + Neutrons - Isotope:
Atoms having same atomic number but different mass numbers. - Principal quantum number (n):
It represents the shell or energy level of an electron. - Maximum electrons in d subshell:
10 - Hund’s Rule:
Electrons occupy orbitals singly before pairing. - Charge on electron:
−1 - Electronic configuration:
Distribution of electrons in different orbitals of an atom.
Section C – Short Answer Questions
21. Rutherford’s Model of Atom
Rutherford proposed that: • Atom has a tiny, dense, positively charged nucleus.
• Electrons revolve around the nucleus.
• Most of the atom is empty space.
22. Difference Between Orbit and Orbital
| Orbit | Orbital |
|---|---|
| Fixed circular path | Probability region |
| 2D path | 3D region |
| Proposed by Bohr | Quantum mechanical model |
23. Bohr’s Postulates
Proposed by Niels Bohr:
• Electrons revolve in fixed orbits.
• Energy levels are quantized.
• Energy emitted/absorbed during transitions.
24. Quantum Numbers
Quantum numbers describe electrons.
Types: • Principal (n) → shell
• Azimuthal (l) → subshell
• Magnetic (m) → orientation
• Spin (s) → spin direction
25. Isotopes
Atoms with same atomic number but different mass numbers.
Examples:
Hydrogen: ¹H, ²H, ³H
26. Pauli Exclusion Principle
No two electrons in an atom have the same four quantum numbers.
Each orbital can hold maximum two electrons with opposite spins.
27. Electronic Configuration of Oxygen (Z=8)
1s² 2s² 2p⁴
28. Heisenberg Uncertainty Principle
It is impossible to determine exact position and momentum of an electron simultaneously.
Section D – Long Answer Questions
29. Cathode Ray Experiment
Performed by J. J. Thomson using a discharge tube.
Observations: • Rays travel straight.
• Deflected by electric field.
• Independent of gas used.
Conclusion:
Cathode rays are electrons (negatively charged particles).
30. Bohr’s Atomic Model & Limitations
Postulates: • Fixed orbits
• Quantized energy
• Energy emitted during transitions
Limitations: • Fails for multi-electron atoms
• Cannot explain fine spectral lines
• Ignores wave nature of electrons
31. Quantum Mechanical Model of Atom
Developed by Schrödinger.
• Electrons exist in orbitals.
• Orbitals represent probability regions.
• Exact path cannot be predicted.
• Based on wave mechanics.
This model best explains atomic structure.
32. Subshells & Shapes of Orbitals
| Subshell | Shape | Max Electrons |
|---|---|---|
| s | spherical | 2 |
| p | dumbbell | 6 |
| d | cloverleaf | 10 |
| f | complex | 14 |
Internal Choice
Aufbau Principle
Electrons fill orbitals in order of increasing energy.
Order: 1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p
Example: Sodium = 1s² 2s² 2p⁶ 3s¹
Isotopes vs Isobars
| Property | Isotopes | Isobars |
|---|---|---|
| Atomic number | Same | Different |
| Mass number | Different | Same |
| Element | Same | Different |
Additional Sample paper 📜
Below is an Additional Sample Question Paper for Class 11 Chemistry – Structure of an Atom, designed with a fresh pattern for revision, practice tests, and school examinations.
Additional Sample Question Paper
Class 11 Chemistry
Chapter: Structure of an Atom
Time: 3 Hours
Maximum Marks: 70
Section A – MCQs (1 × 15 = 15 Marks)
Choose the correct answer:
- The charge-to-mass ratio of electron was determined by:
a) Ernest Rutherford
b) J. J. Thomson
c) Niels Bohr
d) Chadwick - Canal rays consist of:
a) Electrons
b) Protons
c) Neutrons
d) Photons - Number of neutrons in ²³Na is:
a) 11
b) 12
c) 13
d) 23 - Which atomic model proposed a dense nucleus?
a) Thomson
b) Dalton
c) Rutherford
d) Bohr - Maximum electrons in n = 3 shell:
a) 8
b) 18
c) 32
d) 2 - Which subshell has highest energy in n=3?
a) 3s
b) 3p
c) 3d
d) All equal - Shape of d orbital:
a) Spherical
b) Dumbbell
c) Cloverleaf
d) Linear - The quantum number that determines orbital shape is:
a) n
b) l
c) m
d) s - How many orbitals are present in d subshell?
a) 3
b) 5
c) 7
d) 1 - Spin quantum number values are:
a) 0 and 1
b) +1 and −1
c) +½ and −½
d) 1 and 2 - Energy of electron depends on:
a) nucleus
b) orbit number
c) temperature
d) pressure - Which rule explains half-filled stability?
a) Aufbau principle
b) Hund’s rule
c) Pauli principle
d) Heisenberg principle - Atomic number of element with configuration 2,8,1 is:
a) 8
b) 10
c) 11
d) 12 - Orbitals were introduced in:
a) Bohr model
b) Quantum mechanical model
c) Rutherford model
d) Thomson model - Isobars have:
a) same atomic number
b) same mass number
c) same electrons
d) same element
Section B – Very Short Answer (2 × 10 = 20 Marks)
- Define cathode rays.
- Write the value of Planck’s constant.
- What is meant by ground state?
- Define atomic mass unit (amu).
- What is an emission spectrum?
- Define subshell.
- What is degenerate orbital?
- State Pauli Exclusion Principle.
- Maximum electrons in f subshell?
- Define ion.
Section C – Short Answer Questions (3 × 7 = 21 Marks)
- Explain Goldstein’s canal ray experiment.
- Write differences between isotopes and isobars.
- Explain dual nature of electron.
- Describe shapes of s and p orbitals.
- Write electronic configuration of magnesium (Z = 12).
- Explain Heisenberg Uncertainty Principle.
- State Aufbau principle and give order of filling.
Section D – Case-Based / Long Answer (7 × 2 = 14 Marks)
33. Case Study: Atomic Structure
During an experiment, scientists observed that electrons behave both like particles and waves.
Questions: a) Who proposed this concept?
b) Write the equation relating wavelength and momentum.
c) What does this prove about matter?
34. Case Study: Electronic Arrangement
An element has atomic number 16.
Questions: a) Write its electronic configuration.
b) How many valence electrons does it have?
c) To which period does it belong?
d) What type of ion will it form?
Internal Choice (Attempt any ONE)
A. Explain Bohr’s model with its limitations.
OR
B. Explain quantum numbers and their significance.
End of Additional Sample Paper
Complete Solution
Structure of an Atom – Class 11 Chemistry
Section A – MCQs
- Charge-to-mass ratio of electron determined by:
Answer: b) J. J. Thomson - Canal rays consist of:
Answer: b) Protons - Number of neutrons in ²³Na:
Mass number – atomic number = 23 − 11 = 12
Answer: b) 12 - Model proposing dense nucleus:
Answer: c) Rutherford - Maximum electrons in n = 3 shell:
Formula = 2n² = 2(3²) = 18
Answer: b) 18 - Highest energy subshell in n=3:
Answer: c) 3d - Shape of d orbital:
Answer: c) Cloverleaf - Quantum number determining shape:
Answer: b) l (azimuthal quantum number) - Orbitals in d subshell:
Answer: b) 5 - Spin quantum number values:
Answer: c) +½ and −½ - Electron energy depends on:
Answer: b) orbit number - Rule explaining half-filled stability:
Answer: b) Hund’s rule - Atomic number of configuration 2,8,1:
Total electrons = 11
Answer: c) 11 - Orbitals introduced in:
Answer: b) Quantum mechanical model - Isobars have:
Answer: b) same mass number
Section B – Very Short Answers
- Cathode rays:
Streams of negatively charged particles (electrons) emitted from the cathode. - Planck’s constant:
6.626 × 10⁻³⁴ Js - Ground state:
Lowest energy state of an electron. - Atomic mass unit (amu):
1 amu = 1/12 the mass of a carbon-12 atom. - Emission spectrum:
Spectrum produced when excited electrons return to lower energy levels emitting light. - Subshell:
Subdivision of a shell consisting of orbitals (s, p, d, f). - Degenerate orbitals:
Orbitals having equal energy (e.g., three p orbitals). - Pauli Exclusion Principle:
No two electrons have the same set of four quantum numbers. - Maximum electrons in f subshell:
14 - Ion:
An atom or group of atoms carrying a charge due to loss or gain of electrons.
Section C – Short Answer Questions
26. Goldstein’s Canal Ray Experiment
Goldstein used a discharge tube with a perforated cathode. He observed rays moving opposite to cathode rays.
Conclusion:
These positively charged rays led to the discovery of protons.
27. Difference Between Isotopes and Isobars
| Property | Isotopes | Isobars |
|---|---|---|
| Atomic number | Same | Different |
| Mass number | Different | Same |
| Element | Same | Different |
28. Dual Nature of Electron
Proposed by Louis de Broglie.
Electrons behave as: • particles (mass & charge)
• waves (diffraction & interference)
λ = h/mv
29. Shapes of s and p Orbitals
• s orbital: spherical shape
• p orbital: dumbbell shape with three orientations (px, py, pz)
30. Electronic Configuration of Magnesium (Z=12)
1s² 2s² 2p⁶ 3s²
31. Heisenberg Uncertainty Principle
It is impossible to determine both exact position and momentum of an electron simultaneously.
This led to the concept of orbitals.
32. Aufbau Principle
Electrons fill orbitals in increasing energy order.
Order:
1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p
Section D – Case-Based Answers
33. Dual Nature Case
a) Proposed by Louis de Broglie
b) λ = h/mv
c) Matter exhibits both wave and particle nature.
34. Atomic Number 16
a) Electronic configuration:
1s² 2s² 2p⁶ 3s² 3p⁴
b) Valence electrons: 6
c) Period: 3
d) Forms ion: S²⁻ (gains 2 electrons)
Internal Choice
A. Bohr’s Model & Limitations
Postulates: • Electrons revolve in fixed orbits.
• Energy levels are quantized.
• Radiation emitted during transitions.
Limitations: • Not valid for multi-electron atoms
• Cannot explain fine spectral lines
• Ignores wave nature of electrons
B. Quantum Numbers & Significance
Quantum numbers describe electron properties:
• n → energy level
• l → subshell & shape
• m → orientation
• s → spin
They help determine electron arrangement.
Final Note
This solution provides step-by-step answers for complete understanding and revision of Structure of an Atom.
Multiple Choice Question ✨
1.
The smallest particle of an element that retains its properties is:
a) Molecule
b) Atom
c) Ion
d) Electron
Answer: b) Atom
2.
Who proposed the plum pudding model of atom?
a) Dalton
b) J. J. Thomson
c) Ernest Rutherford
d) Niels Bohr
Answer: b) Thomson
3.
The number of protons in an atom determines its:
a) Mass number
b) Atomic number
c) Isotope
d) Density
Answer: b) Atomic number
4.
Which particle was discovered by Chadwick?
a) Proton
b) Electron
c) Neutron
d) Positron
Answer: c) Neutron
5.
Which radiation is deflected by electric and magnetic fields?
a) Alpha rays
b) Cathode rays
c) Gamma rays
d) X-rays
Answer: b) Cathode rays
6.
The mass of electron is approximately:
a) 1/1836 of proton
b) Equal to proton
c) 1/2 of proton
d) Twice proton
Answer: a) 1/1836 of proton
7.
The nucleus of hydrogen atom contains:
a) One proton only
b) One neutron only
c) One proton and one neutron
d) One electron
Answer: a) One proton only
8.
Atomic mass is mainly due to:
a) Electrons
b) Protons only
c) Neutrons only
d) Protons and neutrons
Answer: d) Protons and neutrons
9.
The value of Planck’s constant is:
a) 6.626 × 10⁻³⁴ Js
b) 3 × 10⁸ m/s
c) 9.8 m/s²
d) 1.6 × 10⁻¹⁹ C
Answer: a) 6.626 × 10⁻³⁴ Js
10.
Bohr’s model is applicable to:
a) All atoms
b) Hydrogen atom
c) Molecules
d) Solids
Answer: b) Hydrogen atom
11.
Energy of electron in an atom is:
a) Continuous
b) Quantized
c) Infinite
d) Zero
Answer: b) Quantized
12.
Which quantum number defines size of orbital?
a) n
b) l
c) m
d) s
Answer: a) n
13.
Which quantum number defines orientation of orbital?
a) n
b) l
c) m
d) s
Answer: c) m
14.
Total number of subshells in n = 4 shell:
a) 2
b) 3
c) 4
d) 5
Answer: c) 4
15.
Maximum number of electrons in n = 2 shell:
a) 2
b) 8
c) 18
d) 32
Answer: b) 8
16.
Which of the following has zero mass?
a) Electron
b) Proton
c) Neutron
d) Photon
Answer: d) Photon
17.
The spectral lines of hydrogen are explained by:
a) Thomson model
b) Rutherford model
c) Bohr model
d) Dalton model
Answer: c) Bohr model
18.
Which series lies in visible region of hydrogen spectrum?
a) Lyman
b) Balmer
c) Paschen
d) Brackett
Answer: b) Balmer
19.
Wave nature of electron was proposed by:
a) Einstein
b) Bohr
c) Louis de Broglie
d) Dalton
Answer: c) Louis de Broglie
20.
The uncertainty principle was given by:
a) Heisenberg
b) Bohr
c) Rutherford
d) Maxwell
Answer: a) Heisenberg
21.
An orbital can accommodate maximum:
a) 1 electron
b) 2 electrons
c) 3 electrons
d) 4 electrons
Answer: b) 2 electrons
22.
Which subshell has highest energy in the same shell?
a) s
b) p
c) d
d) f
Answer: d) f
23.
Number of p orbitals present in one subshell:
a) 1
b) 2
c) 3
d) 5
Answer: c) 3
24.
Which rule states electrons fill orbitals singly first?
a) Pauli principle
b) Aufbau principle
c) Hund’s rule
d) Heisenberg principle
Answer: c) Hund’s rule
25.
Electronic configuration of Neon (Z=10) is:
a) 1s² 2s² 2p⁴
b) 1s² 2s² 2p⁶
c) 1s² 2s² 2p⁵
d) 1s² 2s¹ 2p⁷
Answer: b) 1s² 2s² 2p⁶
26.
The shape of p orbital is:
a) spherical
b) dumbbell
c) linear
d) circular
Answer: b) dumbbell
27.
Which of the following is not a quantum number?
a) Principal
b) Azimuthal
c) Magnetic
d) Orbital radius
Answer: d) Orbital radius
28.
Number of electrons in 3p subshell:
a) 2
b) 3
c) 6
d) 10
Answer: c) 6
29.
If l = 2, the subshell is:
a) s
b) p
c) d
d) f
Answer: c) d
30.
Atoms having same mass number but different atomic numbers are called:
a) Isotopes
b) Isobars
c) Isotones
d) Isoelectronic species
Answer: b) Isobars
Here’s a one-page, 800-word revision sheet for Class 11 Chemistry – Structure of an Atom, perfect for quick exam preparation and last-minute revision. I’ve kept it student-friendly, exam-oriented, and SEO-ready.
Structure of an Atom – Quick Revision Sheet
1. Atomic Theory Overview
- Atom: Smallest particle of an element retaining its chemical properties.
- Dalton’s Atomic Theory (1803): Atoms are indivisible, identical in mass for a given element, and combine in simple ratios to form compounds.
- Discovery of Electron: J. J. Thomson (1897) used cathode ray tube; electron is negatively charged, mass ≈ 1/1836 of proton.
- Proton: Positively charged particle in nucleus, discovered via canal ray experiment.
- Neutron: Neutral particle in nucleus, discovered by James Chadwick.
2. Atomic Models
2.1 Thomson’s Model
- “Plum pudding” model: atom is a positively charged sphere with embedded electrons.
- Could not explain scattering of alpha particles.
2.2 Rutherford’s Model (1911)
- Gold foil experiment: α-particles mostly pass through, some deflect.
- Conclusions: Atom has tiny, dense, positively charged nucleus; electrons revolve around nucleus; most of atom is empty space.
- Limitation: Could not explain atomic stability and spectral lines.
2.3 Bohr’s Model (1913)
- Electrons revolve in fixed circular orbits with quantized energy levels.
- Postulates:
- Electrons move in stable orbits without radiating energy.
- Energy is absorbed/emitted when electrons jump orbits.
- Angular momentum is quantized: L = nħ.
- Energy of electron:
E_n = -2.18 \times 10^{-18} \frac{Z^2}{n^2} \text{J}
- Limitation: Failed for multi-electron atoms; ignored wave nature.
2.4 Quantum Mechanical Model
- Developed by Schrödinger using wave equation: describes electron as a wave.
- Electrons occupy orbitals, regions with maximum probability of finding an electron.
- Incorporates Heisenberg Uncertainty Principle:
\Delta x \cdot \Delta p \geq \frac{h}{4\pi}
3. Subatomic Particles
| Particle | Symbol | Charge | Mass (kg) |
|---|---|---|---|
| Electron | e⁻ | −1 | 9.11 × 10⁻³¹ |
| Proton | p⁺ | +1 | 1.673 × 10⁻²⁷ |
| Neutron | n⁰ | 0 | 1.675 × 10⁻²⁷ |
4. Atomic Numbers, Mass Numbers & Isotopes
- Atomic Number (Z): Number of protons = number of electrons in neutral atom.
- Mass Number (A): Protons + Neutrons.
- Isotopes: Same Z, different A (e.g., ¹H, ²H, ³H).
- Isobars: Same A, different Z.
5. Quantum Numbers
- Principal (n): Shell/energy level, n = 1,2,3…
- Azimuthal (l): Subshell shape, l = 0 to n−1 (s=0, p=1, d=2, f=3).
- Magnetic (m): Orientation of orbital, m = −l to +l.
- Spin (s): Electron spin, +½ or −½.
Rules for Electron Arrangement:
- Aufbau Principle: Fill orbitals of lowest energy first.
- Pauli Exclusion Principle: Max 2 electrons per orbital, opposite spins.
- Hund’s Rule: Single occupancy of degenerate orbitals before pairing.
6. Electron Configuration Examples
| Element | Atomic Number | Configuration |
|---|---|---|
| Hydrogen | 1 | 1s¹ |
| Oxygen | 8 | 1s² 2s² 2p⁴ |
| Sodium | 11 | 1s² 2s² 2p⁶ 3s¹ |
| Magnesium | 12 | 1s² 2s² 2p⁶ 3s² |
Valence electrons: Electrons in outermost shell; determine chemical reactivity.
7. Shapes of Orbitals
| Orbital | Shape | Max Electrons |
|---|---|---|
| s | Spherical | 2 |
| p | Dumbbell | 6 |
| d | Cloverleaf | 10 |
| f | Complex | 14 |
- Degenerate orbitals: Orbitals of same energy (e.g., 3 px, 3 py, 3 pz).
8. Important Formulas
- Energy of electron in nth orbit: J
- Wavelength of matter wave:
- Maximum electrons in shell: 2n²
9. Important Points for Exam
- Rutherford experiment: α-particles, gold foil, nucleus.
- Bohr model: Hydrogen spectrum, n = 1,2,3…
- Quantum mechanical model: Orbitals, probability regions.
- Isotopes: Same chemical properties, different physical properties.
- Rules: Aufbau, Pauli, Hund’s are critical for electronic configuration.
- Spectra: Lyman (UV), Balmer (visible), Paschen (IR).
10. Tips for Competitive Exams
- Memorize subatomic particles’ properties.
- Write electron configurations quickly using Aufbau order.
- Practice drawing orbital shapes (s, p, d).
- Remember quantum numbers and their significance.
- Solve MCQs and numerical problems on mass number, isotopes, and electron configurations.
- Focus on Bohr’s postulates and limitations for conceptual clarity.
- Revise formulas for energy, wavelength, and maximum electrons.









Leave a Reply